Abstract
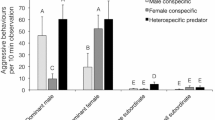
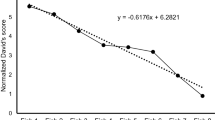
Domestication is still a long and difficult process and it is particularly impacted by species behavioural traits. Indeed, tolerance to high densities in intensive cultures and sociability are major features which facilitate domestication and influence the effectiveness of aquaculture production. Moreover, behavioural domestication predispositions could change at the intraspecific level. Here, we investigate three essential behavioural traits: aggressive interactions, group structure and activity between three allopatric populations of Perca fluviatilis, a fish species at its nascent stage of production. We highlight inter-populational differences in group structure and aggressive interactions but not in activity. A more cohesive and homogeneous group structure was demonstrated for Finnish populations compared to Lake Geneva at 45–46 days post-hatching. In addition, Lake Geneva presented a higher aggressiveness. These inter-populational differences could be used in European perch aquaculture in order to improve production as well as welfare of individuals.


Similar content being viewed by others
Change history
19 August 2019
This article belongs to the special issue European Percid Fish Culture by Żarski et al. and should be included in Volume 27, Issue 4. The link to this special issue can be found here: <ExternalRef><RefSource>https://link.springer.com/journal/10499/27/4</RefSource><RefTarget Address="https://link.springer.com/journal/10499/27/4" TargetType="URL"/></ExternalRef>
Abbreviations
- GEN:
-
Lake Geneva
- VAL:
-
Lake Valkea-Müstajärvi
- ISO:
-
Lake Iso-Valkjärvi
- RAS:
-
Recirculated aquaculture system
References
Amundsen P-A, Svenning M-A, Slikavuoplo SI (1999) An experimental comparison of cannibalistic response in different Arctic charr (Salvelinus alpinus (L.)) stocks. Ecol Freshw Fish 8:43–48. https://doi.org/10.1111/j.1600-0633.1999.tb00051.x
Ashley PJ (2007) Fish welfare: current issues in aquaculture. Appl Anim Behav Sci 104:199–235. https://doi.org/10.1016/j.applanim.2006.09.001
Babiak I, Mandiki SNM, Ratsinjomanana K, Kestemont P (2004) Initial weight and its variation in post-larval Eurasian perch affect quantitative characteristics of juvenile cohorts under controlled conditions. Aquaculture 234:263–276. https://doi.org/10.1016/j.aquaculture.2003.12.010
Baras E (2013) Cannibalism in fish larvae: what have we learned? In: Qin JG (ed) larval fish aquaculture. 167–199
Baras E, Jobling M (2002) Dynamics of intracohort cannibalism in cultured fish. Aquac Res 33:461–479. https://doi.org/10.1046/j.1365-2109.2002.00732.x
Baras E, Kestemont P, Mélard C, Mélard C (2003) Effect of stocking density on the dynamics of cannibalism in sibling larvae of Perca fluviatilis under controlled conditions. Aquaculture 219:241–255. https://doi.org/10.1016/S0044-8486(02)00349-6
Bates D, Maechler M, Bolker B, Walker S (2004) lme4: linear mixed-effects models using Eigen and S4
Behrmann-Godel J, Gerlach G, Eckmann R (2006) Kin and population recognition in sympatric Lake Constance perch (Perca fluviatilis L.): can assortative shoaling drive population divergence? Behav Ecol Sociobiol 59:461–468. https://doi.org/10.1007/s00265-005-0070-3
Bell AM (2004) Behavioural differences between individuals and two populations of stickleback (Gasterosteus aculeatus). J Evol Biol 18:464–473. https://doi.org/10.1111/j.1420-9101.2004.00817.x
Boisclair D, Leggett WC (1989) The importance of activity in bioenergetics models applied to actively foraging fishes. Can J Fish Aquat Sci 46:1859–1867. https://doi.org/10.1139/f89-234
