Abstract
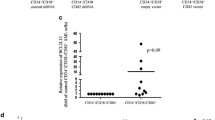
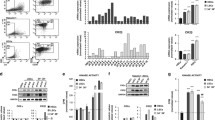
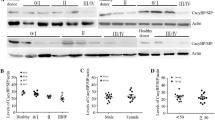
Chronic lymphocytic leukemia (CLL) is a clonal proliferative disease of mature B lymphocytes. To further improve the prognosis of patients, it is necessary to further elucidate the pathogenesis of CLL and find more effective therapeutic targets. Nuclear Factor of Activated T cells 5 (NFAT5) is the major activated transcription factor (TF) upon osmotic pressure increase in mammalian cells, and it also regulates many target genes to affect various cellular functions. The effects of NFAT5 on tumor growth and metastasis have also been widely revealed. However, the effects of NFAT5 on the progression of CLL are still unclear. In this study, we found abnormally high expression of NFAT5 in human CLL patients. Additionally, NFAT5 depletion suppressed proliferation and stimulated apoptosis of CLL cells. Our data further confirmed NFAT5 regulated AQP5 expression and the phosphorylation of p38 MAPK. We also found that AQP5 overexpression reversed the inhibitory effect of NFAT5 depletion on cell proliferation in CLL cells. Furthermore, we revealed STUB1 directly bound to NFAT5 and promoted its degradation. Taken together, our results indicate the involvement of NFAT5 in CLL progression and suggest that NFAT5 could serve as a promising therapeutic target for CLL treatment.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Amara S, Alotaibi D, Tiriveedhi V (2016) NFAT5/STAT3 interaction mediates synergism of high salt with IL-17 towards induction of VEGF-A expression in breast cancer cells. Oncol Lett 12:933–943. https://doi.org/10.3892/ol.2016.4713
Barthel R et al (2020) CD74 is dispensable for development of chronic lymphocytic leukemia in Emicro-TCL1 transgenic mice. Leuk Lymphoma. https://doi.org/10.1080/10428194.2020.1791851
Chae YK et al (2008) Human AQP5 plays a role in the progression of chronic myelogenous leukemia (CML). PLoS ONE 3:e2594. https://doi.org/10.1371/journal.pone.0002594
Fernandez-Castillo N et al (2015) Transcriptomic and genetic studies identify NFAT5 as a candidate gene for cocaine dependence. Transl Psychiatry 5:e667. https://doi.org/10.1038/tp.2015.158
Germann S et al (2012) Dual role of the ddx5/ddx17 RNA helicases in the control of the pro-migratory NFAT5 transcription factor. Oncogene 31:4536–4549. https://doi.org/10.1038/onc.2011.618
Gopalakrishnan S et al (2020) Integrated mechanistic model of minimal residual disease kinetics with venetoclax therapy in chronic lymphocytic leukemia. Clin Pharmacol Ther. https://doi.org/10.1002/cpt.2005
Guo K, Jin F (2015) NFAT5 promotes proliferation and migration of lung adenocarcinoma cells in part through regulating AQP5 expression. Biochem Biophys Res Commun 465:644–649. https://doi.org/10.1016/j.bbrc.2015.08.078
Halterman JA, Kwon HM, Leitinger N, Wamhoff BR (2012) NFAT5 expression in bone marrow-derived cells enhances atherosclerosis and drives macrophage migration. Front Physiol 3:313. https://doi.org/10.3389/fphys.2012.00313
Hashemi M, Mohammadipour M, Rostami S, Soltanpour MS (2020) Promoter DNA methylation frequency and clinicopathological role of miR-129–2 gene in patients with chronic lymphocytic leukemia. Oman Med J 35:e151. https://doi.org/10.5001/omj.2020.71
Hosnijeh FS et al (2020) Proteomic markers with prognostic impact on outcome of chronic lymphocytic leukemia patients under chemo-immunotherapy; results from the HOVON 109 study. Exp Hematol. https://doi.org/10.1016/j.exphem.2020.08.002
Ito T, Fujio Y, Takahashi K, Azuma J (2007) Degradation of NFAT5, a transcriptional regulator of osmotic stress-related genes, is a critical event for doxorubicin-induced cytotoxicity in cardiac myocytes. J Biol Chem 282:1152–1160. https://doi.org/10.1074/jbc.M609547200
Jiang Y et al (2019) Transcription factor NFAT5 contributes to the glycolytic phenotype rewiring and pancreatic cancer progression via transcription of PGK1. Cell Death Dis 10:948. https://doi.org/10.1038/s41419-019-2072-5
Karabon L et al (2020) Abnormal expression of BTLA and CTLA-4 immune checkpoint molecules in chronic lymphocytic leukemia patients. J Immunol Res 2020:6545921. https://doi.org/10.1155/2020/6545921
Kim DH, Kim KS, Ramakrishna S (2018) NFAT5 promotes in vivo development of murine melanoma metastasis. Biochem Biophys Res Commun 505:748–754. https://doi.org/10.1016/j.bbrc.2018.09.171
Koch M et al (2020) Meta-analysis reveals significant sex differences in chronic lymphocytic leukemia progression in the emicro-TCL1 transgenic mouse model. Cancers. https://doi.org/10.3390/cancers12071980
