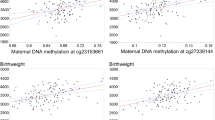
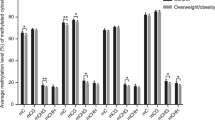
Abstract
DNA methylation status of RXRα gene promoter has been correlated with maternal diet during early pregnancy, and associated with offspring’s adiposity and bone mineral content. In adult life, increased methylation of RXRα promoter region is associated with myocardium pathologies. Early growth response proteins (EGR) are zinc finger transcription factors associated with several cellular pathways such as inflammation, apoptosis, and cardiopathies. DNA-binding sequences of EGR proteins have been reported in the RXRα gene promoter using chromatin immunoprecipitation methods. Here, we used correlations between the maternal pre-pregnancy body mass index (p-BMI), gestational weight gain (GWG), and birth weight (BW) as indirect indicators of the maternal nutritional status as modifier of DNA methylation in the offspring. DNA methylation status from newborns’ umbilical vein blood in full-term pregnancy was evaluated in a short sequence (116 pb) of the RXRα gene promoter that contains the elements of response sequence for EGR proteins. Fifty-three bisulfite-modified DNA samples were assessed through methyl-sensitive high-resolution melting (MS-HRM) analysis. To validate the results, we directly sequenced MS-HRM samples to confirm the presence of CpG-methylated positions. In addition, the RXRα protein levels in extracts of umbilical vein blood were evaluated by western blot. We found differential methylation in a specific locus of the RXRα promoter surrounding the EGR-binding sequence; however, no correlation was found with the level of RXRα protein expression. Variability in the methylation status of the RXRα promoter near the EGR transcription factor binding site in newborn cord blood provides controversial epigenetic insights into RXRα regulation via EGR proteins.




Similar content being viewed by others
References
Baron VT, Pio R, Jia Z, Mercola D (2015) Early growth response 3 regulates genes of inflammation and directly activates IL6 and IL8 expression in prostate cancer. Br J Cancer. https://doi.org/10.1038/bjc.2014.622
Bodega B, Orlando V (2014) Repetitive elements dynamics in cell identity programming, maintenance and disease. Curr Opin Cell Biol 31:67–73. https://doi.org/10.1016/j.ceb.2014.09.002
Chen M, Xiong F, Zhang L (2013) Promoter methylation of Egr-1 site contributes to fetal hypoxia-mediated PKCepsilon gene repression in the developing heart. Am J Physiol Regul Integr Comp Physiol 304:R683–R689. https://doi.org/10.1152/ajpregu.00461.2012
Chorley BN et al (2012) Identification of novel NRF2-regulated genes by ChIP-Seq: influence on retinoid X receptor alpha. Nucleic Acids Res 40:7416–7429. https://doi.org/10.1093/nar/gks409
Chow EK et al (2006) A role for IRF3-dependent RXRalpha repression in hepatotoxicity associated with viral infections. J Exp Med 203:2589–2602. https://doi.org/10.1084/jem.20060929
Corte Z, Venta R (2010) Biological variation of free plasma amino acids in healthy individuals. Clin Chem Lab Med 48:99–104. https://doi.org/10.1515/CCLM.2010.008
Cunningham FG (2010) Laboratory values in normal pregnancy. In: Queenan JT, Hobbins JC, Spong CY (eds) Protocols for high-risk pregnancies: an evidence-based approach, 5th edn. Blackwell Science Ltd, Hoboken
de Koning AP, Gu W, Castoe TA, Batzer MA, Pollock DD (2011) Repetitive elements may comprise over two-thirds of the human genome. PLoS Genet 7:e1002384. https://doi.org/10.1371/journal.pgen.1002384
Duester G (1998) Alcohol dehydrogenase as a critical mediator of retinoic acid synthesis from vitamin A in the mouse embryo. J Nutr 128:459S–462S
Engel SM et al (2014) Neonatal genome-wide methylation patterns in relation to birth weight in the Norwegian mother and child cohort. Am J Epidemiol 179:834–842. https://doi.org/10.1093/aje/kwt433
FAO (1996) The Sixth World food survey. Food and Agriculture Organization of the United Nations, Rome
Fraser CG, Harris EK (1989) Generation and application of data on biological variation in clinical chemistry. Crit Rev Clin Lab Sci 27:409–437. https://doi.org/10.3109/10408368909106595
