Abstract
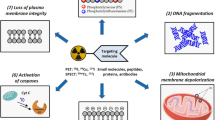
The synthetic bis(zinc(II)-dipicolylamine) (DPAZn2) coordination complexes are known to have a high specific and selective affinity to target the exposed phosphatidylserine (PS) on the surface of dead and dying cells. An 18F-labeled DPAZn2 complex (4-18F-Fluoro-benzoyl-bis(zinc(II)-dipicolylamine), 18F-FB-DPAZn2) as positron emission tomography (PET) tracer was developed and evaluated for in vivo imaging of tumor treated with a chemical agent. The in vitro cell stain studies revealed that fluorescent DPAZn2 complexes (Dansyl-DPAZn2) stained the same cells (apoptotic and necrotic cells) as fluorescein isothiocyanate (FITC) labeled Annexin V (FITC-Annexin V). The radiosynthesis of 18F-FB-DPAZn2 was achieved through the amidation the precursor bis(2,2′-dipicolylamine) derivative (DPA2) with the prosthetic group N-succinimidyl-4-[18F]-fluorobenzoate (18F-SFB) and chelation with zinc nitrate. In the biodistribution study, the fast clearance of 18F-FB-DPAZn2 from blood and kidney was observed and high uptake in liver and intestine within 90 min postinjection was also found. For the PET imaging, significantly higher tumor uptake of 18F-FB-DPAZn2 was observed in the adriamycin (ADM)-treated Hepa1-6 hepatocellular carcinoma-bearing mice than that in the untreated tumor-model mice, while a slightly decreased tumor uptake of 18F-FDG was found in the ADM-treated tumor-bearing mice. The results indicate that 18F-FB-DPAZn2 has the similar capability of apoptosis detection as FITC-Annexin V and seems to be a potential PET tracer for noninvasive evaluation and monitoring of anti-tumor chemotherapy. The high uptake of 18F-FB-DPAZn2 in the abdomen needs to optimize the structure for improving its pharmacokinetics characteristics in the future work.






Similar content being viewed by others
References
Brown JM, Attardi LD (2005) The role of apoptosis in cancer development and treatment response. Nat Rev Cancer 5:231–237
Corsten MF, Hofstra L, Narula J et al (2006) Counting heads in the war against cancer: defining the role of Annexin A5 imaging in cancer treatment and surveillance. Cancer Res 66:1255–1260
Lahorte CM, Vanderheyden JL, Steinmetz N et al (2004) Apoptosis-detecting radioligands: current state of the art and future perspectives. Eur J Nucl Med 31:887–919
Blankenberg FG (2008) In vivo detection of apoptosis. J Nucl Med 49:81S–95S
Grimberg H, Levin G, Shirvan A et al (2009) Monitoring of tumor response to chemotherapy in vivo by a novel small-molecule detector of apoptosis. Apoptosis 14:257–267
Reshef A, Shirvan A, Akselrod-Ballin A et al (2010) Small-molecule biomarkers for clinical PET imaging of apoptosis. J Nucl Med 51:837–840
Niu G, Chen X (2010) Apoptosis imaging: beyond annexin V. J Nucl Med 51:1659–1662
Hanshaw RG, Lakshmi C, Lambert TN et al (2005) Fluorescent detection of apoptotic cells by using zinc coordination complexes with a selective affinity for membrane surfaces enriched with phosphatidylserine. ChemBioChem 6:2214–2220
Smith BA, Akers WJ, Leevy WM et al (2010) Optical imaging of mammary and prostate tumors in living animals using a synthetic near infrared zinc(II)-dipicolylamine probe for anionic cell surfaces. J Am Chem Soc 132:67–69
Leevy WM, Johnson JR, Lakshmi C et al (2006) Selective recognition of bacterial membranes by zinc(II)-coordination complexes. Chem Commun 1595–1597
Leevy WM, Gammon ST, Jiang H et al (2006) Optical imaging of bacterial infection in living mice using a fluorescent near-infrared molecular probe. J Am Chem Soc 128:16476–16477
Leevy WM, Gammon ST, Johnson JR et al (2008) Noninvasive optical imaging of Staphylococcus aureus bacterial infection in living mice using a bis-dipicolylamine-zinc(II) affinity group conjugated to a near-infrared fluorophore. Bioconjugate Chem 19:686–692
