Abstract
Objective
The aim of this study is to evaluate the cytoprotection and potential molecular mechanisms of cyanidin-3-glucoside (C3G) on hydrogen peroxide (H2O2)-induced oxidative damage in HepG2 cells.
Methods
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was carried out to examine the viability of HepG2 cells exposure to H2O2 or C3G. Meanwhile, the antioxidant properties of C3G were measured by determining the activities of superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT) and the malondialdehyde (MDA) levels. Flow cytometry was employed to determine HepG2 cells apoptosis, and HepG2 cells were stained with Hoechst 33342 to observe cell morphology. 2′,7′-dichlorofluorescin diacetate (DCFH-DA) was used to evaluate the production of intracellular reactive oxygen species (ROS). Finally, the expression of apoptosis-related protein was monitored through western blot analysis.
Results
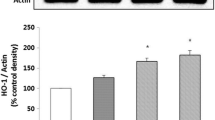
HepG2 cells induced with H2O2 presented a remarkable decrease in cell viability that was suppressed when HepG2 cells were interfered with C3G (2.5–10 μM). C3G interference memorably and dose-dependently inhibited H2O2-induced intracellular ROS and MDA overproduction, while C3G treatment markedly increased H2O2-induced the activities of intracellular SOD, GSH-Px and CAT. Eventually, the relative proteins expression levels of p53, cleaved caspase-9/3, cytochrome c, Fas-L, Fas, FADD and caspase-8 were substantially up-regulated in H2O2-triggered HepG2 cells, and Bax/Bcl-2 ratio and the relative protein expression levels of PARP were dramatically down-regulated. However, the expression levels of these relative proteins were reversed in C3G-interfered HepG2 cells.
Conclusions
C3G could protect HepG2 cells from oxidative damage, and the effects that were mediated by the mitochondrial apoptotic pathways and the external pathways.









Similar content being viewed by others
References
Acquaviva R, Russo A, Galvano F et al (2003) Cyanidin and cyanidin-3-O-β-D-glucoside as DNA cleavage protectors and antioxidants. Cell Biol Toxicol 19(4):243–252
Albano E (2008) Oxidative mechanisms in the pathogenesis of alcoholic liver disease. Mol Aspects Med 29(1–2):9–16. https://doi.org/10.1016/j.mam.2007.09.004
Arumugam T, Pillay Y, Ghazi T et al (2019) Fumonisin B1-induced oxidative stress triggers Nrf2-mediated antioxidant response in human hepatocellular carcinoma (HepG2) cells. Mycotoxin Res 35(1):99–109. https://doi.org/10.1007/s12550-018-0335-0
Carocho M, Ferreira ICFR (2013) A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem Toxicol 51:15–25. https://doi.org/10.1016/j.fct.2012.09.021
Chang CY, Shen CY, Kang CK et al (2014) Taurine protects HK-2 cells from oxidized LDL-induced cytotoxicity via the ROS-mediated mitochondrial and p53-related apoptotic pathways. Toxicol Appl Pharmacol 279(3):351–363. https://doi.org/10.1016/j.taap.2014.06.029
Chen WL, Qian Y, Meng WF et al (2009) A novel marine compound xyloketal B protects against oxidized LDL-induced cell injury in vitro. Biochem Pharmacol 78(8):941–950. https://doi.org/10.1016/j.bcp.2009.05.029
de Groot LES, Sabogal Pineros YS, Bal SM et al (2019) Do eosinophils contribute to oxidative stress in mild asthma? Clin Exp Allergy 49(6):929–931. https://doi.org/10.1111/cea.13389
Hao X, Kang Y, Li J et al (2016) Protective effects of hyperoside against H2O2-induced apoptosis in human umbilical vein endothelial cells. Mol Med Rep 14(1):399–405. https://doi.org/10.3892/mmr.2016.5235
Hong H, Liu GQ (2004) Protection against hydrogen peroxide-induced cytotoxicity in PC12 cells by scutellarin. Life Sci 74(24):2959–2973. https://doi.org/10.1016/j.lfs.2003.09.074