Gabbe SG, Niebyl JR, Simpson JL, Landon MB, Galan HL, Jauniaux ERM, Driscoll DA (2007) Obstetrics: normal and problem pregnancies, 6th edn. Elsevier Saunders, Philadelphia
Gemma C et al (2009) Maternal pregestational BMI is associated with methylation of the PPARGC1A promoter in newborns. Obesity 17:1032–1039. https://doi.org/10.1038/oby.2008.605
Gluckman PD, Hanson MA, Cooper C, Thornburg KL (2008) Effect of in utero and early-life conditions on adult health and disease. N Engl J Med 359:61–73. https://doi.org/10.1056/NEJMra0708473
Godfrey KM et al (2011) Epigenetic gene promoter methylation at birth is associated with child’s later adiposity. Diabetes 60:1528–1534. https://doi.org/10.2337/db10-0979
Gueri M, Jutsum P, Sorhaindo B (1982) Anthropometric assessment of nutritional status in pregnant women: a reference table of weight-for-height by week of pregnancy. Am J Clin Nutr 35:609–616
Hamza MS et al (2009) De-novo identification of PPARgamma/RXR binding sites and direct targets during adipogenesis. PloS ONE 4:e4907. https://doi.org/10.1371/journal.pone.0004907
Hanson MA, Godfrey KM (2008) Commentary: maternal constraint is a pre-eminent regulator of fetal growth. Int J Epidemiol 37:252–254. https://doi.org/10.1093/ije/dyn015
Harvey NC et al (2014) Childhood bone mineral content is associated with methylation status of the RXRA promoter at birth. J Bone Miner Res 29:600–607. https://doi.org/10.1002/jbmr.2056
Hernandez HG, Tse MY, Pang SC, Arboleda H, Forero DA (2013) Optimizing methodologies for PCR-based DNA methylation analysis. Biotechniques 55:181–197. https://doi.org/10.2144/000114087
Kaar JL, Crume T, Brinton JT, Bischoff KJ, McDuffie R, Dabelea D (2014) Maternal obesity, gestational weight gain, and offspring adiposity: the exploring perinatal outcomes among children study. J Pediatr 165:509–515. https://doi.org/10.1016/j.jpeds.2014.05.050
Kuhn RM et al (2009) The UCSC genome browser database: update. Nucleic Acids Res 37:D755–D761. https://doi.org/10.1093/nar/gkn875
Langley-Evans SC (2006) Developmental programming of health and disease. Proc Nutr Soc 65:97–105
Lee JM et al (2014) Gestational weight gain is an important risk factor for excessive fetal growth. Obstet Gynecol Sci 57:442–447. https://doi.org/10.5468/ogs.2014.57.6.442
Li G et al (2006) Identification and characterization of the human retinoid X receptor alpha gene promoter. Gene 372:118–127. https://doi.org/10.1016/j.gene.2005.12.027
Michels KB, Harris HR, Barault L (2011) Birthweight, maternal weight trajectories and global DNA methylation of LINE-1 repetitive elements. PLoS ONE 6:e25254. https://doi.org/10.1371/journal.pone.0025254
Morales E, Groom A, Lawlor DA, Relton CL (2014) DNA methylation signatures in cord blood associated with maternal gestational weight gain: results from the ALSPAC cohort. BMC Res Notes 7:278. https://doi.org/10.1186/1756-0500-7-278
Nielsen JH et al (2014) Impact of fetal and neonatal environment on beta cell function and development of diabetes. Acta Obstet Gynecol Scand 93:1109–1122. https://doi.org/10.1111/aogs.12504
Pagel JI, Deindl E (2011) Early growth response 1–a transcription factor in the crossfire of signal transduction cascades. Indian J Biochem Biophys 48:226–235
Perera F, Herbstman J (2011) Prenatal environmental exposures, epigenetics, and disease. Reprod Toxicol 31:363–373. https://doi.org/10.1016/j.reprotox.2010.12.055
Phaloprakarn C, Manusirivithaya S, Boonyarittipong P (2014) Risk score comprising maternal and obstetric factors to identify late preterm infants at risk for neonatal intensive care unit admission. J Obstet Gynaecol Res. https://doi.org/10.1111/jog.12610
Pilsner JR et al (2009) Influence of prenatal lead exposure on genomic methylation of cord blood DNA. Environ Health Perspect 117:1466–1471. https://doi.org/10.1289/ehp.0800497
Rakyan VK, Down TA, Balding DJ, Beck S (2011) Epigenome-wide association studies for common human diseases. Nat Rev Genet 12:529–541. https://doi.org/10.1038/nrg3000
Ramadas N, Rajaraman B, Kuppuswamy AA, Vedantham S (2014) Early growth response-1 (EGR-1): a key player in myocardial cell injury. Cardiovasc Hematol Agents Med Chem 12:66–71
Rasmussen KM, Yaktine AL (2009) Weight gain during pregnancy: reexamining the guidelines. The National Academies Press, Washington, D.C.
