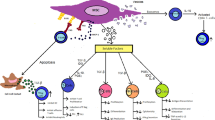
Mesenchymal stem cells (MSC) are characterized by tolerogenic potential and therefore, are used in the treatment of autoimmune diseases such as graft-versus-host disease (GVHD) reactions after allogeneic hematopoietic cell transplantation to improve the transplant functions, as well as for the therapy and prevention of cytokine storm in COVID-19 patients and some other conditions. However, MSC can exhibit proinflammatory activity, which causes risks for their clinical use. We studied the cytokine profile of bone marrow MSC culture and demonstrate intensive production of IL-6, IL-8, and chemokine MCP-1, which participate in the pathogenesis of cytokine storm and GVHD. At the same time, no anti-inflammatory IL-4 and IL-10 were detected. To reduce the risks of MSC application in the GVHD therapeutic protocols, further studies of the conditions promoting generation of MSC with tolerogenic potential and approved clinical standards of MSC use are required.
Article PDF
Similar content being viewed by others
References
Bernardo ME, Fibbe WE. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell. 2013;13(4):392- 402. doi: https://doi.org/10.1016/j.stem.2013.09.006
Chan CK, Wu KH, Lee YS, Hwang SM, Lee MS, Liao SK, Cheng EH, See LC, Tsai CN, Kuo ML, Huang JL. The comparison of interleukin 6-associated immunosuppressive effects of human ESCs, fetal-type MSC, and adult-type MSC. Transplantation. 2012;94(2):132-138. doi: https://doi.org/10.1097/TP.0b013e31825940a4
Clinical Commissioning Policy: Treatments for Graft versus Host Disease (GvHD) following Haematopoietic Stem Cell Transplantation. 31 March 2017. URL: https://www.england.nhs.uk/wp-content/uploads/2017/03/gvhd-heamatopoieticstem- cell.pdf
Connard SS, Linardi RL, Even KM, Berglund AK, Schnabel LV, Ortved KF. Effects of continuous passage on the immunomodulatory properties of equine bone marrow-derived mesenchymal stem cells in vitro. Vet. Immunol. Immunopathol. 2021;234:110203. doi: https://doi.org/10.1016/j.vetimm.2021.110203
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317. doi: https://doi.org/10.1080/14653240600855905
Drobyski WR, Pasquini M, Kovatovic K, Palmer J, Douglas Rizzo J, Saad A, Saber W, Hari P. Tocilizumab for the treatment of steroid refractory graft-versus-host disease. Biol. Blood Marrow Transplant. 2011;17(12):1862-1868. doi: https://doi.org/10.1016/j.bbmt.2011.07.001
Ferrà C, de Sanjosé S, Gallardo D, Berlanga JJ, Rueda F, Marìn D, de la Banda E, Ancìn I, Peris J, Garcìa J, Grañena A. IL-6 and IL-8 levels in plasma during hematopoietic progenitor transplantation. Haematologica. 1998;83(12):1082- 1087.
Gazdic M, Volarevic V, Arsenijevic N, Stojkovic M. Mesenchymal stem cells: a friend or foe in immune-mediated diseases. Stem Cell Rev. Rep. 2015;11(2):280-287. doi: https://doi.org/10.1007/s12015-014-9583-3
Gu L, Tseng S, Horner RM, Tam C, Loda M, Rollins BJ. Control of TH2 polarization by the chemokine monocyte chemoattractant protein-1. Nature. 2000;404:407-411. doi: https://doi.org/10.1038/35006097
Kiselevskiy M, Shubina I, Chikileva I, Sitdikova S, Samoylenko I, Anisimova N, Kirgizov K, Suleimanova A, Gorbunova T, Varfolomeeva S. Immune pathogenesis of COVID-19 intoxication: storm or silence? Pharmaceuticals (Basel). 2020; 13(8):166. doi: https://doi.org/10.3390/ph13080166.
Kiselevskiy M, Vlasenko R, Reshetnikova V, Chikileva I, Shubina I, Osmanov E, Valiev T, Sidorova N, Batmanova N, Stepanyan N, Kirgizov K, Varfolomeeva S. Potential use of mesenchymal multipotent cells for hemopoietic stem cell transplantation: pro and contra. J. Pediatr. Hematol. Oncol. 2021;43(3):90-94. doi: https://doi.org/10.1097/MPH.0000000000002065
Kyurkchiev D, Bochev I, Ivanova-Todorova E, Mourdjeva M, Oreshkova T, Belemezova K, Kyurkchiev S. Secretion of immunoregulatory cytokines by mesenchymal stem cells. World J. Stem Cells. 2014;6(5):552-570. doi: https://doi.org/10.4252/wjsc.v6.i5.552.
