Abstract
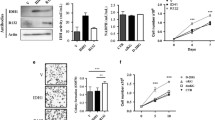
The gene encoding isocitrate dehydrogenase (IDH) is somatically mutated predominantly in secondary glioblastoma multiforme. Glioma-specific mutations in IDH1 always produced a single amino acid substitution at R132, but mutations in IDH2 were exclusively at R172 which was the analogous site to R132 in IDH1. Mutations of IDH1 and IDH2 led to simultaneous loss and gain of activities in the production of α-ketoglutarate and 2-hydroxyglutarate, respectively. Matrix metalloproteinases (MMPs) are zinc-dependent endoproteinases involved in the degradation of the extracellular matrix. The exact role of IDH2 mutant on MMPs activity and cell migration has not been fully studied. Here, we show that in response to IDH2 mutations, low levels of α-ketoglutarate increased the stabilization of HIF-1α which can contribute to tumor growth. Moreover, mutant IDH2-induced HIF-1α improved the secretion levels of pro-MMP-2 and pro-MMP-9 as well as the conversion from pro-MMP-2 to its active form, giving C6 glioma cells a higher migration potential. The HIF-1α pathway is probably a critical pathway for release of MMPs in the glioma cancer harboring IDH mutant.



Similar content being viewed by others
References
Badiga AV, Chetty C, Kesanakurti D, Are D, Gujrati M, Klopfenstein JD, Dinh DH, Rao JS (2011) MMP-2 siRNA inhibits radiation-enhanced invasiveness in glioma cells. PloS One 6:e20614
Berg G, Miksztowicz V, Schreier L (2011) Metalloproteinases in metabolic syndrome. Clin Chim Acta 412:1731–1739
Castro MM, Kandasamy AD, Youssef N, Schulz R (2011) Matrix metalloproteinase inhibitor properties of tetracyclines: therapeutic potential in cardiovascular diseases. Pharmacol Res 64(6):551–560
Chen RX, Cui JF, Xu CD, Xue TC, Guo K, Gao DM, Liu YK, Ye SL, Ren ZG (2011) The significance of MMP-9 over MMP-2 in HCC invasiveness and recurrence of hepatocellular carcinoma after curative resection. Ann Surg Oncol (Online)
Deshane J, Garner CC, Sontheimer H (2003) Chlorotoxin inhibits glioma cell invasion via matrix metalloproteinase-2. J Biol Chem 278:4135–4144
Fu YJ, Huang R, Du J, Yang RJ, An N, Liang AH (2010) Glioma-derived mutations in IDH: from mechanism to potential therapy. Biochem Biophys Res Commun 397:127–130
Fu YJ, An N, Chan KG, Wu YB, Zheng SH, Liang AH (2011a) A model of BmK CT in inhibiting glioma cell migration via matrix metalloproteinase-2 from experimental and molecular dynamics simulation study. Biotechnol Lett 33:1309–1317
Fu YJ, Huang R, Zheng YL, Zhang ZY, Liang AH (2011b) Glioma-derived mutations in isocitrate dehydrogenase 2 beneficial to traditional chemotherapy. Biochem Biophys Res Commun 410:218–223
Giese A, Westphal M (1996) Glioma invasion in the central nervous system. Neurosurgery 39:235–250
Gladson CL (1999) The extracelular matrix of gliomas: modulation of cell function. J Neuropathol Exp Neurol 58:1029–1040
Ji HT, Wang J, Fang B, Fang X, Lu Z (2011) α-Catenin inhibits glioma cell migration, invasion, and proliferation by suppression of β-catenin transactivation. J Neurooncol 103:445–451
Lee SY, Park SY, Kim SH, Choi EC (2011) Expression of matrix metalloproteiases and their inhibition in squamous cell carcinoma of the tonsil and their clinical significance. Clin Exp Otorhinolaryngol 4:88–94
Li LW, Chen P, Ling Y, Song XM, Lu ZJ, He QH, Li ZY, Lu N, Guo QL (2011) Inhibitory effects of GL-V9 on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-2/9. Eur J Pharm Sci 43:393–399
Ranogajec I, Razumovic JJ, Puzovic V, Gabrilovac J (2011) Prognostic value of matrix metalloproteinase-2 (MMP-2), matrix metalloproteinase-9 (MMP-9) and aminopeptidase N/CD13 in breast cancer patients. Med Oncol (Online)
Shao WL, Wang W, Xiong XG, Cao C, Yan TD, Chen GQ, Chen HZ, Yin WQ, Liu J, Gu YY, Mo MC, He JX (2011) Prognostic impact of MMP-2 and MMP-9 expression in pathologic stage IA non-small cell lung cancer. J Surg Oncol 104(7):841–846
Zhao SM, Lin Y, Xu W, Jiang WQ, Zha ZY, Wang P, Yu W, Li ZQ, Gong LL, Peng YJ, Ding JP, Lei QY, Guan KL, Xiong Y (2009) Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1α. Science 324:261–265
Acknowledgments
This project was supported by grants from the “National Natural Science Foundation of China” (No. 30700534, 31071924), the “Natural Science Foundation of Shanxi Province” (2008021039, 2010011040-1), and “Shanxi Scholarship Council of China”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, Y., Zheng, Y., Li, K. et al. Mutations in isocitrate dehydrogenase 2 accelerate glioma cell migration via matrix metalloproteinase-2 and 9. Biotechnol Lett 34, 441–446 (2012). https://doi.org/10.1007/s10529-011-0800-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-011-0800-8