Kumar R, DuMond JF, Khan SH, Thompson EB, He Y, Burg MB, Ferraris JD (2020) NFAT5, which protects against hypertonicity, is activated by that stress via structuring of its intrinsically disordered domain. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1911680117
Kuper C, Beck FX, Neuhofer W (2014) NFAT5-mediated expression of S100A4 contributes to proliferation and migration of renal carcinoma cells. Front Physiol 5:293. https://doi.org/10.3389/fphys.2014.00293
Lee JH, Kim JW, Im YS, Seong GJ, Lee HK (2011) Cyclosporine a induces nerve growth factor expression via activation of MAPK p38 and NFAT5. Cornea 30(Suppl 1):S19–S24. https://doi.org/10.1097/ICO.0b013e3182281028
Meng X, Li Z, Zhou S, Xiao S, Yu P (2019) miR-194 suppresses high glucose-induced non-small cell lung cancer cell progression by targeting NFAT5. Thorac Cancer 10:1051–1059. https://doi.org/10.1111/1759-7714.13038
Park J et al (2014) Increased NFAT5 expression stimulates transcription of Hsp70 in preeclamptic placentas. Placenta 35:109–116. https://doi.org/10.1016/j.placenta.2013.12.005
Qin X, Wang Y, Li J, Xiao Y, Liu Z (2017) NFAT5 inhibits invasion and promotes apoptosis in hepatocellular carcinoma associated with osmolality. Neoplasma 64:502–510. https://doi.org/10.4149/neo_2017_403
Remo A et al (2015) Systems biology analysis reveals NFAT5 as a novel biomarker and master regulator of inflammatory breast cancer. J Transl Med 13:138. https://doi.org/10.1186/s12967-015-0492-2
Sedlarikova L, Petrackova A, Papajik T, Turcsanyi P, Kriegova E (2020) Resistance-associated mutations in chronic lymphocytic leukemia patients treated with novel agents. Front Oncol 10:894. https://doi.org/10.3389/fonc.2020.00894
Trama J, Lu Q, Hawley RG, Ho SN (2000) The NFAT-related protein NFATL1 (TonEBP/NFAT5) is induced upon T cell activation in a calcineurin-dependent manner. J Immunol 165:4884–4894. https://doi.org/10.4049/jimmunol.165.9.4884
Villanueva S et al (2012) NFAT5 is activated by hypoxia: role in ischemia and reperfusion in the rat kidney. PLoS ONE 7:e39665. https://doi.org/10.1371/journal.pone.0039665
Wang Q, Zhou Y, Rychahou P, Liu C, Weiss HL, Evers BM (2013) NFAT5 represses canonical Wnt signaling via inhibition of beta-catenin acetylation and participates in regulating intestinal cell differentiation. Cell Death Dis 4:e671. https://doi.org/10.1038/cddis.2013.202
Xu S, Sui J, Fu Y, Wu W, Liu T, Yang S, Liang G (2020a) Titanium dioxide nanoparticles induced the apoptosis of RAW264.7 macrophages through miR-29b-3p/NFAT5 pathway. Environ Sci Pollut Res Int 27:26153–26162. https://doi.org/10.1007/s11356-020-08952-5
Xu Z, Sun Y, Wei Z, Jiang J, Xu J, Liu P (2020b) Suppression of CXCL-1 could restore necroptotic pathway in chronic lymphocytic leukemia. OncoTargets Ther 13:6917–6925. https://doi.org/10.2147/OTT.S256993
Yoshimoto S, Morita H, Matsuda M, Katakura Y, Hirata M, Hashimoto S (2020) NFAT5 promotes oral squamous cell carcinoma progression in a hyperosmotic environment. Lab Investig. https://doi.org/10.1038/s41374-020-00486-1
Zhou X, Izumi Y, Burg MB, Ferraris JD (2011) Rac1/osmosensing scaffold for MEKK3 contributes via phospholipase C-gamma1 to activation of the osmoprotective transcription factor NFAT5. Proc Natl Acad Sci USA 108:12155–12160. https://doi.org/10.1073/pnas.1108107108
Zhou X et al (2020) Anti-breast cancer effect of 2-dodecyl-6-Methoxycyclohexa-2,5-diene-1,4-dione in vivo and in vitro through MAPK signaling pathway. Drug Design Devel Ther 14:2667–2684. https://doi.org/10.2147/DDDT.S237699
Author information
Authors and Affiliations
Contributions
BLC and YL designed the study, supervised data collection, SX and YN analyzed the data, interpreted the data, JZ prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors state that there are no conflicts of interest to disclose.
Ethical Approval
Ethical approval was obtained from the Ethics Committee of Affiliated Hospital of Guilin Medical University.
Informed Consent
Written informed consent was obtained from legally authorized representative(s) for anonymized patient information to be published in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, B.L., Li, Y., Xu, S. et al. NFAT5 Regulated by STUB1, Facilitates Malignant Cell Survival and p38 MAPK Activation by Upregulating AQP5 in Chronic Lymphocytic Leukemia. Biochem Genet 59, 870–883 (2021). https://doi.org/10.1007/s10528-021-10040-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-021-10040-3