Abstract
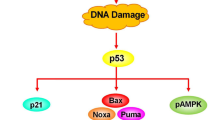
Esophageal squamous cell carcinoma (ESCC) is a common digestive cancer with high mortality rate due to late diagnosis and drug resistance. It is important to identify new molecular target and develop new anticancer strategy. ML323 is a novel USP1 inhibitor and exhibits anticancer activity against several cancers. Herein, we investigated whether ML323 has some cytotoxity effect on ESCC cells and explored the underlying mechanisms. Results revealed that ML323 impeded esophageal cancer cell viability and colony formation. Meanwhile, ML323 blocked cells at G0/G1 phase concomitant with the reduced protein level of c-Myc, cyclin D1, CDK4 and CDK6. ML323 treatment also triggered DNA damage and active p53. Then, ML323 induced apoptosis by p53-Noxa. Additionally, it stimulated protective autophagy. Co-treatment with CQ or BafA1, two classical autophagy inhibitors, enhanced the cytotoxity of ML323. These findings suggested that USP1 inhibitor (ML323) could be used as a viable anti-ESCC approach.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- ESCC:
-
Esophageal squamous cell carcinoma
- HCC:
-
Hepatocellular carcinoma
- MM:
-
Multiple myeloma
- EMT:
-
Epithelial-mesenchymal transition
- TNBC:
-
Triple negative breast cancer
- BafA1:
-
BafilomycinA1
- CQ:
-
Chloroquine
- DMSO:
-
Dimethyl sulfoxide
- PBS:
-
Phosphate-buffered saline
- MMP:
-
Mitochondrial membrane potential
- AO:
-
Acridine orange
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Arnold M, Ferlay J, van Berge Henegouwen MI, Soerjomataram I (2020) Global burden of oesophageal and gastric cancer by histology and subsite in 2018. Gut 69:1564–1571
Zhao Y, Xue C, Xie Z, Ouyang X, Li L (2020) Comprehensive analysis of ubiquitin-specific protease 1 reveals its importance in hepatocellular carcinoma. Cell Prolif 53:e12908
Li Y, Xu Y, Gao C, Sun Y, Zhou K, Wang P, Cheng J, Guo W, Ya C, Fan J, Yang X (2020) USP1 maintains the survival of liver circulating tumor cells by deubiquitinating and stabilizing TBLR1. Front Oncol 10:554809
Liao Y, Shao Z, Liu Y, Xia X, Deng Y, Yu C, Sun W, Kong W, He X, Liu F, Guo Z, Chen G, Tang D, Gan H, Liu J, Huang H (2021) USP1-dependent RPS16 protein stability drives growth and metastasis of human hepatocellular carcinoma cells. J Exp Clin Cancer Res 40:201
Lee JK, Chang N, Yoon Y, Yang H, Cho H, Kim E, Shin Y, Kang W, Oh YT, Mun GI, Joo KM, Nam DH, Lee J (2016) USP1 targeting impedes GBM growth by inhibiting stem cell maintenance and radioresistance. Neuro Oncol 18:37–47
Ma L, Lin K, Chang G, Chen Y, Yue C, Guo Q, Zhang S, Jia Z, Huang TT, Zhou A, Huang S (2019) Aberrant activation of beta-catenin signaling drives glioma tumorigenesis via USP1-mediated stabilization of EZH2. Cancer Res 79:72–85
Liu J, Zhu H, Zhong N, Jiang Z, Xu L, Deng Y, Jiang Z, Wang H, Wang J (2016) Gene silencing of USP1 by lentivirus effectively inhibits proliferation and invasion of human osteosarcoma cells. Int J Oncol 49:2549–2557
Ma A, Tang M, Zhang L, Wang B, Yang Z, Liu Y, Xu G, Wu L, Jing T, Xu X, Yang S, Liu Y (2019) USP1 inhibition destabilizes KPNA2 and suppresses breast cancer metastasis. Oncogene 38:2405–2419
Niu Z, Li X, Feng S, Huang Q, Zhuang T, Yan C, Qian H, Ding Y, Zhu J, Xu W (2020) The deubiquitinating enzyme USP1 modulates ERalpha and modulates breast cancer progression. J Cancer 11:6992–7000