White AG, Fu N, Leevy WM et al (2010) Optical imaging of bacterial infection in living mice using deep-red fluorescent squaraine rotaxane probes. Bioconjugate Chem 21:1297–1304
Smith BA, Gammon ST, Xiao S et al (2011) In vivo optical imaging of acute cell death using a near-infrared fluorescent zinc-dipicolylamine probe. Mol Pharmaceutics 8:583–590
Chen K, Yap L, Park R et al (2011) Evaluation of 64Cu-labeled dipicolylamine (DPA) as a small-molecule PET probe for in vivo imaging of phosphatidylserine exposure. J Nucl Med 52(Suppl 1):1502. http://jnumedmtg.snmjournals.org/cgi/content/meeting abstract/52/1_MeetingAbstracts/1502
Wyffels L, Gray BD, Barber C et al (2011) Synthesis and preliminary evaluation of radiolabeled bis(zinc(II)-dipicolylamine) coordination complexes as cell death imaging agents. Bioorg Med Chem 19:3425–3433
Liu X, Cheng D, Gray BD et al (2012) Radiolabeled Zn-DPA as a potential infection imaging agent. Nucl Med Biol 39(5):709–714
Kimura E, Aoki S, Kikuta E et al (2003) A macrocyclic zinc(II) fluorophore as a detector of apoptosis. PNAS 100:3731–3736
Oltmanns D, Zitzmann-Kolbe S, Mueller A et al (2011) Zn(II)-Bis(Cyclen) complexes and the imaging of apoptosis/necrosis. Bioconjugate Chem 22:2611–2624
Lakshmi C, Hanshaw RG, Smith BD (2004) Fluorophore-linked zinc(II)dipicolylamine coordination complexes as sensors for phosphatidylserine-containing membranes. Tetrahedron 60:11307–11315
Guo XY, Wang HL, Jin YF et al (2011) Synthesis and radiolabelling of [18F]FEDPA as an imaging agent for apoptosis. J Nucl Radiochem 33(4):245–251. http://www.jnrc.org.cn/qikan/public/tjdjl.asp?wenjianming=1104-33-09.pdf
Tang G, Zeng W, Yu M et al (2008) Facile synthesis of N-succinimidyl 4-[18F]fluorobenzoate ([18F]SFB) for protein labeling. J Label Compd Radiopharm 51:68–71
Brindle KM (2008) New approaches for imaging tumour responses to treatment. Nat Rev Cancer 8:94–107
Hanshaw RG, Smith BD (2005) New reagents for phosphatidylserine recognition and detection of apoptosis. Bioorg Med Chem 13:5035–5042
Koulov AV, Stucker KA, Lakshmi C et al (2003) Detection of apoptotic cells using a synthetic fluorescent sensor for membrane surfaces that contain phosphatidylserine. Cell Death Differ 10(12):1357–1359
Fahrni CJ, O’Halloran TV (1999) Aqueous coordination chemistry of quinoline-based fluorescence probes for the biological chemistry of zinc. J Am Chem Soc 121:11448–11458
Nasir MS, Fahrni CJ, Suhy DA et al (1999) The chemical cell biology of zinc: structure and intracellular fluorescence of a zinc-quinolinesulfonamide complex. J Biol Inorg Chem 4:775–783
Murakami Y, Takamatsu HJ, Taki M et al (2004) 18F-labelled annexin V: a PET tracer for apoptosis imaging. Eur J Nucl Med 31:469–474
Yagle KJ, Eary JF, Tait JF et al (2005) Evaluation of 18F-annexin V as a PET imaging agent in an animal model of apoptosis. J Nucl Med 6:658–666
Grierson JR, Yagle KJ, Eary JF et al (2004) Production of [F-18]fluoroannexin for imaging apoptosis with PET. Bioconjugate Chem 15:373–379
Acknowledgments
The authors would like to thank Dr. Zhong Pei for his technical support and Dr. Xiangsong Zhang for his useful assistance. This work was supported by the National Natural Science Foundation (No. 81101076, No. 30970856, No. 81201116), Postdoctoral Science Foundation of China (20110490964), Science and Technology Planning Project of Guangdong Province, China (2010B031600054), Science and Technology Planning Project of Guangzhou (2011J5200025) and The Project Sponsored by the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry (No. [2010]609).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, H., Tang, X., Tang, G. et al. Noninvasive positron emission tomography imaging of cell death using a novel small-molecule probe, 18F labeled bis(zinc(II)-dipicolylamine) complex. Apoptosis 18, 1017–1027 (2013). https://doi.org/10.1007/s10495-013-0852-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-013-0852-4