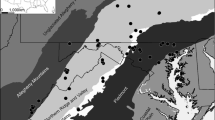
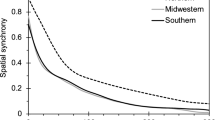
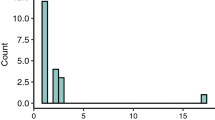
Abstract
The population dynamics and impacts of non-native species often change following their initial establishment, with impacts either increasing or decreasing over time. The reasons why the abundance of an invading species may change are varied but often reflect changes in the way in which populations interact with resident communities. Here we analyze changes in the outbreak dynamics of Lymantria dispar (formerly known to as the “gypsy moth”), a Eurasian foliage-feeding insect that has been established in N. America for ca. 150 years. We find that during the course of this species’ presence in N. America, it has continually exhibited population dynamics in which populations reach outbreak levels, resulting in defoliation of large forested areas. However, there is evidence of some changes in both the periodicity and synchrony of these outbreaks. We hypothesize that the accidental introduction of an entomopathogenic nucleopolyhedrosis virus around 1906 resulted in populations shifting from a pattern of sustained outbreaks to oscillatory dynamics with periodic outbreaks synchronized over large distances. We analyze historical L. dispar population data that provide some evidence in support of this hypothesis. There is also evidence that the more recent establishment of the fungal pathogen Entomophaga maimaiga has caused a decrease in the amplitude of L. dispar outbreaks since its emergence in 1989.





Similar content being viewed by others
References
Abbott KC, Dwyer G (2008) Using mechanistic models to understand synchrony in forest insect populations: the North American gypsy moth as a case study. Am Nat 172:613–624
Allstadt AJ, Haynes KJ, Liebhold AM, Johnson DM (2013) Long-term shifts in the cyclicity of outbreaks of a forest-defoliating insect. Oecologia 172:141–151
Allstadt AJ, Liebhold AM, Johnson DM, Davis RE, Haynes KJ (2015) Temporal variation in the synchrony of weather and its consequences for spatiotemporal population dynamics. Ecology 96:2935–2946
Bjørnstad ON (2000) Cycles and synchrony: two historical ‘experiments’ and one experience. J Anim Ecol 69:869–873
Bjørnstad ON, Falck W (2001) Nonparametric spatial covariance functions: estimation and testing. Environ Ecol Stat 8:53–70
Bjørnstad ON, Liebhold AM, Johnson DM (2008) Transient synchronization following invasion: revisiting Moran’s model and a case study. Pop Ecol 50:379–389
Bjørnstad ON, Robinet C, Liebhold AM (2010) Geographic variation in the North American gypsy moth cycles: sub-harmonics, generalist predators and spatial coupling. Ecology 91:106–118
Blackburn TM, Essl F, Evans T, Hulme PE, Jeschke JM, Kühn I, Kumschick S, Marková Z, Mrugała A, Nentwig W, Pergl J (2014) A unified classification of alien species based on the magnitude of their environmental impacts. PLoS Biol 12:e1001850
Burgess AF, Baker WL (1938) The gypsy and brown-tail moths and their control. US Dept Agric Circ 464
Burgess AF (1944) The gypsy moth and the brown-tail moth - history of the work for prevention of spread and extermination of these insects in North America. 343 p. online at https://naldc.nal.usda.gov/catalog/6124364
Campbell RW (1967) The analysis of numerical change in gypsy moth populations. For Sci Monogr 33:1–34
Clifton EH, Castrillo LA, Gryganskyi A, Hajek AE (2019) A pair of native fungal pathogens drives decline of a new invasive herbivore. PNAS 116:9178–9180
Colautti RI, Ricciardi A, Grigorovich IA, MacIsaac HJ (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733
Dalgleish HJ, Nelson CD, Scrivani JA, Jacobs DF (2015) Consequences of shifts in abundance and distribution of American chestnut for restoration of a foundation forest tree. Forests 7:4