Garcia-Santisteban I, Peters GJ, Giovannetti E, Rodriguez JA (2013) USP1 deubiquitinase: cellular functions, regulatory mechanisms and emerging potential as target in cancer therapy. Mol Cancer 12:91
Das DS, Das A, Ray A, Song Y, Samur MK, Munshi NC, Chauhan D, Anderson KC (2017) Blockade of deubiquitylating enzyme USP1 inhibits DNA repair and triggers apoptosis in multiple myeloma cells. Clin Cancer Res 23:4280–4289
Xu X, Li S, Cui X, Han K, Wang J, Hou X, Cui L, He S, Xiao J, Yang Y (2019) Inhibition of ubiquitin specific protease 1 sensitizes colorectal cancer cells to DNA-damaging chemotherapeutics. Front Oncol 9:1406
Cui SZ, Lei ZY, Guan TP, Fan LL, Li YQ, Geng XY, Fu DX, Jiang HW, Xu SH (2020) Targeting USP1-dependent KDM4A protein stability as a potential prostate cancer therapy. Cancer Sci 111:1567–1581
Gong H, Liu L, Cui L, Ma H, Shen L (2021) ALKBH5-mediated m6A-demethylation of USP1 regulated T-cell acute lymphoblastic leukemia cell glucocorticoid resistance by Aurora B. Mol Carcinog 60:644–657
Xu J, Li B, Song W, Cao L, Zhu C, Lin S (2021) Tumor suppressor functions of miRNA-375 in nasopharyngeal carcinoma through inhibition of ubiquitin-specific protease 1 expression. Int J Biochem Cell Biol 141:106092
Liao Y, Liu Y, Shao Z, Xia X, Deng Y, Cai J, Yao L, He J, Yu C, Hu T, Sun W, Liu F, Tang D, Liu J, Huang H (2021) A new role of GRP75-USP1-SIX1 protein complex in driving prostate cancer progression and castration resistance. Oncogene 40:4291–4306
Kuang X, Xiong J, Lu T, Wang W, Zhang Z, Wang J (2021) Inhibition of USP1 induces apoptosis via ID1/AKT pathway in B-cell acute lymphoblastic leukemia cells. Int J Med Sci 18:245–255
Ishikawa C, Mori N (2021) The antipsychotic drug pimozide is effective against human T-cell leukemia virus type 1-infected T cells. Eur J Pharmacol 908:174373
Han D, Wang L, Chen B, Zhao W, Liang Y, Li Y, Zhang H, Liu Y, Wang X, Chen T, Li C, Song X, Luo D, Li Z, Yang Q (2021) USP1-WDR48 deubiquitinase complex enhances TGF-beta induced epithelial-mesenchymal transition of TNBC cells via stabilizing TAK1. Cell Cycle 20:320–331
Williams SA, Maecker HL, French DM, Liu J, Gregg A, Silverstein LB, Cao TC, Carano RA, Dixit VM (2011) USP1 deubiquitinates ID proteins to preserve a mesenchymal stem cell program in osteosarcoma. Cell 146:918–930
Murai J, Yang K, Dejsuphong D, Hirota K, Takeda S, D’Andrea AD (2011) The USP1/UAF1 complex promotes double-strand break repair through homologous recombination. Mol Cell Biol 31:2462–2469
Cukras S, Lee E, Palumbo E, Benavidez P, Moldovan GL, Kee Y (2016) The USP1-UAF1 complex interacts with RAD51AP1 to promote homologous recombination repair. Cell Cycle 15:2636–2646
Kim JM, Parmar K, Huang M, Weinstock DM, Ruit CA, Kutok JL, D’Andrea AD (2009) Inactivation of murine Usp1 results in genomic instability and a Fanconi anemia phenotype. Dev Cell 16:314–320
Oestergaard VH, Langevin F, Kuiken HJ, Pace P, Niedzwiedz W, Simpson LJ, Ohzeki M, Takata M, Sale JE, Patel KJ (2007) Deubiquitination of FANCD2 is required for DNA crosslink repair. Mol Cell 28:798–809
Mistry H, Hsieh G, Buhrlage SJ, Huang M, Park E, Cuny GD, Galinsky I, Stone RM, Gray NS, D’Andrea AD, Parmar K (2013) Small-molecule inhibitors of USP1 target ID1 degradation in leukemic cells. Mol Cancer Ther 12:2651–2662
