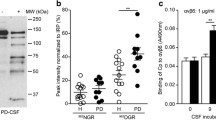
Serum concentration of copper, immunoreactive polypeptides of ceruloplasmin and its oxidase activity, and the number of copper atoms per ceruloplasmin molecule were decreased in patients with Parkinson’s disease in comparison with the corresponding parameters in age-matched healthy individuals, but the ratio of apoceruloplasmin to holoceruloplasmin in patients with Parkinson’s disease was similar in both groups. Treatment of blood serum with Helex 100, a high-affinity copper chelator, revealed reduced content of labile copper atoms per ceruloplasmin molecule in patients with Parkinson’s disease in comparison with that in healthy controls. The mechanism underlying impaired metabolic incorporation of labile copper atoms into CP molecule is discussed as a possible cause of copper dyshomeostasis associated with Parkinson’s disease.
Similar content being viewed by others
References
Sokolov AV, Kostevich VA, Romanico DN, Zakharova ET, Vasilyev VB. Two-stage method for purification of ceruloplasmin based on its interaction with neomycin. Biochemistry (Moscow). 2012;77(6):631-638.
Bento I, Peixoto C, Zaitsev VN, Lindley PF. Ceruloplasmin revisited: structural and functional roles of various metal cation-binding sites. Acta Crystallogr. D Biol. Crystallogr. 2007;63(Pt 2):240-248.
Dusek P, Roos PM, Litwin T, Schneider SA, Flaten TP, Aaseth J. The neurotoxicity of iron, copper and manganese in Parkinson’s and Wilson’s diseases. J. Trace Elem. Med. Biol. 2015;31:193-203.
Ilyechova EY, Saveliev AN, Skvortsov AN, Babich PS, Zatulovskaia YA, Pliss MG, Korzhevskii DE, Tsymbalenko NV, Puchkova L.V. The effects of silver ions on copper metabolism in rats. Metallomics. 2014;6(10):1970-1987.
Itoh S, Ozumi K, Kim HW, Nakagawa O, McKinney RD, Folz RJ, Zelko IN, Ushio-Fukai M, Fukai T. Novel mechanism for regulation of extracellular SOD transcription and activity by copper: role of antioxidant-1. Free Radic. Biol. Med. 2009;46(1):95-104.
Lutsenko S, Tsivkovskii R, Walker JM. Functional properties of the human copper-transporting ATPase ATP7B (the Wilson’s disease protein) and regulation by metallochaperone Atox1. Ann. N.Y. Acad. Sci. 2003;986:204-211.
Mukhopadhyay CK, Mazumder B, Lindley PF, Fox PL. Identification of the prooxidant site of human ceruloplasmin: A model for oxidative damage by copper bound to protein surfaces. Proc. Natl Acad. Sci. USA. 1997;94(21):11,546-11,551.
Polishchuk R, Lutsenko S. Golgi in copper homeostasis: a view from the membrane trafficking field. Histochem. Cell Biol. 2013;140(3):285-295.
Redenšek S, Trošt M, Dolžan V. Genetic determinants of Parkinson’s disease: can they help to stratify the patients based on the underlying molecular defect? Front. Aging Neurosci. 2017;9:20. doi: https://doi.org/10.3389/fnagi.2017.00020.
Valensin D, Dell’Acqua S, Kozlowski H, Casella L. Coordination and redox properties of copper interaction with α-synuclein. J. Inorg. Biochem. 2016;163:292-300.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 164, No. 11, pp. 547-551, November, 2017
Rights and permissions
About this article
Cite this article
Karpenko, M.N., Ilyicheva, E.Y., Muruzheva, Z.M. et al. Role of Copper Dyshomeostasis in the Pathogenesis of Parkinson’s Disease. Bull Exp Biol Med 164, 596–600 (2018). https://doi.org/10.1007/s10517-018-4039-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-018-4039-4