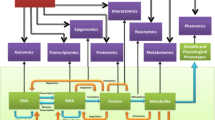
Abstract
Ageing is a biological process guided by genetic and environmental factors that ultimately lead to adverse outcomes for organismal lifespan and healthspan. Determination of molecular pathways that are affected with age and increase disease susceptibility is crucial. The gene expression profile of the ideal ageing model, namely the nematode Caenorhabditis elegans mapped with the microarray technology initially led to the identification of age-dependent gene expression alterations that characterize the nematode's ageing process. The list of differentially expressed genes was then utilized to construct a network of molecular interactions with their first neighbors/interactors using the interactions listed in the WormBase database. The subsequent network analysis resulted in the unbiased selection of 110 candidate genes, among which well-known ageing regulators appeared. More importantly, our approach revealed candidates that have never been linked to ageing before, thus suggesting promising potential targets/ageing regulators.





Similar content being viewed by others
Data availability
The dataset supporting the conclusions of this article is available in the GEO repository, under the accession number GSE163253.
Code availability
The BioInfoMiner platform is available online at the website https://bioinfominer.com. Cytoscape is a free software available for download at https://cytoscape.org/download.html and all of its plugin applications are available at the Cytoscape App store https://apps.cytoscape.org/. R is available for download at the website https://cran.r-project.org/ and RStudio IDE also available at https://rstudio.com/products/rstudio/download/.
References
Ashburner M, Ball CA, Blake JA et al (2000) Gene ontology: tool for the unification of biology. Nat Genet 25:25–29
Assenov Y, Ramírez F, Schelhorn SESE et al (2008) Computing topological parameters of biological networks. Bioinformatics 24:282–284. https://doi.org/10.1093/bioinformatics/btm554
Azzi C, Aeschimann F, Neagu A, Großhans H (2020) A branched heterochronic pathway directs Juvenile-to-adult transition through two LIN-29 isoforms. Elife. https://doi.org/10.7554/eLife.53387
Barabási AL, Oltvai ZN (2004) Network biology: Understanding the cell’s functional organization. Nat Rev Genet 5:101–113
Beets I, Zhang G, Fenk LA et al (2020) Natural variation in a dendritic scaffold protein remodels experience-dependent plasticity by altering neuropeptide expression. Neuron 105:106-121.e10. https://doi.org/10.1016/j.neuron.2019.10.001
Benarroch R, Austin JM, Ahmed F, Isaacson RL (2019) The roles of cytosolic quality control proteins, SGTA and the BAG6 complex, in disease. In: Advances in Protein Chemistry and Structural Biology. Academic Press Inc., pp 265–313
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bis JC, Jian X, Kunkle BW et al (2020) Whole exome sequencing study identifies novel rare and common Alzheimer’s-Associated variants involved in immune response and transcriptional regulation. Mol Psychiatry 25:1859–1875. https://doi.org/10.1038/s41380-018-0112-7
Bishop NA, Guarente L (2007) Two neurons mediate diet-restriction-induced longevity in C. elegans. Nature 447:545–549. https://doi.org/10.1038/nature05904
Brenner S (1974) The genetics of Caenorhabditis elegans. Genetics 77:71–94
Budovskaya YV, Wu K, Southworth LK et al (2008) An elt-3/elt-5/elt-6 GATA Transcription Circuit Guides Aging in C. elegans. Cell 134:291–303. https://doi.org/10.1016/j.cell.2008.05.044
Budovsky A, Tacutu R, Yanai H et al (2009) Common gene signature of cancer and longevity. Mech Ageing Dev 130:33–39. https://doi.org/10.1016/j.mad.2008.04.002
Bunu G, Toren D, Ion CF et al (2020) SynergyAge, a curated database for synergistic and antagonistic interactions of longevity-associated genes. Sci Data. https://doi.org/10.1038/s41597-020-00710-z