Chen J, Dexheimer TS, Ai Y, Liang Q, Villamil MA, Inglese J, Maloney DJ, Jadhav A, Simeonov A, Zhuang Z (2011) Selective and cell-active inhibitors of the USP1/ UAF1 deubiquitinase complex reverse cisplatin resistance in non-small cell lung cancer cells. Chem Biol 18:1390–1400
Liang Q, Dexheimer TS, Zhang P, Rosenthal AS, Villamil MA, You C, Zhang Q, Chen J, Ott CA, Sun H, Luci DK, Yuan B, Simeonov A, Jadhav A, Xiao H, Wang Y, Maloney DJ, Zhuang Z (2014) A selective USP1-UAF1 inhibitor links deubiquitination to DNA damage responses. Nat Chem Biol 10:298–304
Dexheimer TS, Rosenthal AS, Luci DK, Liang Q, Villamil MA, Chen J, Sun H, Kerns EH, Simeonov A, Jadhav A, Zhuang Z, Maloney DJ (2014) Synthesis and structure-activity relationship studies of N-benzyl-2-phenylpyrimidin-4-amine derivatives as potent USP1/UAF1 deubiquitinase inhibitors with anticancer activity against nonsmall cell lung cancer. J Med Chem 57:8099–8110
Dexheimer TS, Rosenthal AS, Liang Q, Chen J, Villamil MA, Kerns EH, Simeonov A, Jadhav A, Zhuang Z, Maloney DJ (2010) Discovery of ML323 as a Novel Inhibitor of the USP1/UAF1 Deubiquitinase Complex, Probe Reports from the NIH Molecular Libraries Program, Bethesda.
Kemp M (2016) Recent advances in the discovery of deubiquitinating enzyme inhibitors. Prog Med Chem 55:149–192
Rahme GJ, Zhang Z, Young AL, Cheng C, Bivona EJ, Fiering SN, Hitoshi Y, Israel MA (2016) PDGF engages an E2F-USP1 signaling pathway to support ID2-mediated survival of proneural glioma cells. Cancer Res 76:2964–2976
el Dakir H, Pickard A, Srivastava K, McCrudden CM, Gross SR, Lloyd S, Zhang SD, Margariti A, Morgan R, Rudland PS, El-Tanani M (2018) The anti-psychotic drug pimozide is a novel chemotherapeutic for breast cancer. Oncotarget 9:34889–34910
Locke MN, Rape M (2014) DNA damage: walking the edge. Nat Chem Biol 10:243–244
Park E, Kim H, Kim JM, Primack B, Vidal-Cardenas S, Xu Y, Price BD, Mills AA, D’Andrea AD (2013) FANCD2 activates transcription of TAp63 and suppresses tumorigenesis. Mol Cell 50:908–918
Logue SE, Elgendy M, Martin SJ (2009) Expression, purification and use of recombinant annexin V for the detection of apoptotic cells. Nat Protoc 4:1383–1395
Oancea M, Mazumder S, Crosby ME, Almasan A (2006) Apoptosis assays. Methods Mol Med 129:279–290
Tavakoli MB, Kheirollahi M, Kiani A, Kazemi M, Javanmard SH, Mohebat L (2015) Annexin V FITC conjugated as a radiation toxicity indicator in lymphocytes following radiation overexposure in radiotherapy programs. Adv Biomed Res 4:119
Wang L, Li M, Sha B, Hu X, Sun Y, Zhu M, Xu Y, Li P, Wang Y, Guo Y, Li J, Shi J, Li P, Hu T, Chen P (2021) Inhibition of deubiquitination by PR-619 induces apoptosis and autophagy via ubi-protein aggregation-activated ER stress in oesophageal squamous cell carcinoma. Cell Prolif 54:e12919
Hermeking H, Rago C, Schuhmacher M, Li Q, Barrett JF, Obaya AJ, O’Connell BC, Mateyak MK, Tam W, Kohlhuber F, Dang CV, Sedivy JM, Eick D, Vogelstein B, Kinzler KW (2000) Identification of CDK4 as a target of c-MYC. Proc Natl Acad Sci USA 97:2229–2234
Gu J, Wang X, Zhang L, Xiang J, Li J, Chen Z, Zhang Y, Chen J, Shen J (2021) Matrine suppresses cell growth of diffuse large B-cell lymphoma via inhibiting CaMKIIgamma/c-Myc/CDK6 signaling pathway. BMC Complement Med Ther 21:163
Mateyak MK, Obaya AJ, Sedivy JM (1999) c-Myc regulates cyclin D-Cdk4 and -Cdk6 activity but affects cell cycle progression at multiple independent points. Mol Cell Biol 19:4672–4683
