Abstract
Objectives
Different cultivation conditions and parameters were evaluated to improve the production and secretion of a recombinant Phanerochaete chrysosporium lipH8 gene in Komagataella phaffii (Pichia pastoris).
Results
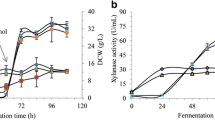
The recombinant lipH8 gene with its native secretion signal was successfully cloned and expressed in Komagataella phaffii (Pichia pastoris) under the control of the alcohol oxidase 1 promoter (PAOX1). The results revealed that co-feeding with sorbitol and methanol increased rLiP secretion by 5.9-fold compared to the control conditions. The addition of 1 mM FeSO4 increased LiP activity a further 6.0-fold during the induction phase. Moreover, the combination of several optimal conditions and parameters yielded an extracellular rLiP activity of 20.05 U l−1, which is more than ten-fold higher relative to standard growth conditions (BMM10 medium, pH 6 and 30 °C).
Conclusion
Extracellular activity of a recombinant LiP expressed in P. pastoris increased more than ten-fold when co-feeding sorbitol and methanol as carbon sources, together with urea as nitrogen source, FeSO4 supplementation, lower pH and lower cultivation temperature.




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahmad M, Hirz M, Pichler H, Schwab H (2014) Protein expression in Pichia pastoris: recent achievements and perspectives for heterologous protein production. Appl Microbiol Biotechnol 98:5301–5317. https://doi.org/10.1007/s00253-014-5732-5
Bankefa OE, Wang M, Zhu T, Li Y (2018) Enhancing the secretion pathway maximizes the effects of mixed feeding strategy for glucose oxidase production in the methylotrophic yeast Pichia pastoris. Bioresour Bioprocess 5:25. https://doi.org/10.1186/s40643-018-0211-y
Biko ODV, Viljoen-Bloom M, van Zyl WH (2020) Microbial lignin peroxidases: applications, production challenges and future perspectives. Enzyme Microb Technol 141:109669. https://doi.org/10.1016/j.enzmictec.2020.109669
Çalik P, Bozkurt B, Zerze GH, Inankur B, Bayraktar E, Boy E, Orman MA, Açik E, Özdamar TH (2013) Effect of co-substrate sorbitol different feeding strategies on human growth hormone production by recombinant Pichia pastoris. J Chem Technol Biotechnol 88:1631–1640. https://doi.org/10.1002/jctb.4011
Chen L, Mohsin A, Chu J, Zhuang Y, Liu Y, Guo M (2017) Enhanced protein production by sorbitol co-feeding with methanol in recombinant Pichia pastoris strains. Biotechnol Bioproc Eng 773:767–773. https://doi.org/10.1007/s12257-017-0011-9
Ciofalo V, Barton N, Kreps J, Coats I, Shanahan D (2006) Safety evaluation of a lipase enzyme preparation, expressed in Pichia pastoris, intended for use in the degumming of edible vegetable oil. Regul Toxicol Pharmacol 45:1–8. https://doi.org/10.1016/j.yrtph.2006.02.001
Cos O, Ramón R, Montesinos JL, Valero F (2006) Operational strategies, monitoring and control of heterologous protein production in the methylotrophic yeast Pichia pastoris under different promoters: a review. Microb Cell Fact 5:17. https://doi.org/10.1186/1475-2859-5-17
Dehnavi E, Ranaei Siadat SO, Fathi Roudsari M, Khajeh K (2016) Cloning and high-level expression of β-xylosidase from Selenomonas ruminantium in Pichia pastoris by optimizing of pH, methanol concentration and temperature conditions. Protein Expr Purif 124:55–61. https://doi.org/10.1016/j.pep.2016.05.001
Doyle WA, Blodig W, Veitch NC, Piontek K, Smith AT (1998) Two substrate interaction sites in lignin peroxidase revealed by site- directed mutagenesis. Biochemistry 37:15097–15105. https://doi.org/10.1021/bi981633h
Fang Z, Xu L, Pan D, Jiao L, Liu Z, Yan Y (2014) Enhanced production of Thermomyces lanuginosus lipase in Pichia pastoris via genetic and fermentation strategies. J Ind Microbiol Biotechnol 41:1541–1551. https://doi.org/10.1007/s10295-014-1491-7
Gasser B, Maurer M, Rautio J, Sauer M, Bhattacharyya A, Saloheimo M, Mattanovich PMD (2007) Monitoring of transcriptional regulation in Pichia pastoris under protein production conditions. BMC Genom 8:179. https://doi.org/10.1186/1471-2164-8-179
Gold MH, Kuwahara M, Chiu AA, Glenn JK (1984) Purification and characterization of an extracellular H2O2-requiring diarylpropane oxygenase from the white rot basidiomycete, Phanerochaete chrysosporium. Arch Biochem Biophys 234:353–362. https://doi.org/10.1016/0003-9861(84)90280-7