C. elegans Sequencing Consortium (1998) Genome sequence of the nematode C. elegans: a platform for investigating biology. Science 282:2012–2018. https://doi.org/10.1126/science.282.5396.2012
Chang W, Tilmann C, Thoemke K et al (2004) A forkhead protein controls sexual identity of the C. elegans male somatic gonad. Development 131:1425–1436
Chew YL, Fan X, Götz J, Nicholas HR (2013) Aging in the nervous system of Caenorhabditis elegans. Commun. Integr. Biol. 6
Cho J, Grove C, Van Auken K, et al (2018) 2018 Update on Protein-Protein Interaction Data in WormBase. microPublication Biol https://doi.org/https://doi.org/10.17912/micropub.biology.000074
Chondrogianni N, Vasilopoulou MA, Kapetanou M, Gonos ES (2019) Proteasome modulation: A way to delay aging? In: Encyclopedia of Biomedical Gerontology. Elsevier, pp 92–104
Cordeiro Rodrigues RJ, De Jesus Domingues AM, Hellmann S et al (2019) PETISCO is a novel protein complex required for 21U RNA biogenesis and embryonic viability. Genes Dev. https://doi.org/10.1101/gad.322446.118
Curran SP, Ruvkun G (2007) Lifespan regulation by evolutionarily conserved genes essential for viability. PLoS Genet 3:0479–0487. https://doi.org/10.1371/journal.pgen.0030056
de Magalhães JP, Toussaint O (2004) GenAge: a genomic and proteomic network map of human ageing. FEBS Lett 571:243–247. https://doi.org/10.1016/j.febslet.2004.07.006
Denzel MS, Lapierre LR, Mack HID (2019) Emerging topics in C. elegans aging research: Transcriptional regulation, stress response and epigenetics. Mech Ageing Dev 177:4–21
Doncheva NT, Morris JH, Gorodkin J, Jensen LJ (2019) Cytoscape StringApp: network analysis and visualization of proteomics data. J Proteome Res 18:623–632. https://doi.org/10.1021/acs.jproteome.8b00702
Eckley DM, Rahimi S, Mantilla S et al (2013) Molecular characterization of the transition to mid-life in Caenorhabditis elegans. Age (Omaha) 35:689–703. https://doi.org/10.1007/s11357-012-9401-2
Eckley DM, Coletta CE, Orlov NV et al (2018) Transcriptome states reflect imaging of aging states. J Gerontol - Ser A Biol Sci Med Sci 73:893–901. https://doi.org/10.1093/gerona/glx236
Edifizi D, Nolte H, Babu V et al (2017) Multilayered Reprogramming In Response To Persistent DNA damage in C. elegans. Cell Rep 20:2026–2043. https://doi.org/10.1016/j.celrep.2017.08.028
Ertl I, Porta-De-La-Riva M, Gómez-Orte E et al (2016) Functional interplay of two paralogs encoding SWI/SNF chromatin-remodeling accessory subunits during Caenorhabditis elegans development. Genetics 202:961–975. https://doi.org/10.1534/genetics.115.183533
Feldman JL, Priess JR (2012) A role for the centrosome and PAR-3 in the hand-off of MTOC function during epithelial polarization. Curr Biol 22:575–582. https://doi.org/10.1016/j.cub.2012.02.044
Feng Y, Wang Q, Wang T (2017) Drug target protein-protein interaction networks: a systematic perspective. Biomed Res Int 2017
Firestein BL, Rongo C (2001) DLG-1 is a MAGUK similar to SAP97 and is required for adherens junction formation. Mol Biol Cell 12:3465–3475. https://doi.org/10.1091/mbc.12.11.3465
Friedman DB, Johnson TE (1988) A mutation in the age-1 gene in Caenorhabditis elegans lengthens life and reduces hermaphrodite fertility
Golden TR, Melov S (2004) Microarray analysis of gene expression with age in individual nematodes. Aging Cell 3:111–124. https://doi.org/10.1111/j.1474-9728.2004.00095.x
Golden TR, Melov S (2007) Gene expression changes associated with aging in C. elegans. WormBook 1–12
Golden TR, Hubbard A, Dando C et al (2008) Age-related behaviors have distinct transcriptional profiles in Caenorhabditis.elegans. Aging Cell 7:850–865. https://doi.org/10.1111/j.1474-9726.2008.00433.x