Reik W, Dean W, Walter J (2001) Epigenetic reprogramming in mammalian development. Science 293:1089–1093. https://doi.org/10.1126/science.1063443
Saben J, Zhong Y, Gomez-Acevedo H, Thakali KM, Borengasser SJ, Andres A, Shankar K (2013) Early growth response protein-1 mediates lipotoxicity-associated placental inflammation: role in maternal obesity. Am J Physiol Endocrinol Metab 305:E1–14. https://doi.org/10.1152/ajpendo.00076.2013
Schaiff WT, Bildirici I, Cheong M, Chern PL, Nelson DM, Sadovsky Y (2005) Peroxisome proliferator-activated receptor-gamma and retinoid X receptor signaling regulate fatty acid uptake by primary human placental trophoblasts. J Clin Endocrinol Metabol 90:4267–4275. https://doi.org/10.1210/jc.2004-2265
Schroeder JW et al (2011) Neonatal DNA methylation patterns associate with gestational age. Epigenetics 6:1498–1504. https://doi.org/10.4161/epi.6.12.18296
Shin SY, Kim JH, Baker A, Lim Y, Lee YH (2010) Transcription factor Egr-1 is essential for maximal matrix metalloproteinase-9 transcription by tumor necrosis factor alpha. Mol Cancer Res MCR 8:507–519. https://doi.org/10.1158/1541-7786.MCR-09-0454
Shukla S et al (2011) CTCF-promoted RNA polymerase II pausing links DNA methylation to splicing. Nature 479:74–79. https://doi.org/10.1038/nature10442
Siegfried Z, Eden S, Mendelsohn M, Feng X, Tsuberi BZ, Cedar H (1999) DNA methylation represses transcription in vivo. Nat Genet 22:203–206. https://doi.org/10.1038/9727
Sonagra AD, Biradar SM, Murthy KD (2014) Normal pregnancy-a state of insulin resistance. JCDR 8:CC01–CC03. https://doi.org/10.7860/jcdr/2014/10068.5081
Soubry A et al (2013) Paternal obesity is associated with IGF2 hypomethylation in newborns: results from a Newborn Epigenetics Study (NEST) cohort. BMC Med 11:29. https://doi.org/10.1186/1741-7015-11-29
Taschner S, Koesters C, Platzer B, Jorgl A, Ellmeier W, Benesch T, Strobl H (2007) Down-regulation of RXRalpha expression is essential for neutrophil development from granulocyte/monocyte progenitors. Blood 109:971–979. https://doi.org/10.1182/blood-2006-04-020552
Teh AL et al (2014) The effect of genotype and in utero environment on interindividual variation in neonate DNA methylomes. Genome Res 24:1064–1074. https://doi.org/10.1101/gr.171439.113
Tobi EW et al (2009) DNA methylation differences after exposure to prenatal famine are common and timing- and sex-specific. Hum Mol Genet 18:4046–4053. https://doi.org/10.1093/hmg/ddp353
Uhlen M et al (2005) A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol Cell Proteom MCP 4:1920–1932. https://doi.org/10.1074/mcp.M500279-MCP200
Vera E, Canela A, Fraga MF, Esteller M, Blasco MA (2008) Epigenetic regulation of telomeres in human cancer. Oncogene 27:6817–6833. https://doi.org/10.1038/onc.2008.289
Wendling O, Chambon P, Mark M (1999) Retinoid X receptors are essential for early mouse development and placentogenesis. Proc Natl Acad Sci USA 96:547–551
Whitaker RC (2004) Predicting preschooler obesity at birth: the role of maternal obesity in early pregnancy. Pediatrics 114:e29–e36
Wojdacz TK, Dobrovic A (2007) Methylation-sensitive high resolution melting (MS-HRM): a new approach for sensitive and high-throughput assessment of methylation. Nucleic Acids Res 35:e41. https://doi.org/10.1093/nar/gkm013
Wojdacz TK, Moller TH, Thestrup BB, Kristensen LS, Hansen LL (2010) Limitations and advantages of MS-HRM and bisulfite sequencing for single locus methylation studies. Expert Rev Mol Diagn 10:575–580. https://doi.org/10.1586/erm.10.46
Zandarashvili L, White MA, Esadze A, Iwahara J (2015) Structural impact of complete CpG methylation within target DNA on specific complex formation of the inducible transcription factor Egr-1. FEBS Lett 589:1748–1753. https://doi.org/10.1016/j.febslet.2015.05.022
Zhang D et al (2010) Genetic control of individual differences in gene-specific methylation in human brain. Am J Hum Genet 86:411–419. https://doi.org/10.1016/j.ajhg.2010.02.005
Zhang J, Ma X, Wang H, Ma D, Huang G (2014) Elevated methylation of the RXRA promoter region may be responsible for its downregulated expression in the myocardium of patients with TOF. Pediatr Res 75:588–594. https://doi.org/10.1038/pr.2014.17
Acknowledgements
This project was supported by the Dirección General de Asuntos del Personal Académico (DGAPA), UNAM postdoctoral fellowship for ECS, and funds from Fundación Miguel Alemán, A.C. We would like to thank Soledad López-Alvarez and Beatriz Sánchez-Hernández (CINVESTAV-IPN and INCMNSZ) for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no known conflicts of interest associated with this publication. The present project was conducted with full compliance of research ethics norms, and more specifically the rules, specifications, and good practices established in the Official Mexican Standards for health and by the Mexico City Ministry of Health.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chávez-Lizárraga, D., Zárate-Segura, P., Beltrán-Montoya, J. et al. DNA Methylation Variability in a Single Locus of the RXRα Promoter from Umbilical Vein Blood at Term Pregnancy. Biochem Genet 56, 210–224 (2018). https://doi.org/10.1007/s10528-017-9838-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-017-9838-1