Cook Sangar ML, Genovesi LA, Nakamoto MW, Davis MJ, Knobluagh SE, Ji P, Millar A, Wainwright BJ, Olson JM (2017) Inhibition of CDK4/6 by Palbociclib Significantly Extends Survival in Medulloblastoma Patient-Derived Xenograft Mouse Models. Clin Cancer Res 23:5802–5813
Burmakin M, Shi Y, Hedstrom E, Kogner P, Selivanova G (2013) Dual targeting of wild-type and mutant p53 by small molecule RITA results in the inhibition of N-Myc and key survival oncogenes and kills neuroblastoma cells in vivo and in vitro. Clin Cancer Res 19:5092–5103
O’Connor MJ (2015) Targeting the DNA damage response in cancer. Mol Cell 60:547–560
Raimondi M, Cesselli D, Di Loreto C, La Marra F, Schneider C, Demarchi F (2019) USP1 (ubiquitin specific peptidase 1) targets ULK1 and regulates its cellular compartmentalization and autophagy. Autophagy 15:613–630
Klionsky DJ, Abdel-Aziz AK, Abdelfatah S, Abdellatif M, Abdoli A, Abel S, Abeliovich H, Abildgaard MH, Abudu YP, Acevedo-Arozena A, Adamopoulos IE, Adeli K, Adolph TE, Adornetto A, Aflaki E, Agam G, Agarwal A, Aggarwal BB, Agnello M, Agostinis P, Agrewala JN, Agrotis A, Aguilar PV, Ahmad ST, Ahmed ZM, Ahumada-Castro U, Aits S, Aizawa S, Akkoc Y, Akoumianaki T, Akpinar HA, Al-Abd AM, Al-Akra L, Al-Gharaibeh A, Alaoui-Jamali MA, Alberti S, Alcocer-Gomez E, Alessandri C, Ali M, Alim Al-Bari MA, Aliwaini S, Alizadeh J, Almacellas E, Almasan A, Alonso A, Alonso GD, Altan-Bonnet N, Altieri DC, Alvarez EMC, Alves S et al (2021) Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition)(1). Autophagy 17:1–382
Lei LC, Yu VZ, Ko JMY, Ning L, Lung ML (2020) FANCD2 confers a malignant phenotype in esophageal squamous cell carcinoma by regulating cell cycle progression. Cancers (Basel) 12.
Bretones G, Delgado MD, Leon J (2015) Myc and cell cycle control. Biochim Biophys Acta 1849:506–516
Fry DW, Harvey PJ, Keller PR, Elliott WL, Meade M, Trachet E, Albassam M, Zheng X, Leopold WR, Pryer NK, Toogood PL (2004) Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Mol Cancer Ther 3:1427–1438
Yap CS, Peterson AL, Castellani G, Sedivy JM, Neretti N (2011) Kinetic profiling of the c-Myc transcriptome and bioinformatic analysis of repressed gene promoters. Cell Cycle 10:2184–2196
Hou Y, Liu R, Xia M, Sun C, Zhong B, Yu J, Ai N, Lu JJ, Ge W, Liu B, Chen X (2021) Nannocystin ax, an eEF1A inhibitor, induces G1 cell cycle arrest and caspase-independent apoptosis through cyclin D1 downregulation in colon cancer in vivo. Pharmacol Res 173:105870
Stacey DW (2003) Cyclin D1 serves as a cell cycle regulatory switch in actively proliferating cells. Curr Opin Cell Biol 15:158–163
Alao JP (2007) The regulation of cyclin D1 degradation: roles in cancer development and the potential for therapeutic invention. Mol Cancer 6:24
Morinaga N, Yahiro K, Matsuura G, Moss J, Noda M (2008) Subtilase cytotoxin, produced by Shiga-toxigenic Escherichia coli, transiently inhibits protein synthesis of Vero cells via degradation of BiP and induces cell cycle arrest at G1 by downregulation of cyclin D1. Cell Microbiol 10:921–929
Masamha CP, Benbrook DM (2009) Cyclin D1 degradation is sufficient to induce G1 cell cycle arrest despite constitutive expression of cyclin E2 in ovarian cancer cells. Cancer Res 69:6565–6572
Rezaei PF, Fouladdel S, Ghaffari SM, Amin G, Azizi E (2012) Induction of G1 cell cycle arrest and cyclin D1 down-regulation in response to pericarp extract of Baneh in human breast cancer T47D cells. Daru 20:101