Greer EL, Maures TJ, Hauswirth AG et al (2010) Members of the H3K4 trimethylation complex regulate lifespan in a germline-dependent manner in C. elegans. Nature 466:383–387. https://doi.org/10.1038/nature09195
Grompone G, Martorell P, Llopis S et al (2012) Anti-inflammatory lactobacillus rhamnosus CNCM I-3690 strain protects against oxidative stress and increases lifespan in caenorhabditis elegans. PLoS One 7:e52493. https://doi.org/10.1371/journal.pone.0052493
Harris TW, Arnaboldi V, Cain S et al (2020) WormBase: a modern model organism information resource. Nucleic Acids Res 48:D762–D767. https://doi.org/10.1093/nar/gkz920
He L, Chen Y, Feng J et al (2017) Cellular senescence regulated by SWI/SNF complex subunits through p53/p21 and p16/pRB pathway. Int J Biochem Cell Biol 90:29–37. https://doi.org/10.1016/j.biocel.2017.07.007
Herndon LA, Wolkow CA, Driscoll M, Hall DH (2017) Effects of Ageing on the Basic Biology and Anatomy of C. elegans. 9–39. https://doi.org/https://doi.org/10.1007/978-3-319-44703-2_2
Herndon LA, Wolkow C, Hall DH (2018) WormAtlas Aging Handbook - Introduction to Aging in C. elegans. WormAtlas. https://doi.org/https://doi.org/10.3908/wormatlas.8.4
Hetherington S, Gally C, Fritz JA et al (2011) PAT-12, a potential anti-nematode target, is a new spectraplakin partner essential for Caenorhabditis elegans hemidesmosome integrity and embryonic morphogenesis. Dev Biol 350:267–278. https://doi.org/10.1016/j.ydbio.2010.11.025
Hou L, Wang D, Chen D et al (2016) A systems approach to reverse engineer lifespan extension by dietary restriction. Cell Metab 23:529–540. https://doi.org/10.1016/j.cmet.2016.02.002
Hsu AL, Murphy CT, Kenyon C (2003) Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300:1142–1145. https://doi.org/10.1126/science.1083701
Inukai S, Pincus Z, De Lencastre A, Slack FJ (2018) A microRNA feedback loop regulates global microRNA abundance during aging. RNA 24:159–172. https://doi.org/10.1261/rna.062190.117
Irizarry RA, Hobbs B, Collin F et al (2003) Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4:249–264. https://doi.org/10.1093/biostatistics/4.2.249
Irizarry RA, Warren D, Spencer F et al (2005) Multiple-laboratory comparison of microarray platforms. Nat Methods 2:345–349. https://doi.org/10.1038/nmeth756
Jaccard P (1901) Article in Bulletin de la Societe Vaudoise des Sciences Naturelles. Bull la Société Vaudoise des Sci Nat
Kenyon C (2011) The first long-lived mutants: discovery of the insulin/IGF-1 pathway for ageing. Philos Trans R Soc B Biol Sci 366:9–16. https://doi.org/10.1098/rstb.2010.0276
Kiehl TR, Shibata H, Pulst SM (2000) The ortholog of human ataxin-2 is essential for early embryonic patterning in C. elegans. J Mol Neurosci 15:231–241. https://doi.org/10.1385/JMN:15:3:231
Kim W, Underwood RS, Greenwald I, Shaye DD (2018) Ortholist 2: A new comparative genomic analysis of human and caenorhabditis elegans genes. Genetics 210:445–461. https://doi.org/10.1534/genetics.118.301307
Kirkwood TBL, Austad SN (2000) Why do we age? Nature 408:233–238
Koutrouli M, Karatzas E, Paez-Espino D, Pavlopoulos GA (2020) A guide to conquer the biological network era using graph theory. Front Bioeng Biotechnol 8:34
Koutsandreas T, Binenbaum I, Pilalis E et al (2016) Analyzing and visualizing genomic complexity for the derivation of the emergent molecular networks. Int J Monit Surveill Technol Res 4:30–49. https://doi.org/10.4018/ijmstr.2016040103
Kulkarni M, Shakes DC, Guevel K, Smith HE (2012) SPE-44 implements sperm cell fate. PLoS Genet. https://doi.org/10.1371/journal.pgen.1002678
Large EE, Mathies LD (2014) Caenorhabditis elegans SWI/SNF subunits control sequential developmental stages in the somatic gonad. G3 Genes. Genomes, Genet 4:471–483. https://doi.org/10.1534/g3.113.009852
Leclerc RD (2008) Survival of the sparsest: Robust gene networks are parsimonious. Mol Syst Biol 4:213. https://doi.org/10.1038/msb.2008.52
