Abstract
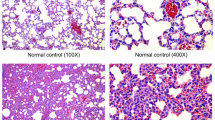
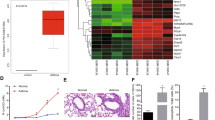
The aim of this study was to investigate the underlying mechanism of miR-9-5p in airway smooth muscle cells (ASMCs) of asthmatic mice. An asthmatic mouse model was established through the intraperitoneal injection of ovalbumin. Histopathological changes in lung tissues of asthmatic mice were observed using HE staining. ASMCs was identified using immunofluorescence staining and cell morphology. The mRNA expressions of miR-9-5p, KLF5, and IL-1β were measured using RT-qPCR. Additionally, CCK8 assay and flow cytometry were applied for ASMC proliferation and apoptosis, respectively. The protein levels of OPN, KLF5, and IL-1β were assessed using western blotting. The results showed that miR-9-5p was abnormally downregulated in lung tissues and ASMCs of asthmatic mice. Dual-Luciferase Reporter Assay and Chromatin immunoprecipitation confirmed that miR-9-5p targeted KLF5 that bounds to IL-1β promoter. Besides, miR-9-5p negatively regulated IL-1β mRNA and protein level via KLF5. Moreover, miR-9-5p was found to positively regulate ASMC apoptosis, negatively regulate ASMC proliferation and OPN protein expression, albeit with partial reversal by KLF5. Mechanistically, the regulation of ASMC proliferation and apoptosis by miR-9-5p is achieved by targeting KLF5/IL-1β axis.





Similar content being viewed by others
Data Availability
The data could be obtained upon request to the corresponding author.
References
Abe K, Sugiura H, Hashimoto Y, Ichikawa T, Koarai A, Yamada M, Numakura T, Onodera K, Tanaka R, Sato K, Yanagisawa S, Okazaki T, Tamada T, Kikuchi T, Ichinose M (2016) Possible role of Kruppel-like factor 5 in the remodeling of small airways and pulmonary vessels in chronic obstructive pulmonary disease. Respir Res 17:7. https://doi.org/10.1186/s12931-016-0322-y
Ali Syeda Z, Langden SSS, Munkhzul C, Lee M, Song SJ (2020) Regulatory mechanism of microRNA expression in cancer. Int J Mol Sci 21(5):1723. https://doi.org/10.3390/ijms21051723
Asher MI, Garcia-Marcos L, Pearce NE, Strachan DP (2020) Trends in worldwide asthma prevalence. Eur Respir J 56(6):2002094. https://doi.org/10.1183/13993003.02094-2020
Banno A, Reddy AT, Lakshmi SP, Reddy RC (2020) Bidirectional interaction of airway epithelial remodeling and inflammation in asthma. Clin Sci (lond) 134:1063–1079. https://doi.org/10.1042/CS20191309
Britt RD Jr, Ruwanpathirana A, Ford ML, Lewis BW (2023) Macrophages orchestrate airway inflammation, remodeling, and resolution in Asthma. Int J Mol Sci 24(13):10451. https://doi.org/10.3390/ijms241310451
Dang X, Yang L, Guo J, Hu H, Li F, Liu Y, Pang Y (2019) miR-145-5p is associated with smoke-related chronic obstructive pulmonary disease via targeting KLF5. Chem Biol Interact 300:82–90. https://doi.org/10.1016/j.cbi.2019.01.011
Fehrenbach H, Wagner C, Wegmann M (2017) Airway remodeling in asthma: what really matters. Cell Tissue Res 367:551–569. https://doi.org/10.1007/s00441-016-2566-8
Francisco-Garcia AS, Garrido-Martin EM, Rupani H, Lau LCK, Martinez-Nunez RT, Howarth PH, Sanchez-Elsner T (2019) Small RNA species and microRNA profiles are altered in severe asthma nanovesicles from broncho alveolar lavage and associate with impaired lung function and inflammation. Noncoding RNA 5(4):51. https://doi.org/10.3390/ncrna5040051
Gao Y, Liu Y, Zheng D, Ho C, Wen D, Sun J, Huang L, Liu Y, Li Q, Zhang Y (2022) HDAC5-mediated Smad7 silencing through MEF2A is critical for fibroblast activation and hypertrophic scar formation. Int J Biol Sci 18(15):5724–5739. https://doi.org/10.7150/ijbs.76140