Buske C, Gerlai R (2011a) Early embryonic ethanol exposure impairs shoaling and the dopaminergic and serotoninergic systems in adult zebrafish. Neurotoxicol Teratol 33:698–707. https://doi.org/10.1016/j.ntt.2011.05.009
Buske C, Gerlai R (2011b) Shoaling develops with age in zebrafish (Danio rerio). Prog Neuro-Psychopharmacol Biol Psychiatry 35:1409–1415. https://doi.org/10.1016/j.pnpbp.2010.09.003
Buske C, Gerlai R (2012) Maturation of shoaling behavior is accompanied by changes in the dopaminergic and serotoninergic systems in zebrafish. Dev Psychobiol 54:28–35. https://doi.org/10.1002/dev.20571
Colchen T, Teletchea F, Fontaine P, Pasquet A (2016) Temperature modifies activity, inter-individual relationships and group structure in fish. Curr Zool:1–9. https://doi.org/10.1093/cz/zow048
Colchen T, Fontaine P, Ledoré Y, Teletchea F, Pasquet A (2019) Intra-cohort cannibalism in early life stages of pikeperch. Aquac Res. https://doi.org/10.1111/are.13966
Damsgård B, Huntingford F (2012) Fighting and aggression. In: Aquaculture and behavior. Wiley-Blackwell, Oxford, pp 248–285
DeWitt TJ, Scheiner SM (2004) Phenotypic variation from single genotypes: a primer. In phenotypic plasticity: functional and conceptual approaches. Oxford University Press, New York, NY
Diamond J (2002) Evolution, consequences and future of plant and animal domestication. Nature 418:700–707. https://doi.org/10.1038/nature01019
Fiogbé ED, Kestemont P (2003) Optimum daily ration for Eurasian perch Perca fluviatilis L. reared at its optimum growing temperature. Aquaculture 216:243–252. https://doi.org/10.1016/S0044-8486(02)00409-X
Foster SA (1999) The geography of behaviour: an evolutionary perspective. Trends Ecol Evol 14:190–195
Foster SA, Endler JA (1999) Geographic variation in behavior: perspectives on evolutionary mechanisms. Oxford University Press
Gastwirth JL, Gel YR, Hui W, Lyubchich V, Miao W, Nogushi K (2015) Lawstat - tools for biostatistics, Public Policy, and Law
Gepts P, Famula TR, Bettinger RL et al (2012) Biodiversity in agriculture domestication, evolution, and sustainability. Cambridge University Press, Davis
Graeb BD, Galarowicz T, Wahl DH et al (2005) Foraging behavior, morphology, and life history variation determine the ontogeny of piscivory in two closely related predators. Can J Fish Aquat Sci 62:2010–2020. https://doi.org/10.1139/f05-112
Guma’a SA (1982) Retinal development and retinomotor responses in perch, Perca fluviatilis L. J Fish Biol 20:611–618. https://doi.org/10.1111/j.1095-8649.1982.tb03960.x
Hellström G, Magnhagen C (2011) The influence of experience on risk taking: results from a common-garden experiment on populations of Eurasian perch. Behav Ecol Sociobiol 65:1917–1926. https://doi.org/10.1007/s00265-011-1201-7
Hinde RA (1976) Interactions, relationships and social structure. Man 11:1–17. https://doi.org/10.2307/2800384
Huizinga M, Ghalambor CK, Reznick DN (2009) The genetic and environmental basis of adaptive differences in shoaling behaviour among populations of Trinidadian guppies, Poecilia reticulata. J Evol Biol 22:1860–1866. https://doi.org/10.1111/j.1420-9101.2009.01799.x
Huntingford FA (2004) Implications of domestication and rearing conditions for the behaviour of cultivated fishes. J Fish Biol 65:122–142. https://doi.org/10.1111/j.0022-1112.2004.00562.x
Huntingford FA (1982) Do inter- and intraspecific aggression vary in relation to predation pressure in sticklebacks? Anim Behav 30:909–916. https://doi.org/10.1016/S0003-3472(82)80165-6
Huntingford F, Jobling M, Kadri S (2012) Wiley InterScience (Online service) Aquaculture and behavior. John Wiley & Sons
Imsland AK, Foss A, Naevdal G et al (2000) Countergradient variation in growth and food conversion efficiency of juvenile turbot. J Fish Biol 57:1213–1226. https://doi.org/10.1006/jfbi.2000.1384