Li YH, Dong MQ, Guo Z (2010) Systematic analysis and prediction of longevity genes in Caenorhabditis elegans. Mech Ageing Dev 131:700–709. https://doi.org/10.1016/j.mad.2010.10.001
Li MM, Nilsen A, Shi Y et al (2013) ALKBH4-dependent demethylation of actin regulates actomyosin dynamics. Nat Commun. https://doi.org/10.1038/ncomms2863
Lionaki E, Tavernarakis N (2013) Assessing aging and senescent decline in Caenorhabditis elegans : Cohort survival analysis. Methods Mol Biol 965:473–484. https://doi.org/10.1007/978-1-62703-239-1_31
Liu F, Kuo WP, Jenssen TK, Hovig E (2012) Performance comparison of multiple microarray platforms for gene expression profiling. Methods Mol Biol 802:141–155. https://doi.org/10.1007/978-1-61779-400-1_10
Liu H, Wang S, Hang W et al (2018) LET-413/Erbin acts as a RAB-5 effector to promote RAB-10 activation during endocytic recycling. J Cell Biol 217:299–314. https://doi.org/10.1083/jcb.201705136
Lund J, Tedesco P, Duke K et al (2002) Transcriptional profile of aging in C. elegans. Curr Biol 12:1566–1573. https://doi.org/10.1016/S0960-9822(02)01146-6
Mansfeld J, Urban N, Priebe S et al (2015) Branched-chain amino acid catabolism is a conserved regulator of physiological ageing. Nat Commun 6:1–12. https://doi.org/10.1038/ncomms10043
Mao G, Zhang N (2013) Analysis of average shortest-path length of scale-free network. J Appl Math. https://doi.org/10.1155/2013/865643
Martin GM (2007) Modalities of gene action predicted by the classical evolutionary biological theory of aging. In: Annals of the New York Academy of Sciences. Blackwell Publishing Inc., pp 14–20
Martínez Corrales G, Alic N (2020) Evolutionary conservation of transcription factors affecting longevity. Trends Genet 36:373–382
Miwa T, Inoue K, Sakamoto H (2019) MRG-1 is required for both chromatin-based transcriptional silencing and genomic integrity of primordial germ cells in Caenorhabditis elegans. Genes Cells 24:377–389. https://doi.org/10.1111/gtc.12683
Mori MA, Raghavan P, Thomou T et al (2012) Role of microRNA processing in adipose tissue in stress defense and longevity. Cell Metab 16:336–347. https://doi.org/10.1016/j.cmet.2012.07.017
Murphy CT, McCarroll SA, Bargmann CI et al (2003) Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424:277–284. https://doi.org/10.1038/nature01789
Müthel S, Uyar B, He M et al (2019) The conserved histone chaperone LIN-53 is required for normal lifespan and maintenance of muscle integrity in Caenorhabditis elegans. Aging Cell. https://doi.org/10.1111/acel.13012
Neukomm LJ, Zeng S, Frei AP et al (2014) Small GTPase CDC-42 promotes apoptotic cell corpse clearance in response to PAT-2 and CED-1 in C. elegans. Cell Death Differ 21:845–853. https://doi.org/10.1038/cdd.2014.23
Niwa R, Zhou F, Li C, Slack FJ (2008) The expression of the Alzheimer’s amyloid precursor protein-like gene is regulated by developmental timing microRNAs and their targets in Caenorhabditis elegans. Dev Biol 315:418–425. https://doi.org/10.1016/j.ydbio.2007.12.044
Oliveros JC (2015) Venny 2.1.0. In: An Interact. tool Comp. List. with Venn´s diagrams. https://bioinfogp.cnb.csic.es/tools/venny/. Accessed 4 Feb 2021.
Panowski SH, Wolff S, Aguilaniu H et al (2007) PHA-4/Foxa mediates diet-restriction-induced longevity of C. elegans. Nature 447:550–555. https://doi.org/10.1038/nature05837
Pincus Z, Slack FJ (2008) Transcriptional (dys)regulation and aging in Caenorhabditis elegans. Genome Biol 9:233
Portman DS (2006) Profiling C. elegans gene expression with DNA microarrays. WormBook 1–11
Princz A, Pelisch F, Tavernarakis N (2020) SUMO promotes longevity and maintains mitochondrial homeostasis during ageing in Caenorhabditis elegans. Sci Rep 10:15513. https://doi.org/10.1038/s41598-020-72637-9
R Core Team (2020) R: A language and environment for statistical computing. R Found. Stat. Comput.