Hou X, Tong Q, Wang W et al (2015) Dihydromyricetin protects endothelial cells from hydrogen peroxide-induced oxidative stress damage by regulating mitochondrial pathways. Life Sci 130:38–46. https://doi.org/10.1016/j.lfs.2015.03.007
Huang W, Yan Z, Li D et al (2018) Antioxidant and anti-inflammatory effects of blueberry anthocyanins on high glucose-induced human retinal capillary endothelial cells. Oxid Med Cell Longev 2018:1862462. https://doi.org/10.1155/2018/1862462
Jia C, Xu L, Han T et al (2018) Generation of reactive oxygen species in heterogeneously sonoporated cells by microbubbles with single-pulse ultrasound. Ultrasound Med Biol 44(5):1074–1085. https://doi.org/10.1016/j.ultrasmedbio.2018.01.006
Klaunig JE, Wang Z, Pu X et al (2011) Oxidative stress and oxidative damage in chemical carcinogenesis. Toxicol Appl Pharmacol 254(2):86–99. https://doi.org/10.1016/j.taap.2009.11.028
Kwok HH, Ng WY, Yang MSM et al (2010) The ginsenoside protopanaxatriol protects endothelial cells from hydrogen peroxide-induced cell injury and cell death by modulating intracellular redox status. Free Radic Biol Med 48(3):437–445. https://doi.org/10.1016/j.freeradbiomed.2009.11.013
Lee JS, Kim YR, Song IG et al (2015) Cyanidin-3-glucoside isolated from mulberry fruit protects pancreatic β-cells against oxidative stress-induced apoptosis. Int J Mol Med 35(2):405–412. https://doi.org/10.3892/ijmm.2014.2013
Li L, Zhao J, Ge C et al (2018) Dehydroepiandrosterone rehabilitate BRL-3A cells oxidative stress damage induced by hydrogen peroxide. J Cell Physiol 233(8):6262–6272. https://doi.org/10.1002/jcp.26458
Liu T, Zhou Y, Wang J et al (2016) Coronary Microembolization induces cardiomyocyte apoptosis in swine by activating the LOX-1-dependent mitochondrial pathway and caspase-8-dependent pathway. J Cardiovasc Pharm 21(2):209–218. https://doi.org/10.1177/1074248415599265
Madungwe NB, Feng Y, Lie M et al (2018) Mitochondrial inner membrane protein (mitofilin) knockdown induces cell death by apoptosis via an AIF-PARP-dependent mechanism and cell cycle arrest. Am J Physiol Cell Physiol 315(1):C28–C43. https://doi.org/10.1152/ajpcell.00230.2017
Martinou JC, Youle RJ (2011) Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell 21(1):92–101. https://doi.org/10.1016/j.devcel.2011.06.017
Min J, Yu SW, Baek SH et al (2011) Neuroprotective effect of cyanidin-3-O-glucoside anthocyanin in mice with focal cerebral ischemia. Neurosci Lett 500(3):157–161. https://doi.org/10.1016/j.neulet.2011.05.048
Potukuchi A, Addepally U, Sindhu K et al (2018) Increased total DNA damage and oxidative stress in brain are associated with decreased longevity in high sucrose diet fed WNIN/Gr-Ob obese rats. Nutr Neurosci 21(9):648–656. https://doi.org/10.1080/1028415X.2017.1332509
Prasanna G, Jing P (2018) Cyanidin-3-O-glucoside functions like chemical chaperone and attenuates the glycation mediated amyloid formation in albumin. Arch Biochem Biophys 643:50–56. https://doi.org/10.1016/j.abb.2018.02.012
Qi G, Mi Y, Fan R et al (2017) Tea polyphenols ameliorate hydrogen peroxide-and constant darkness-triggered oxidative stress via modulating the Keap1/Nrf2 transcriptional signaling pathway in HepG2 cells and mice liver. RSC Adv 7(51):32198–32208
Sharma V, Anderson D, Dhawan A (2012) Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2). Apoptosis 17(8):852–870. https://doi.org/10.1007/S10495-012-0705-6
Shen H, Yuan Y, Ding F et al (2008) The protective effects of Achyranthes bidentata polypeptides against NMDA-induced cell apoptosis in cultured hippocampal neurons through differential modulation of NR2A- and NR2B-containing NMDA receptors. Brain Res Bull 77(5):274–281. https://doi.org/10.1016/j.brainresbull.2008.08.002