Imsland AK, Jonassen TM, Langston A, Hoare R, Wergeland H, FitzGerald R, Mulcahy M, Stefansson SO (2002) The interrelation of growth and disease resistance of different populations of juvenile Atlantic halibut (Hippoglossus hippoglossus L.). Aquaculture 204:167–177. https://doi.org/10.1016/S0044-8486(01)00656-1
Jobling M (2010) Farmed species and their characteristics. In: Le François N, Jobling M, Carter C, Blier P (eds) Finfish aquaculture diversification, CABI. pp 88–99
Jonassen TM, Imsland AK, Fitzgerald R et al (2000) Geographic variation in growth and food conversion efficiency of juvenile Atlantic halibut related to latitude. J Fish Biol 56:279–294. https://doi.org/10.1006/jfbi.1999.1159
Jourdan S, Fontaine P, Boujard T, Vandeloise E, Gardeur JN, Anthouard M, Kestemont P (2000) Influence of daylength on growth, heterogeneity, gonad development, sexual steroid and thyroid levels, and N and P budgets in Perca fluviatilis. Aquaculture 186:253–265
Kelly SA, Panhuis TM, Stoehr AM (2012) Phenotypic plasticity: molecular mechanisms and adaptive significance. In: Comprehensive physiology. John Wiley & Sons, Inc., Hoboken, pp 1417–1439
Kestemont P, Dabrowski K, Summerfelt RC (2015) Biology and culture of percid fishes: principles and practices
Kestemont P, Jourdan S, Houbart M, Mélard C, Paspatis M, Fontaine P, Cuvier A, Kentouri M, Baras E (2003) Size heterogeneity, cannibalism and competition in cultured predatory fish larvae: biotic and abiotic influences. Aquaculture 227:333–356. https://doi.org/10.1016/S0044-8486(03)00513-1
Kestemont P, Mélard C (2000) Chap. 11—aquaculture. In: Craig JF (ed) percid fishes. Systematics, ecology and exploitation. Blackwell science, Oxford, pp 191–224
Kestemont P, Mélard C, Fiogbe E, Vlavonou R, Masson G (1996) Nutritional and animal husbandry aspects of rearing early life stages of Eurasian perch Perca fluviatilis. J Appl Ichthyol 12:157–165. https://doi.org/10.1111/j.1439-0426.1996.tb00082.x
Kestemont P, Rougeot MCJ, Toner D (2008) Larval and juvenile production. Farming Eurasian Perch Spec Publ BIM 24:30–41
Kohane MJ, Parsons PA (1988) Domestication: evolutionary change under stress. Evol Biol 23:31–48
Kristiansen TS, Fernö A, Holm JC, Privitera L, Bakke S, Fosseidengen JE (2004) Swimming behaviour as an indicator of low growth rate and impaired welfare in Atlantic halibut (Hippoglossus hippoglossus L.) reared at three stocking densities. Aquaculture 230:137–151. https://doi.org/10.1016/S0044-8486(03)00436-8
Krol J, Dauchot N, Mandiki SNM et al (2015) Cannibalism in cultured Eurasian perch, Perca fluviatilis (Actinopterygii: Perciformes: Percidae) - implication of maternal influence, kinship, and sex ratio of progenies. Acta Ichthyol Piscat 45:65–73
Lahti K, Laurila A, Enberg K, Piironen J (2001) Variation in aggressive behaviour and growth rate between populations and migratory forms in the brown trout, Salmo trutta. Anim Behav 62:935–944. https://doi.org/10.1006/anbe.2001.1821
Le François NR, Otton D, Werstink G (2010) Considerations for the selection and commercialization of new or alternate species. In: Jobling M, Carter C, Blier P (eds) Le François N. CABI, Finfish Aquaculture Diversification, pp 100–114
Lecocq T (2018) Insects: the disregarded domestication histories. In: Teletchea F (ed) Animal domestication. IntechOpen. https://doi.org/10.5772/intechopen.81834
Leithner S, Wanzenböck J (2015) Rearing larvae of different strains of Coregonus lavaretus under cold water conditions: comparison of a special cold-water line with a standard agglomerated microdiet. J Agric Sci 7:28–36. https://doi.org/10.5539/jas.v7n5p28