Ritchie ME, Phipson B, Wu D et al (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47. https://doi.org/10.1093/nar/gkv007
Rosas I, Martínez C, Clarimón J et al (2020) Role for ATXN1, ATXN2, and HTT intermediate repeats in frontotemporal dementia and Alzheimer’s disease. Neurobiol Aging 87:139.e1-139.e7. https://doi.org/10.1016/j.neurobiolaging.2019.10.017
Russell RL, Seppa RI (1987) Genetic and environmental manipulation of aging in Caenorhabditis elegans. Basic Life Sci 42:35–48. https://doi.org/10.1007/978-1-4613-1939-9_3
Samuelson AV, Carr CE, Ruvkun G (2007) Gene activities that mediate increased life span of C. elegans insulin-like signaling mutants. Genes Dev 21:2976–2994. https://doi.org/10.1101/gad.1588907
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Shaw WM, Luo S, Landis J et al (2007) The C. elegans TGF-β dauer pathway regulates longevity via insulin signaling. Curr Biol 17:1635–1645. https://doi.org/10.1016/j.cub.2007.08.058
Sheaffer KL, Updike DL, Mango SE (2008) The target of rapamycin pathway antagonizes pha-4/FoxA to control development and aging. Curr Biol 18:1355–1364. https://doi.org/10.1016/j.cub.2008.07.097
Shimomura Y, Ito M (2005) Human hair keratin-associated proteins. J Investig Dermatol Symp Proc 10:230–233. https://doi.org/10.1111/j.1087-0024.2005.10112.x
Tacutu R, Shore DE, Budovsky A et al (2012) Prediction of C. elegans longevity genes by human and worm longevity networks. PLoS One 7:e48282. https://doi.org/10.1371/journal.pone.0048282
Tacutu R, Thornton D, Johnson E et al (2018) Human ageing genomic resources: new and updated databases. Nucleic Acids Res 46:D1083–D1090. https://doi.org/10.1093/nar/gkx1042
Takeuchi T, Nemoto-Sasaki Y, Sugiura K et al (2013) Galectin LEC-1 plays a defensive role against damage due to oxidative stress in Caenorhabditis elegans. J Biochem 154:455–464. https://doi.org/10.1093/jb/mvt074
Tarkhov AE, Alla R, Ayyadevara S et al (2019) A universal transcriptomic signature of age reveals the temporal scaling of Caenorhabditis elegans aging trajectories. Sci Rep 9:1–18. https://doi.org/10.1038/s41598-019-43075-z
Tissenbaum HA (2015) Using C. elegans for aging research. Invertebr Reprod Dev 59:59–63
Tocchini C, Keusch JJ, Miller SB et al (2014) The TRIM-NHL Protein LIN-41 controls the onset of developmental plasticity in caenorhabditis elegans. PLoS Genet. https://doi.org/10.1371/journal.pgen.1004533
Tornieri K, Zlatic SA, Mullin AP et al (2013) Vps33b pathogenic mutations preferentially affect VIPAS39/SPE-39-positive endosomes. Hum Mol Genet 22:5215–5228. https://doi.org/10.1093/hmg/ddt378
Tóth ML, Sigmond T, Borsos É et al (2008) Longevity pathways converge on autophagy genes to regulate life span in Caenorhabditis elegans. Autophagy 4:330–338. https://doi.org/10.4161/auto.5618
Toth ML, Melentijevic I, Shah L et al (2012) Neurite sprouting and synapse deterioration in the aging Caenorhabditis elegans nervous system. J Neurosci 32:8778–8790. https://doi.org/10.1523/JNEUROSCI.1494-11.2012
Tsang WY, Sayles LC, Grad LI et al (2001) Mitochondrial respiratory chain deficiency in caenorhabditis elegans results in developmental arrest and increased life span. J Biol Chem 276:32240–32246. https://doi.org/10.1074/jbc.M103999200
Uno M, Honjoh S, Matsuda M et al (2013) a fasting-responsive signaling pathway that extends life span in C. elegans. Cell Rep 3:79–91. https://doi.org/10.1016/j.celrep.2012.12.018
Van Voorhies WA, Ward S (1999) Genetic and environmental conditions that increase longevity in Caenorhabditis elegans decrease metabolic rate. Proc Natl Acad Sci USA 96:11399–11403. https://doi.org/10.1073/pnas.96.20.11399
Van Gilst MR, Hadjivassiliou H, Jolly A, Yamamoto KR (2005) Nuclear hormone receptor NHR-49 controls fat consumption and fatty acid composition in C. elegans. PLoS Biol 3:0301–0312. https://doi.org/10.1371/journal.pbio.0030053
Wang XF, Chen G (2003) Complex networks: Small-world, scale-free and beyond. IEEE Circuits Syst Mag 3:6–20
Wang BY, Xu XS, Cui YX et al (2014) Caenorhabditis elegans eyes absent ortholog EYA-1 Is required for stress resistance. Biochem 79:653–662. https://doi.org/10.1134/S0006297914070074
Wang SY, Mao H, Shibuya H et al (2019) The demethylase NMAD-1 regulates DNA replication and repair in the Caenorhabditis elegans germline. PLoS Genet. https://doi.org/10.1371/journal.pgen.1008252