Song J, Zhao M, Liu X et al (2013) Protection of cyanidin-3-glucoside against oxidative stress induced by acrylamide in human MDA-MB-231 cells. Food Chem Toxicol 58:306–310. https://doi.org/10.1016/j.fct.2013.05.003
Subramaniam SR, Ellis EM (2011) Esculetin-induced protection of human hepatoma HepG2 cells against hydrogen peroxide is associated with the Nrf2-dependent induction of the NAD(P)H: Quinone oxidoreductase 1 gene. Toxicol Appl Pharmacol 250(2):130–136. https://doi.org/10.1016/j.taap.2010.09.025
Sun J, Wen Y, Zhou Y et al (2018) p53 attenuates acetaminophen-induced hepatotoxicity by regulating drug-metabolizing enzymes and transporter expression. Cell Death Dis 9(5):536. https://doi.org/10.1038/s41419-018-0507-z
Tan L, Yang H, Pang W et al (2014) Cyanidin-3-O-galactoside and blueberry extracts supplementation improves spatial memory and regulates hippocampal ERK expression in senescence-accelerated mice. Biomed Environ Sci 27(3):186–196. https://doi.org/10.3967/bes2014.007
Timon-Gomez A, Nyvltova E, Abriata LA et al (2018) Mitochondrial cytochrome c oxidase biogenesis: recent developments. Semin Cell Dev Biol 76:163–178. https://doi.org/10.1016/j.semcdb.2017.08.055
Tung CL, Ju DT, Velmurugan BK et al (2019) Carthamus tinctorius L. extract activates insulin-like growth factor-I receptor signaling to inhibit FAS-death receptor pathway and suppress lipopolysaccharides-induced H9c2 cardiomyoblast cell apoptosis. Environ Toxicol 34(12):1320–1328. https://doi.org/10.1002/tox.22833
Volpe CMO, Villar-Delfino PH, Dos Anjos PMF et al (2018) Cellular death, reactive oxygen species (ROS) and diabetic complications. Cell Death Dis 9(2):119. https://doi.org/10.1038/s41419-017-0135-z
Wang C, Youle RJ (2009) The role of mitochondria in apoptosis. Annu Rev Genet 43:95–118. https://doi.org/10.1146/annurev-genet-102108-134850
Wu D, Liang M, Dang H et al (2018a) Hydrogen protects against hyperoxia-induced apoptosis in type II alveolar epithelial cells via activation of PI3K/Akt/Foxo3a signaling pathway. Biochem Biophys Res Commun 495(2):1620–1627. https://doi.org/10.1016/j.bbrc.2017.11.193
Wu Z, Wang H, Fang S et al (2018b) Roles of endoplasmic reticulum stress and autophagy on H2O2-induced oxidative stress injury in HepG2 cells. Mol Med Rep 18(5):4163–4174. https://doi.org/10.3892/mmr.2018.9443
Xue H, Shen L, Wang X et al (2019) Isolation and purification of anthocyanin from blueberry using macroporous resin combined sephadex LH-20 techniques. Food Sci Technol Res 25(1):29–38
Xu Q, Ma P, Yu W et al (2010) Chitooligosaccharides protect human embryonic hepatocytes against oxidative stress induced by hydrogen peroxide. Mar Biotechnol 12(3):292–298. https://doi.org/10.1007/s10126-009-9222-1
Yang D, Zhu D, Zhu S et al (2018) 17β-Estradiol/extrogen receptor β alleviates apoptosis and enhances matrix biosynthesis of nucleus pulposus cells through regulating oxidative damage under a high glucose condition. Biomed Pharmacother 107:1004–1009. https://doi.org/10.1016/j.biopha.2018.08.084
Yuan L, Ren X, Wu Y et al (2013) Isoorientin protects BRL-3A rat liver cell against hydrogen peroxide-induced apoptosis by inhibiting mitochondrial dysfunction, inactivating MAPKs, activating Akt and scavenging ROS and NO. Biomed Aging Pathol 3(3):153–159. https://doi.org/10.1016/j.biomag.2013.06.004
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors of this paper declare no conflict of interest.
Ethical approval
This article does not contain any researches on human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tan, J., Li, P., Xue, H. et al. Cyanidin-3-glucoside prevents hydrogen peroxide (H2O2)-induced oxidative damage in HepG2 cells. Biotechnol Lett 42, 2453–2466 (2020). https://doi.org/10.1007/s10529-020-02982-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02982-2