Guo C, Huang Y, Zheng H, Tang L, He J, Xiang L, Liu D, Jiang H (2012) Secretion and activity of antimicrobial peptide cecropin D expressed in Pichia pastoris. Exp Ther Med 4:1063–1068. https://doi.org/10.3892/etm.2012.719
He H, Wu S, Mei M, Ning J, Li C, Ma L, Zhang G, Yi L (2020) A combinational strategy for effective heterologous production of functional human lysozyme in Pichia pastoris. Front Bioeng Biotechnol 8:1–12. https://doi.org/10.3389/fbioe.2020.00118
Ilić Đurđić K, Ece S, Ostafe R, Vogel S, Balaž AM, Schillberg S, Fischer R, Prodanović R (2020) Flow cytometry-based system for screening of lignin peroxidase mutants with higher oxidative stability. J Biosc Bioeng 129:664–671. https://doi.org/10.1016/j.jbiosc.2019.12.009
Jayachandran C, Athiyaman BP, Sankaranarayanan M (2017) Formate co-feeding improved Candida antarctica Lipase B activity in Pichia pastoris. Res J Biotechnol 12:29–36
Karbalaei M, Rezaee SA, Farsiani H (2020) Pichia pastoris: a highly successful expression system for optimal synthesis of heterologous proteins. J Cell Physiol 235:5867–5881. https://doi.org/10.1002/jcp.29583
Kaushik N, Lamminmäki U, Khanna N, Batra G (2020) Enhanced cell density cultivation and rapid expression-screening of recombinant Pichia pastoris clones in microscale. Sci Rep 10:1–11. https://doi.org/10.1038/s41598-020-63995-5
Kensy F, Zang E, Faulhammer C, Tan R-K, Büchs J (2009) Validation of a high-throughput fermentation system based on online monitoring of biomass and fluorescence in continuously shaken microtiter plates. Microb Cell Fact 8:1–17. https://doi.org/10.1186/1475-2859-8-31
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Annu Rev Microbiol 1987:465–505
Krahulec J, Šafránek M (2021) Impact of media components from different suppliers on enterokinase productivity in Pichia pastoris. BMC Biotechnol 21:19. https://doi.org/10.1186/s12896-021-00681-y
Krainer FW, Capone S, Jäger M, Vogl T, Gerstmann M, Glieder A, Herwig C, Spadiut O (2015) Optimizing cofactor availability for the production of recombinant heme peroxidase in Pichia pastoris. Microb Cell Fact 14:4. https://doi.org/10.1186/s12934-014-0187-z
Li C, Zhao X, Wang A, Huber GW, Zhang T (2015) Catalytic transformation of lignin for the production of chemicals and fuels. Chem Rev 115:11559–11624. https://doi.org/10.1021/acs.chemrev.5b00155
Lin-Cereghino J, Wong WW, Xiong S, Giang W, Luong LT, Vu J, Johnson SD, Lin-Cereghino GP (2005) Condensed protocol for competent cell preparation and transformation of the methylotrophic yeast Pichia pastoris. Biotechniques 38:44–48. https://doi.org/10.2144/05381BM04
Long Q, Liu X, Yang Y, Li L, Harvey L, McNeil B, Bai Z (2014) The development and application of high throughput cultivation technology in bioprocess development. J Biotechnol 192:323–338. https://doi.org/10.1016/j.jbiotec.2014.03.028
Macauley-Patrick S, Fazenda ML, McNeil B, Harvey LM (2005) Heterologous protein production using the Pichia pastoris expression system. Yeast 22:249–270. https://doi.org/10.1002/yea.1208
Majeke BM, García-Aparicio M, Biko OD, Viljoen-Bloom M, van Zyl WH, Görgens JF (2020) Synergistic codon optimization and bioreactor cultivation toward enhanced secretion of fungal lignin peroxidase in Pichia pastoris: enzymatic valorization of technical (industrial) lignins. Enzyme Microb Technol 139:109593. https://doi.org/10.1016/j.enzmictec.2020.109593
Martínez AT, Ruiz-Dueñas FJ, Camarero S, Serrano A, Linde D, Lund H, Vind J, Tovborg M, Herold-Majumdar OM, Hofrichter M, Liers C, Ulrich R, Scheibner K, Sannia G, Piscitelli A, Pezzella C, Sener ME, Kılıç S, van Berkel WJH, Guallar V, Lucas MF, Zuhse R, Ludwig R, Hollmann F, Fernández-Fueyo E, Record E, Faulds CB, Tortajada M, Winckelmann I, Rasmussen J-A, Gelo-Pujic M, Gutiérrez A, del Río JC, Rencoret J, Alcalde M (2017) Oxidoreductases on their way to industrial biotransformations. Biotechnol Adv 35:815–831. https://doi.org/10.1016/j.biotechadv.2017.06.003
Mombeni M, Arjmand S, Siadat SOR, Alizadeh H, Abbasi A (2020) pMOX: a new powerful promoter for recombinant protein production in yeast Pichia pastoris. Enzyme Microb Technol 139:109582. https://doi.org/10.1016/j.enzmictec.2020.109582
Nobary MB, Maleki A, Ahmadi H, Salmani AS, Nejati M, Norouzian D (2012) Evaluation of pH/buffering conditions effect on the optimization of recombinant human erythropoietin expression in the methylotrophic yeast, Pichia pastoris. J Pharm Health Sci 1:47–55