Witten TM, Bonchev D (2007) Predicting aging/longevity-related genes in the nematode Caenorhabditis elegans. Chem Biodivers 4:2639–2655. https://doi.org/10.1002/cbdv.200790216
Yanai H, Budovsky A, Barzilay T et al (2017) Wide-scale comparative analysis of longevity genes and interventions. Aging Cell 16:1267–1275. https://doi.org/10.1111/acel.12659
Youngman MJ, Rogers ZN, Kim DH (2011) A decline in p38 MAPK signaling underlies immunosenescence in caenorhabditis elegans. PLoS Genet. https://doi.org/10.1371/journal.pgen.1002082
Zhang Y, Shao Z, Zhai Z et al (2009) The HIF-1 hypoxia-inducible factor modulates lifespan in C elegans. PLoS One. https://doi.org/10.1371/journal.pone.0006348
Zhu GD, L’Hernault SW (2003) The Caenorhabditis elegans spe-39 gene is required for intracellular membrane reorganization during spermatogenesis. Genetics
Acknowledgements
We acknowledge financial support from the European Union and Greek national funds through the Operational Program Competitiveness, Entrepreneurship and Innovation, under the call RESEARCH—CREATE—INNOVATE (project code: T1EDK-01610) to NC. BioInfoMiner is a product developed by e-NIOS Applications PC, for the automated functional interpretation of various omics data streams.
Funding
Financial support was received from the European Union and Greek national funds through the Operational Program Competitiveness, Entrepreneurship and Innovation, under the call RESEARCH—CREATE—INNOVATE (project code: T1EDK-01610) to NC. BioInfoMiner is a product developed by e-NIOS Applications PC, for the automated functional interpretation of various omics data streams.
Author information
Authors and Affiliations
Contributions
FA, KV and NC contributed to the study conception and design. Material preparation and data collection were performed by NP. Data analysis was performed by FA. NC, KV and AC supervised the research. FA and NC wrote the manuscript, review and editing were performed by all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
AC is the Chief Executive Officer of e-NIOS Applications PC.
Ethical Approval
Compliance with ethical standards.
Consent for publication
All authors read and approved the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10522_2021_9920_MOESM1_ESM.eps
Online Resource 1 Principal component analysis (PCA) of individual samples in the dataset GSE21784. The data suggest that ageing is a characteristic transcriptomic drift along a single direction while PC1 and PC2 accounted for 91.7% of variance observed (EPS 443 kb)
10522_2021_9920_MOESM2_ESM.xlsx
Online Resource 2 Tables of the differentially expressed genes in ageing organized into upregulated and downregulated genes in every comparison based on the corresponding log2-fold change values (XLSX 1906 kb)
10522_2021_9920_MOESM3_ESM.xlsx
Online Resource 3 Tables of the common differentially expressed genes in ageing found between the two datasets in the following comparisons of gene expression: day 12 vs day 6 (GSE163253) and day 15 vs day 6 (GSE21784) (XLSX 311 kb)
10522_2021_9920_MOESM4_ESM.xlsx
Online Resource 4 Tables of BioInfoMiner functional enrichment analysis results for the upregulated and downregulated differentially expressed genes of every comparison of gene expression (XLSX 29 kb)
10522_2021_9920_MOESM5_ESM.sif
Online Resource 5 Network of the common differentially expressed genes between the two datasets in old animals compared to middle aged ones and their first neighbors based on WormBase interactions (SIF 859 kb)
10522_2021_9920_MOESM7_ESM.xlsx
Online Resource 7 Table of StringApp functional enrichment analysis results for the genes highlighted as candidate ageing regulators (XLSX 31 kb)
10522_2021_9920_MOESM8_ESM.cys
Supporting data 1 Detailed representations of the networks created and analyzed in this study. The networks in this Cytoscape session file can be viewed with Cytoscape, a free software available for download at the website https://cytoscape.org/download.html (cys 4198 kb)
Rights and permissions
About this article
Cite this article
Aktypi, F., Papaevgeniou, N., Voutetakis, K. et al. Network analysis in aged C. elegans reveals candidate regulatory genes of ageing. Biogerontology 22, 345–367 (2021). https://doi.org/10.1007/s10522-021-09920-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-021-09920-3