Desurmont GA, Donoghue MJ, Clement WL, Agrawal AA (2011) Evolutionary history predicts plant defense against an invasive pest. PNAS 108:7070–7074
Dwyer G, Dushoff J, Yee SH (2004) The combined effects of pathogens and predators on insect outbreaks. Nature 430:341–345
Elkinton JS, Liebhold AM (1990) Population dynamics of gypsy moth in North America. Ann Rev Entomol 35:571–596
Epanchin-Niell RS, Liebhold AM (2015) Benefits of invasion prevention: effect of time lags, spread rates, and damage persistence. Ecol Econ 116:146–153
Fei S, Morin RS, Oswalt CM, Liebhold AM (2019) Biomass losses resulting from insect and disease invasions in US forests. PNAS 116:17371–17376
Forbush EH, Fernald CH (1896) The gypsy moth. Porthetria dispar (Linn.). The gypsy moth. Porthetria dispar (Linn.). Wright & Potter, Boston
Gandhi KJ, Herms DA (2010) Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol Invas 12:389–405
Glaser RW (1915) Wilt of gypsy-moth caterpillar. J Agric Res 4:101–126
Hajek AE, Eilenberg J (2018) Natural enemies: an introduction to biological control. Cambridge University Press, Cambridge
Hajek AE, Humber RA, Elkinton JS (1995) Mysterious origin of Entomophaga maimaiga in North America. Am Entomol 41:31–43
Hajek AE, Tobin PC, Haynes KJ (2015) Replacement of a dominant viral pathogen by a fungal pathogen does not alter the collapse of a regional forest insect outbreak. Oecologia 177:785–797
Hamed KH, Rao RA (1998) A modified Mann-Kendall trend test for autocorrelated data. J Hydrol 204:182–196
Haynes KJ, Walter JA, Liebhold AM (2019) Population spatial synchrony enhanced by periodicity and low detuning with environmental forcing. Proc R Soc B 286:20182828
Howard LO, Fiske WF (1911) The importation into the United States of the parasites of the gipsy moth and the brown-tail moth: a report of progress, with some consideration of previous and concurrent efforts of this kind. US Dept Agric Bull No. 91. US Government Printing Office, Washington
Jeffries MJ, Lawton JH (1984) Enemy free space and the structure of ecological communities. Biol J Linn Soc 23:269–286
Johnson DM, Liebhold AM, Bjørnstad ON (2006) Geographical variation in the periodicity of gypsy moth outbreaks. Ecography 29:367–374
Johnson DM, Liebhold AM, Bjørnstad ON, McManus ML (2005) Circumpolar variation in periodicity and synchrony among gypsy moth populations. J Anim Ecol 74:882–892
Jones JN (1910) Further studies on the nature of the wilt disease of the gypsy moth Larvae. In: Rane FW (ed) The State Forester of Massachusetts, Seventh Annual Report, 1910. Boston. pp 100–105
Liebhold AM (1992) Are North American populations of gypsy moth (Lepidoptera: Lymantriidae) bimodal? Environ Entomol 21:221–229
Liebhold AM, Simons EE, Sior A, Unger JD (1993) Forecasting defoliation caused by the gypsy moth from field measurements. Environ Entomol 22:26–32
Liebhold AM, Gottschalk KW, Mason DA, Bush RR (1997b) Evaluation of forest susceptibility to the gypsy moth across the conterminous United States. J for 95:20–24
Liebhold AM, Haynes KJ, Bjørnstad ON (2012) Spatial synchrony of insect outbreaks. pages. In: Barbosa P, LeTourneau DK, Agrawal AA (eds) Insect outbreaks revisited. Wiley, New York, pp 113–125
Liebhold A, Thorpe K, Ghent J, Lyons DB (1994) Gypsy moth egg mass sampling for decision-making: a user’s guide. US Dept Agric Forest Service NA-TP-04–94
Liebhold AM, Gottschalk KW, Luzader ER, Bush R, Twardus DB (1997a) Gypsy moth in the United States: an atlas. US Dept Agric Forest Service Gen Tech Rep NE-233
Morin RS, Liebhold AM (2016) Invasive forest defoliator contributes to the impending downward trend of oak dominance in eastern North America. Forestry 89:284–289
Nielsen C, Milgroom MG, Hajek AE (2005) Genetic diversity in the gypsy moth fungal pathogen Entomophaga maimaiga from founder populations in North America and source populations in Asia. Mycol Res 109:941–950