Le Blanc K, Ringdén O. Immunobiology of human mesenchymal stem cells and future use in hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2005;11(5):321- 334. doi: https://doi.org/10.1016/j.bbmt.2005.01.005
Li T, Luo C, Zhang J, Wei L, Sun W, Xie Q, Liu Y, Zhao Y, Xu S, Wang L. Efficacy and safety of mesenchymal stem cells co-infusion in allogeneic hematopoietic stem cell transplantation: a systematic review and meta-analysis. Stem Cell Res Ther. 2021;12(1):246. doi: https://doi.org/10.1186/s13287-021-02304-x
Ma S, Xie N, Li W, Yuan B, Shi Y, Wang Y. Immunobiology of mesenchymal stem cells. Cell Death Differ. 2014;21(2):216- 225. doi: https://doi.org/10.1038/cdd.2013.158.
Martin PJ, Uberti JP, Soiffer RJ, Klingemann H, Waller EK, Daly AS, Herrmann RP, Kebriaei P. Prochymal improves response rates in patients with steroid-refractory acute graft versus host disease (SR-GVHD) involving the liver and gut: results of a randomized, placebo-controlled, multicenter phase III trial in GVHD. Biol. Blood Marrow Transplant. 2010;16(2):S169-170. doi: https://doi.org/10.1016/j.bbmt.2009.12.057
McElvaney OJ, Curley GF, Rose-John S, McElvaney NG. Interleukin-6: obstacles to targeting a complex cytokine in critical illness. Lancet Respir. Med. 2021;9(6):643-654. doi: https://doi.org/10.1016/S2213-2600(21)00103-X
Pricola KL, Kuhn NZ, Haleem-Smith H, Song Y, Tuan RS. Interleukin-6 maintains bone marrow-derived mesenchymal stem cell stemness by an ERK1/2-dependent mechanism. J. Cell. Biochem. 2009;108(3):577-588. doi: https://doi.org/10.1002/jcb.22289
Siegel G, Schäfer R, Dazzi F. The immunosuppressive properties of mesenchymal stem cells. Transplantation. 2009;87(9, Suppl):S45-S49. doi: https://doi.org/10.1097/TP.0b013e3181a285b0
Steensberg A, Fischer CP, Keller C, Møller K, Pedersen BK. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Endocrinol. Metab. 2003;285(2):E433-E437. doi: https://doi.org/10.1152/ajpendo.00074.2003
Szabolcs P, Visani G, Locatelli F, Kleiner G, Talano J, Nemecek E, Kurtzberg J. Treatment of steroid-refractory acute GVHD with mesenchymal stem cells improves outcomes in pediatric patients; Results of the pediatric subset in a phase III randomized, placebo-controlled study. Biol. Blood Marrow Transplant. 2010;16(2):S298. doi: https://doi.org/10.1016/j.bbmt.2009.12.426
Wu X, Chen L, Xu Y, Wen J, Ruan Y, He Y, Li C. The relationship between some cytokines and graft versus host disease after allogeneic hematopoietic stem cell transplantation in thalassemia major patients. Blood. 2015. 126(23):5471. doi: https://doi.org/10.1182/blood.V126.23.5471.5471
Zumla A, Wang FS, Ippolito G, Petrosillo N, Agrati C, Azhar EI, Chang C, El-Kafrawy SA, Osman M, Zitvogel L, Galle PR, Locatelli F, Gorman E, Cordon-Cardo C, O’Kane C, McAuley D, Maeurer M. Reducing mortality and morbidity in patients with severe COVID-19 disease by advancing ongoing trials of Mesenchymal Stromal (stem) Cell (MSC) therapy — Achieving global consensus and visibility for cellular hostdirected therapies. Int. J. Infect. Dis. 2020;96:431-439. doi: https://doi.org/10.1016/j.ijid.2020.05.040
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 3, pp. 171-175, September, 2021
Rights and permissions
About this article
Cite this article
Kiselevskii, M.V., Vlasenko, R.Y., Stepanyan, N.G. et al. Secretome of Mesenchymal Bone Marrow Stem Cells: Is It Immunosuppressive or Proinflammatory?. Bull Exp Biol Med 172, 250–253 (2021). https://doi.org/10.1007/s10517-021-05371-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05371-5