García-Marcos L, Chiang CY, Asher MI, Marks GB, El Sony A, Masekela R, Bissell K, Ellwood E, Ellwood P, Pearce N, Strachan DP, Mortimer K, Morales E, Global Asthma Network Phase I Study Group (2023) Asthma management and control in children, adolescents, and adults in 25 countries: a Global Asthma Network Phase I cross-sectional study. Lancet Glob Health 11:e218–e228. https://doi.org/10.1016/S2214-109X(22)00506-X
Hirota N, Martin JG (2013) Mechanisms of airway remodeling. Chest 144:1026–1032. https://doi.org/10.1378/chest.12-3073
Khalfaoui L, Pabelick CM (2023) Airway smooth muscle in contractility and remodeling of asthma: potential drug target mechanisms. Expert Opin Ther Targets 27:19–29. https://doi.org/10.1080/14728222.2023.2177533
Kim RY, Pinkerton JW, Essilfie AT, Robertson AAB, Baines KJ, Brown AC, Mayall JR, Ali MK, Starkey MR, Hansbro NG, Hirota JA, Wood LG, Simpson JL, Knight DA, Wark PA, Gibson PG, O’Neill LAJ, Cooper MA, Horvat JC, Hansbro PM (2017) Role for NLRP3 inflammasome-mediated, IL-1β-dependent responses in severe, steroid-resistant asthma. Am J Respir Crit Care Med 196:283–297. https://doi.org/10.1164/rccm.201609-1830OC
Konno S, Kurokawa M, Uede T, Nishimura M, Huang SK (2011) Role of osteopontin, a multifunctional protein, in allergy and asthma. Clin Exp Allergy 41:1360–1366. https://doi.org/10.1111/j.1365-2222.2011.03775.x
Kumar MS, Lu J, Mercer KL, Golub TR, Jacks T (2007) Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nat Genet 39:673–677. https://doi.org/10.1038/ng2003
Kuo PL, Hsu YL, Huang MS, Chiang SL, Ko YC (2011) Bronchial epithelium-derived IL-8 and RANTES increased bronchial smooth muscle cell migration and proliferation by Kruppel-like factor 5 in areca nut-mediated airway remodeling. Toxicol Sci 121:177–190. https://doi.org/10.1093/toxsci/kfr030
Li B, Ding T, Chen H, Li C, Chen B, Xu X, Huang P, Hu F, Guo L (2023) CircStrn3 targeting microRNA-9-5p is involved in the regulation of cartilage degeneration and subchondral bone remodelling in osteoarthritis. Bone Joint Res 12:33–45. https://doi.org/10.1302/2046-3758.121.BJR-2022-0231.R1
Liu Y, Gao X, Miao Y, Wang Y, Wang H, Cheng Z, Wang X, Jing X, Jia L, Dai L, Liu M, An L (2018) NLRP3 regulates macrophage M2 polarization through up-regulation of IL-4 in asthma. Biochem J 475:1995–2008. https://doi.org/10.1042/BCJ20180086
Liu M, Liu S, Li F, Li C, Chen S, Gao X, Wang X (2023) The miR-124-3p regulates the allergic airway inflammation and remodeling in an ovalbumin-asthmatic mouse model by inhibiting S100A4. Immun Inflamm Dis 11:e730. https://doi.org/10.1002/iid3.730
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Method Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Luo Y, Chen C (2021) The roles and regulation of the KLF5 transcription factor in cancers. Cancer Sci 112:2097–2117. https://doi.org/10.1111/cas.14910
Mahmutovic Persson I, Menzel M, Ramu S, Cerps S, Akbarshahi H, Uller L (2018) IL-1beta mediates lung neutrophilia and IL-33 expression in a mouse model of viral-induced asthma exacerbation. Respir Res 19:16. https://doi.org/10.1186/s12931-018-0725-z
Peebles RS Jr (2017) Is IL-1beta inhibition the next therapeutic target in asthma? J Allergy Clin Immunol 139:1788–1789. https://doi.org/10.1016/j.jaci.2017.03.018
Rao SS, Mu Q, Zeng Y, Cai PC, Liu F, Yang J, Xia Y, Zhang Q, Song LJ, Zhou LL, Li FZ, Lin YX, Fang J, Greer PA, Shi HZ, Ma WL, Su Y, Ye H (2017) Calpain-activated mTORC2/Akt pathway mediates airway smooth muscle rem odelling in asthma. Clin Exp Allergy 47:176–189. https://doi.org/10.1111/cea.12805