Liao IC, Huang YS (2000) Methodological approach used for the domestication of potential candidates for aquaculture. Recent Adv Mediterr Aquac finfish species Diversif - Cah Options Méditerranéennes 47:97–107
Magnhagen C (2006) Risk-taking behaviour in foraging young-of-the-year perch varies with population size structure. Oecologia 147:734–743. https://doi.org/10.1007/s00442-005-0302-2
Magnhagen C (2015) Behaviour of percid fishes in the wild and its relevance for culture. In: Biology and culture of percid fishes. Springer Netherlands, Dordrecht, pp 399–416
Magnhagen C, Verli BT et al (2015) Behavioural responses in a net restraint test predict interrenal reactivity in Arctic charr Salvelinus alpinus. J Fish Biol 87:88–99. https://doi.org/10.1111/jfb.12691
Magurran AE, Seghers BH (1991) Variation in schooling and aggression amongst guppy (Poecilia reticulata) populations in Trinidad. Behaviour 118:214–234. https://doi.org/10.1163/156853991X00292
Mandiki SNM, Blanchard G, Mélard C, Koskela J, Kucharczyk D, Fontaine P, Kestemont P (2004) Effects of geographic origin on growth and food intake in Eurasian perch (Perca fluviatilis L.) juveniles under intensive culture conditions. Aquaculture 229:117–128. https://doi.org/10.1016/S0044-8486(03)00359-4
Martins CIM, Galhardo L, Noble C, Damsgård B, Spedicato MT, Zupa W, Beauchaud M, Kulczykowska E, Massabuau JC, Carter T, Planellas SR, Kristiansen T (2012) Behavioural indicators of welfare in farmed fish. Fish Physiol Biochem 38:17–41. https://doi.org/10.1007/s10695-011-9518-8
Mayr E (1963) Animal species and evolution. In: animal species and their evolution. Belknap Pr. Harvard University Press, Cambridge, Massachusetts, p 797
Mousseau TA, Fox CW (1998) The adaptive significance of maternal effects. Trends Ecol Evol 13:403–407. https://doi.org/10.1016/S0169-5347(98)01472-4
Naumowicz K, Pajdak J, Terech-Majewska E, Szarek J (2017) Intracohort cannibalism and methods for its mitigation in cultured freshwater fish. Rev Fish Biol Fish 27:193–208. https://doi.org/10.1007/s11160-017-9465-2
Olin M, Jutila J, Lehtonen H, Vinni M, Ruuhijärvi J, Estlander S, Rask M, Kuparinen A, Lappalainen J (2012) Importance of maternal size on the reproductive success of perch, Perca fluviatilis, in small forest lakes: implications for fisheries management. Fish Manag Ecol 19:363–374. https://doi.org/10.1111/j.1365-2400.2012.00845.x
Otton DD (2004) Establishment of an agribusiness model for assessing the commercial viability of new species for aquaculture. PhD Thesis. University of Tasmania
Overturf K, Casten MT, LaPatra SL et al (2003) Comparison of growth performance, immunological response and genetic diversity of five strains of rainbow trout (Oncorhynchus mykiss). Aquaculture 217:93–106. https://doi.org/10.1016/S0044-8486(02)00014-5
Pigliucci M, Murren CJ, Schlichting CD (2006) Phenotypic plasticity and evolution by genetic assimilation. J Exp Biol 209:2362–2367. https://doi.org/10.1242/jeb.02070
Pitcher TJ, Parrish JK (1993) Functions of shoaling behaviour in teleosts. In: Pitcher TJ (ed) Behaviour of teleost fishes. Chapman & Hall, London, pp 363–439
Pohlert T (2015) PMCMR: calculate pairwise multiple comparisons of mean rank sums
R Core Team (2017) R: a language and environment for statistical computing
Rosenau ML, McPhail JD (1987) Inherited differences in agonistic behavior between two populations of Coho Salmon. Trans Am Fish Soc 116:646–654. https://doi.org/10.1577/1548-8659(1987)116<646:IDIABB>2.0.CO;2
Song Z, Boenke MC, Rodd FH (2011) Interpopulation differences in shoaling behaviour in guppies (Poecilia reticulata): roles of social environment and population origin. Ethology 117:1009–1018. https://doi.org/10.1111/j.1439-0310.2011.01952.x