Noseda DG, Recúpero MN, Blasco M, Ortiz GE, Galvagno MA (2013) Cloning, expression and optimized production in a bioreactor of bovine chymosin B in Pichia (Komagataella) pastoris under AOX1 promoter. Protein Expr Purif 92:235–244. https://doi.org/10.1016/j.pep.2013.08.018
Peña DA, Gasser B, Zanghellini J, Steiger MG, Mattanovich D (2018) Metabolic engineering of Pichia pastoris. Met Eng 50:2–15. https://doi.org/10.1016/j.ymben.2018.04.017
Pla IA, Damasceno LM, Vannelli T, Ritter G, Batt CA, Shuler ML (2006) Evaluation of Mut+ and MutS Pichia pastoris phenotypes for high level extracellular scFv expression under feedback control of the methanol concentration. Biotechnol Prog 22:881–888. https://doi.org/10.1021/bp060012+
Reddy CA, D’Souza TM (1994) Physiology and molecular biology of the lignin peroxidases of Phanerochaete chrysosporium. FEMS Microbiol Rev 13:137–152. https://doi.org/10.1111/j.1574-6976.1994.tb00040.x
Sáez-Jiménez V, Fernández-Fueyo E, Medrano FJ, Romero A, Martínez AT, Ruiz-Dueñas FJ (2015) Improving the pH-stability of versatile peroxidase by comparative structural analysis with a naturally-stable manganese peroxidase. PLoS ONE 10:e0143267. https://doi.org/10.1371/journal.pone.0143267
Safder I, Khan S, Islam I, Ali MK, Bibi Z, Waqas M (2018) Pichia pastoris expression system : a potential candidate to express protein in industrial and biopharmaceutical domains. Biomed Lett 4:1–14
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning a laboratory manual. Cold Spring Harbor, NY
Schwarzhans JP, Luttermann T, Geier M, Kalinowski J, Friehs K (2017) Towards systems metabolic engineering in Pichia pastoris. Biotechnol Adv 35:681–710. https://doi.org/10.1016/j.biotechadv.2017.07.009
Sun FF, Bai R, Yang H, Wang F, He J, Wang C, Tu M (2016) Heterologous expression of codon optimized Trichoderma reesei Cel6A in Pichia pastoris. Enzyme Microb Technol 92:107–116. https://doi.org/10.1016/j.enzmictec.2016.07.004
Tavasoli T, Arjmand S, Siadat SOR, Shojaosadati SA, Lotfi AS (2017) Enhancement of alpha 1-antitrypsin production in Pichia pastoris by designing and optimizing medium using elemental analysis. Iran J Biotechnol 15:224–231. https://doi.org/10.15171/ijb.1808
Wang W, Wen X (2009) Expression of lignin peroxidase H2 from Phanerochaete chrysosporium by multi-copy recombinant Pichia strain. J Environ Sci 21:218–222. https://doi.org/10.1016/S1001-0742(08)62254-8
Weis R, Luiten R, Skranc W, Schwab H, Wubbolts M, Glieder A (2004) Reliable high-throughput screening with Pichia pastoris by limiting yeast cell death phenomena. FEMS Yeast Res 5:179–189. https://doi.org/10.1016/j.femsyr.2004.06.016
Yang Z, Zhang Z (2018) Engineering strategies for enhanced production of protein and bio-products in Pichia pastoris: a review. Biotechnol Adv 36:182–195. https://doi.org/10.1016/j.biotechadv.2017.11.002
Zhou WJ, Yang JK, Mao L, Miao LH (2015) Codon optimization, promoter and expression system selection that achieved high-level production of Yarrowia lipolytica lipase in Pichia pastoris. Enzyme Microb Technol 71:66–72. https://doi.org/10.1016/j.enzmictec.2014.10.007
Zhu W, Xu R, Gong G, Xu L, Hu Y, Xie L (2021) Medium optimization for high yield production of human serum albumin in Pichia pastoris and its efficient purification. Prot Expr Purif 181:105831. https://doi.org/10.1016/j.pep.2021.105831
Acknowledgements
Opinions expressed and conclusions arrived at are those of the authors and are not necessarily to be attributed to the NRF.
Funding
This work was supported by the National Research Foundation (NRF) linked to the Chair of Energy Research: Biofuels and Other Clean Alternative Fuels (Grant 86423 to WHvZ) and the Competitive Programme for Rated Researchers (Grant 118528 to MVB).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ODB. The first draft of the manuscript was written by ODB subsequent revisions done by MVB and WHvZ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Biko, O.D., Viljoen-Bloom, M. & van Zyl, W.H. Medium optimization for enhanced production of recombinant lignin peroxidase in Pichia pastoris. Biotechnol Lett 45, 105–113 (2023). https://doi.org/10.1007/s10529-022-03321-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-022-03321-3