Rodrigues M, de Castro MF, Paciência I, Barros R, Padrão P, Cavaleiro Rufo J, Silva D, Delgado L, Moreira A, Moreira P (2023) Diet quality, asthma and airway inflammation in school-aged children. Eur Ann Allergy Clin Immunol. https://doi.org/10.23822/EurAnnACI.1764-1489.301
Santarlasci V, Cosmi L, Maggi L, Liotta F, Annunziato F (2013) IL-1 and T helper immune responses. Front Immunol 4:182. https://doi.org/10.3389/fimmu.2013.00182
Simoes DC, Xanthou G, Petrochilou K, Panoutsakopoulou V, Roussos C, Gratziou C (2009) Osteopontin deficiency protects against airway remodeling and hyperresponsiveness in chronic asthma. Am J Respir Crit Care Med 179:894–902. https://doi.org/10.1164/rccm.200807-1081OC
Stern J, Pier J, Litonjua AA (2020) Asthma epidemiology and risk factors. Semin Immunopathol 42:5–15. https://doi.org/10.1007/s00281-020-00785-1
Trinh HKT, Nguyen TVT, Kim SH, Cao TBT, Luu QQ, Kim SH, Park HS (2020) Osteopontin contributes to late-onset asthma phenotypes in adult asthma patients. Exp Mol Med 52:253–265. https://doi.org/10.1038/s12276-020-0376-2
Wang X, Xu R, Chi D, Dai C, Sheng M (2021) Role of NEAT1/miR-9-5p/SLC26A2 pathway on human airway smooth muscle cell. Yonsei Med J 62:858–867. https://doi.org/10.3349/ymj.2021.62.9.858
Wang Z, Song Y, Jiang J, Piao Y, Li L, Bai Q, Xu C, Liu H, Li L, Piao H, Yan G (2022) MicroRNA-182-5p attenuates asthmatic airway inflammation by targeting NOX4. Front Immunol 13:853848. https://doi.org/10.3389/fimmu.2022.853848
Wei W, Chen W, He N (2021) HDAC4 induces the development of asthma by increasing Slug-upregulated CXCL12 expression through KLF5 deacetylation. J Transl Med 19:258. https://doi.org/10.1186/s12967-021-02812-7
Xiong Y, Luo Y, Yuwen T, Li J, Chen R, Shi F (2023) The regulatory role of miR-107-Cdk6-Rb pathway in airway smooth muscle cells in asthma. J Asthma Allergy 16:433–445. https://doi.org/10.2147/JAA.S405457
Yang Q, Miao Q, Chen H, Li D, Luo Y, Chiu J, Wang HJ, Chuvanjyan M, Parmacek MS, Shi W (2023) Myocd regulates airway smooth muscle cell remodeling in response to chronic asthmatic injury. J Pathol 259:331–341. https://doi.org/10.1002/path.6044
Zhai W, Eynott PR, Oltmanns U, Leung SY, Chung KF (2004) Mitogen-activated protein kinase signalling pathways in IL-1 beta-dependent rat airway smooth muscle proliferation. Br J Pharmacol 143:1042–1049. https://doi.org/10.1038/sj.bjp.0705971
Zhao J, Pu J, Hao B, Huang L, Chen J, Hong W, Zhou Y, Li B, Ran P (2020) LncRNA RP11–86H7.1 promotes airway inflammation induced by TRAPM2.5 by acting as a ceRNA of miRNA-9-5p to regulate NFKB1 in HBECS. Sci Rep 10:11587. https://doi.org/10.1038/s41598-020-68327-1
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
CX and HH designed the study, JZ wrote the paper, and all authors participated in the experiments, data analysis, and approved the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Ethical Approval
All the animal experiments were approved by the Institutional Animal Care and Use Committee of Jinzhou Medical University.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10528_2023_10640_MOESM1_ESM.tif
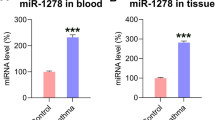
Supplementary file1 (TIF 121 kb) Supplementary figure 1. MiR-9-5p expression in peripheral blood from asthmatic patients and healthy people.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, C., Huang, H., Zou, H. et al. The miR-9-5p/KLF5/IL-1β Axis Regulates Airway Smooth Muscle Cell Proliferation and Apoptosis to Aggravate Airway Remodeling and Inflammation in Asthma. Biochem Genet (2024). https://doi.org/10.1007/s10528-023-10640-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-023-10640-1