Svenning M-A, Borgstrøm DR (2005) Cannibalism in Arctic charr: do all individuals have the same propensity to be cannibals? J Fish Biol 66:957–965. https://doi.org/10.1111/j.1095-8649.2005.00646.x
Swain DP, Lindsey CC (1986) Meristic variation in a clone of the cyprinodont fish Rivulus marmoratus related to temperature history of the parents and of the embryos. Can J Zool 64:1444–1455. https://doi.org/10.1139/z86-216
Tamazouzt L, Chatain B, Fontaine P (2000) Tank wall colour and light level affect growth and survival of Eurasian perch larvae (Perca fluviatilis L.). Aquaculture 182:85–90. https://doi.org/10.1016/S0044-8486(99)00244-6
Teletchea F, Fontaine P (2014) Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish Fish 15:181–195. https://doi.org/10.1111/faf.12006
Vlavonou RS (1996) Elevage expérimental de la perche Perca fluviatilis L. : développement larvaire et croissance. PhD thesis, University of Lorraine
Ward A, Webster M (2016) Sociality: the behaviour of group-living animals. Springer International Publishing, Cham
Wark AR, Greenwood AK, Taylor EM, Yoshida K, Peichel CL (2011) Heritable differences in schooling behavior among Threespine stickleback populations revealed by a novel assay. PLoS One 6:e18316. https://doi.org/10.1371/journal.pone.0018316
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, New-York
Wright D, Rimmer LB, Pritchard VL, Krause J, Butlin RK (2003) Inter and intra-population variation in shoaling and boldness in the zebrafish (Danio rerio). Naturwissenschaften 90:374–377. https://doi.org/10.1007/s00114-003-0443-2
Yang S, Yang K, Liu C, Sun J, Zhang F, Zhang X, Song Z (2015) To what extent is cannibalism genetically controlled in fish? A case study in juvenile hybrid catfish Silurus meridionalis–asotus and the progenitors. Aquaculture 437:208–214. https://doi.org/10.1016/J.AQUACULTURE.2014.12.005
Youngson NA, Whitelaw E (2008) Transgenerational epigenetic effects. Annu Rev Genomics Hum Genet 9:233–257. https://doi.org/10.1146/annurev.genom.9.081307.164445
Acknowledgements
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement no. 652831 (AQUAEXCEL2020). This output reflects only the author’s view, and the European Union cannot be held responsible for any use that may be made of the information contained therein. LT is supported by a grant from the French “Ministère de l’Enseignement Supérieur et de la Recherche”. TV is supported by the Ministry of Education, Youth and Sports of the Czech Republic—projects CENAKVA (No. CZ.1.05/2.1.00/01.0024), CENAKVA II (No. LO1205 under the NPU I programme), GAJU (No. 060/2016/Z) and NAZV (No. QJ 1510117). Authors acknowledge Laurent Espinat and Chloé Goulon (INRA, Thonon-Les-Bains, France) for their help with egg ribbon collection from Lake Geneva.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All along experimental procedures, individuals were handled as little as possible. All procedures were in accordance with the national and international guidelines for protection of animal welfare (Directive 2010/63/EU). This study was conducted with the approval of Animal Care Committee of Lorraine (CELMA no. 66) and the Ministry of Higher Education, Research, and Innovation (APAFIS13368-2018020511226118).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed by the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Toomey, L., Bláha, M., Mauduit, E. et al. When behavioural geographic differentiation matters: inter-populational comparison of aggressiveness and group structure in the European perch. Aquacult Int 27, 1177–1191 (2019). https://doi.org/10.1007/s10499-019-00343-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-019-00343-z