Jansen-Durr P, Meichle A, Steiner P, Pagano M, Finke K, Botz J, Wessbecher J, Draetta G, Eilers M (1993) Differential modulation of cyclin gene expression by MYC. Proc Natl Acad Sci USA 90:3685–3689
Hanson KD, Shichiri M, Follansbee MR, Sedivy JM (1994) Effects of c-myc expression on cell cycle progression. Mol Cell Biol 14:5748–5755
Dominguez-Sola D, Gautier J (2014) MYC and the control of DNA replication. Cold Spring Harb Perspect Med 4:a014423
Albert MC, Brinkmann K, Kashkar H (2014) Noxa and cancer therapy: tuning up the mitochondrial death machinery in response to chemotherapy. Mol Cell Oncol 1:e29906
Perez-Galan P, Roue G, Villamor N, Montserrat E, Campo E, Colomer D (2006) The proteasome inhibitor bortezomib induces apoptosis in mantle-cell lymphoma through generation of ROS and Noxa activation independent of p53 status. Blood 107:257–264
Guikema JE, Amiot M, Eldering E (2017) Exploiting the pro-apoptotic function of NOXA as a therapeutic modality in cancer. Expert Opin Ther Targets 21:767–779
Zou J, Li S, Chen Z, Lu Z, Gao J, Zou J, Lin X, Li Y, Zhang C, Shen L (2018) A novel oral camptothecin analog, gimatecan, exhibits superior antitumor efficacy than irinotecan toward esophageal squamous cell carcinoma in vitro and in vivo. Cell Death Dis 9:661
Ji J, Wu K, Wu M, Zhan Q (2010) p53 functional activation is independent of its genotype in five esophageal squamous cell carcinoma cell lines. Front Med China 4:412–418
Celeste A, Petersen S, Romanienko PJ, Fernandez-Capetillo O, Chen HT, Sedelnikova OA, Reina-San-Martin B, Coppola V, Meffre E, Difilippantonio MJ, Redon C, Pilch DR, Olaru A, Eckhaus M, Camerini-Otero RD, Tessarollo L, Livak F, Manova K, Bonner WM, Nussenzweig MC, Nussenzweig A (2002) Genomic instability in mice lacking histone H2AX. Science 296:922–927
Carrassa L, Damia G (2017) DNA damage response inhibitors: mechanisms and potential applications in cancer therapy. Cancer Treat Rev 60:139–151
Smith J, Tho LM, Xu N, Gillespie DA (2010) The ATM-Chk2 and ATR-Chk1 pathways in DNA damage signaling and cancer. Adv Cancer Res 108:73–112
Guervilly JH, Renaud E, Takata M, Rosselli F (2011) USP1 deubiquitinase maintains phosphorylated CHK1 by limiting its DDB1-dependent degradation. Hum Mol Genet 20:2171–2181
Peng H, Yang F, Hu Q, Sun J, Peng C, Zhao Y, Huang C (2020) The ubiquitin-specific protease USP8 directly deubiquitinates SQSTM1/p62 to suppress its autophagic activity. Autophagy 16:698–708
Acknowledgements
This work was supported by the National Natural Science Foundation Grant of China (Grant No. 81672421), Program for Innovation Research Team (in Science and Technology) in University of Henan Province (Grant No. 20IRTSTHN026) and Open Project Fund of Henan Key Laboratory of Precision Clinical Pharmacy (Grant No. HKLPCP-2020-06).
Author information
Authors and Affiliations
Contributions
Conceptualization, TH and PC; Methodology, BS, YS, SZ, ML, WH, YZ, JY, ZL, JT, and PD; Writing-Original draft preparation, BS and YS; Writing- Reviewing and Editing, JS, TH and PC; Supervision, TH and PC; Funding acquisition, PC and PL. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, Y., Sha, B., Huang, W. et al. ML323, a USP1 inhibitor triggers cell cycle arrest, apoptosis and autophagy in esophageal squamous cell carcinoma cells. Apoptosis 27, 545–560 (2022). https://doi.org/10.1007/s10495-022-01736-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-022-01736-x