Patakamuri SK, O'Brien N (2019) modifiedmk: Modified Versions of Mann Kendall and Spearman's Rho Trend Tests. R package version 1.3.0. https://CRAN.R-project.org/package=modifiedmk
Reardon RC, Podgwaite JD (1976) Disease-parasitoid relationships in natural populations of Lymantria dispar (Lep.: Lymantriidae) in the northeastern United States. Entomophaga 21:333–341
Reardon RC (1981) Rearing, evaluating, and attempting to establish exotic species and redistributing established species, attempts before 1960. In: Doane CC, McManus ML (eds) The gypsy moth: research toward integrated pest management. US Dept Agric Tech Bull, Washington, pp 348–351
Reiff W (1911) The “wilt disease” or" flacherie" of the gypsy moth. Wright & Potter, Boston
Rogers DM, Burgess AF (1910) Report on the field work against the gipsy moth and the brown-tail moth. US Dept Agrc Bur Entomol Bulletin No 87, Washington
Simberloff D, Gibbons L (2004) Now you see them, now you don’t!–population crashes of established introduced species. Biol Invas 6:161–172
Smith HR, Lautenschlager RA, Doane CC, McManus ML (1981) Gypsy moth predators. In: Doane CC, McManus ML (eds) The gypsy moth: research toward integrated pest management. US Dept Agric Tech Bull, Washington, pp 96–125
Speare AT, Colley RH (1912) The artificial use of the browntail fungus in Massachusetts, with practical suggestions for private experiment, and a brief note on a fungus disease of the gypsy caterpillar. Wright & Potter, Boston
U.S. Forest Service (2020) Gypsy moth digest (online database). https://www.fs.usda.gov/naspf/programs/forest-health-protection/gypsy-moth-digest
Vilà M, Hulme PE (eds) (2017) Impact of biological invasions on ecosystem services. Springer International Publishing, Cham
Weseloh RM (1998) Possibility for recent origin of the gypsy moth (Lepidoptera: Lymantriidae) fungal pathogen Entomophaga maimaiga (Zygomycetes: Entomophthorales) in North America. Environ Entomol 27:171–177
Williams DW, Liebhold AM (1995) Influence of weather on the synchrony of gypsy moth (Lepidoptera: Lymantriidae) outbreaks in New England. Environ Entomol 24:987–995
Williams DW, Fuester RW, Metterhouse WW, Balaam RJ, Bullock RH, Chianesei RJ (1991) Oak defoliation and population density relationships for the gypsy moth (Lepidoptera: Lymantriidae). J Econ Entomol 84:1508–1514
Williamson M (1996) Biological Invasions. Chapman & Hall, London
Woods SA, Elkinton JS, Murray KD, Liebhold AM, Gould JR, Podgwaite JD (1991) Transmission dynamics of a nuclear polyhedrosis virus and predicting mortality in gypsy moth (Lepidoptera: Lymantriidae) populations. J Econ Entomol 84:423–430
Acknowledgements
This work was supported by the USDA Forest Service, The Charles Bullard Fellowship Program at Harvard Forest and Grants from the National Science Foundation Macro systems Biology Program (Grant Numbers 1241932, 1638702) and Grant EVA4.0, No. CZ.02.1.01/0.0/0.0/16_019/0000803 financed by OP RDE. JAW was supported by The Nature Conservancy and NSF grant OAC-1839024. We thank Eugene Luzader for technical assistance. The authors are grateful to reviewers and editors whose comments greatly improved the paper.
Funding
USDA Forest Service, The Charles Bullard Fellowship Program at Harvard Forest, National Science Foundation Macrosystems Biology Program, Czech Ministry of Education Operational Programme for Research, Development and Education, The Nature Conservancy
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors that they have declare no conflict of interest.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Code used during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liebhold, A.M., Hajek, A.E., Walter, J.A. et al. Historical change in the outbreak dynamics of an invading forest insect. Biol Invasions 24, 879–889 (2022). https://doi.org/10.1007/s10530-021-02682-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-021-02682-6