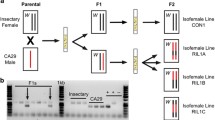
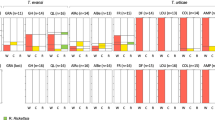
Abstract
Maternally transmitted symbionts such as Cardinium and Wolbachia are widespread in arthropods. Both Cardinium and Wolbachia can cause cytoplasmic incompatibility, a reproductive phenotype that interferes with the development of uninfected eggs that are fertilized by infected sperm. In haplodiploid hosts, these symbionts can also distort sex allocation to facilitate their spread through host populations. Without other fitness effects, symbionts that induce strong reproductive phenotypes tend to spread to high and stable infection frequencies, whereas variants that induce weak reproductive phenotypes are typically associated with intermediate and variable frequencies. To study the spread of Cardinium in a haplodiploid host, we sampled Iranian populations of the economically important spider mite Panonychus ulmi in apple orchards. Within several field populations, we also studied the Wolbachia infection frequencies. All P. ulmi field populations carried a Cardinium infection and exhibited high infection frequencies. In contrast, Wolbachia frequency ranged between ca. 10% and ca. 70% and was only found in co-infected mites. To test whether Cardinium induce reproductive phenotypes in P. ulmi, a Cardinium-cured derived line was generated by antibiotic treatment from a co-infected field population. Genetic crosses indicated that Cardinium do not induce demonstrable levels of cytoplasmic incompatibility and sex allocation distortion in co-infected P. ulmi. However, Cardinium infection was associated with a longer developmental time and reduced total fecundity for co-infected females. We hypothesize that Cardinium spread through P. ulmi populations via uncharacterized fitness effects and that co-infection with Wolbachia might impact these drive mechanisms.



Similar content being viewed by others
Data availability
Count and read data are uploaded as Supplementary Material.
References
Badieinia F, Khajehali J, Nauen R et al (2020) Metabolic mechanisms of resistance to spirodiclofen and spiromesifen in Iranian populations of Panonychus ulmi. Crop Prot 134:105166. https://doi.org/10.1016/j.cropro.2020.105166
Bajda S, Dermauw W, Greenhalgh R et al (2015) Transcriptome profiling of a spirodiclofen susceptible and resistant strain of the European red mite Panonychus ulmi using strand-specific RNA-seq. BMC Genomics. https://doi.org/10.1186/s12864-015-2157-1
Barton NH, Turelli M (2011) Spatial waves of advance with Bistable dynamics: cytoplasmic and genetic analogues of Allee effects. Am Nat 178:E48–E75. https://doi.org/10.1086/661246
Bird FT (1967) A virus disease of the European red mite Panonychus ulmi (Koch). Can J Microbiol 13:1131–1131. https://doi.org/10.1139/m67-156
Breeuwer JAJ (1997) Wolbachia and cytoplasmic incompatibility in the spider mites Tetranychus urticae and T. turkestani. Heredity 79:41–47. https://doi.org/10.1038/hdy.1997.121
Cooper BS, Ginsberg PS, Turelli M, Matute DR (2017) Wolbachia in the Drosophila yakuba complex: pervasive frequency variation and weak cytoplasmic incompatibility, but no apparent effect on reproductive isolation. Genetics 205:333–351. https://doi.org/10.1534/genetics.116.196238
Duron O, Bouchon D, Boutin S et al (2008) The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol 6:27. https://doi.org/10.1186/1741-7007-6-27
Egas M, Vala F, Breeuwer JAJ (2002) On the evolution of cytoplasmic incompatibility in haplodiploid species. Evolution 56:1101–1109. https://doi.org/10.1111/j.0014-3820.2002.tb01424.x
Engelstädter J, Hurst GDD (2009) The ecology and evolution of microbes that manipulate host reproduction. Annu Rev Ecol Evol Syst 40:127–149. https://doi.org/10.1146/annurev.ecolsys.110308.120206
Gotoh T, Noda H, Hong X-Y (2003) Wolbachia distribution and cytoplasmic incompatibility based on a survey of 42 spider mite species (Acari: Tetranychidae) in Japan. Heredity 91:208–216. https://doi.org/10.1038/sj.hdy.6800329
Gotoh T, Noda H, Ito S (2007) Cardinium symbionts cause cytoplasmic incompatibility in spider mites. Heredity 98:13–20. https://doi.org/10.1038/sj.hdy.6800881
Hague MTJ, Mavengere H, Matute DR, Cooper BS (2020) Environmental and genetic contributions to imperfect wMel-Like Wolbachia transmission and frequency variation. Genetics 215:1117–1132. https://doi.org/10.1534/genetics.120.303330
Hague MTJ, Shropshire JD, Caldwell CN et al (2022) Temperature effects on cellular host-microbe interactions explain continent-wide endosymbiont prevalence. Curr Biol 32:878-888.e8. https://doi.org/10.1016/j.cub.2021.11.065
Hamm CA, Begun DJ, Vo A et al (2014) Wolbachia do not live by reproductive manipulation alone: infection polymorphism in Drosophila suzukii and D. subpulchrella. Mol Ecol 23:4871–4885. https://doi.org/10.1111/mec.12901
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS (2018) UFBoot2: improving the ultrafast bootstrap approximation. Mol Biol Evol 35(2):518–522. https://doi.org/10.1093/molbev/msx281
Hoffmann AA, Turelli M, Harshman LG (1990) Factors affecting the distribution of cytoplasmic incompatibility in Drosophila Simulans. Genetics 126:933–948
Hunter MS, Perlman SJ, Kelly SE (2003) A bacterial symbiont in the Bacteroidetes induces cytoplasmic incompatibility in the parasitoid wasp Encarsia pergandiella. Proc R Soc Lond B 270:2185–2190. https://doi.org/10.1098/rspb.2003.2475
Itoh H, Tago K, Hayatsu M, Kikuchi Y (2018) Detoxifying symbiosis: microbe-mediated detoxification of phytotoxins and pesticides in insects. Nat Prod Rep 35:434–454. https://doi.org/10.1039/C7NP00051K
Jeppson LR, Keifer HH, Baker EW (1975) Mites injurious to economic plants. University of California Press, Berkeley
Kalyaanamoorthy S, Minh BQ, Wong TKF, von Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nature Methods 14(6):587–589. https://doi.org/10.1038/nmeth.4285
Katlav A, Cook JM, Riegler M (2022) Common endosymbionts affect host fitness and sex allocation via egg size provisioning. Proc R Soc B 289:20212582. https://doi.org/10.1098/rspb.2021.2582
Katoh K, Toh H (2010) Parallelization of the MAFFT multiple sequence alignment program. Bioinformatics 26:1899–1900. https://doi.org/10.1093/bioinformatics/btq224
Kaur R, Shropshire JD, Cross KL et al (2021) Living in the endosymbiotic world of Wolbachia: a centennial review. Cell Host Microbe 29:879–893. https://doi.org/10.1016/j.chom.2021.03.006
Li C-X, Shi M, Tian J-H et al (2015) Unprecedented genomic diversity of RNA viruses in arthropods reveals the ancestry of negative-sense RNA viruses. Elife 4:e05378. https://doi.org/10.7554/eLife.05378
Martinez J, Longdon B, Bauer S et al (2014) Symbionts commonly provide broad spectrum resistance to viruses in insects: a comparative analysis of Wolbachia strains. PLoS Pathog 10:e1004369. https://doi.org/10.1371/journal.ppat.1004369
Martinez J, Klasson L, Welch JJ, Jiggins FM (2021) Life and death of selfish genes: comparative genomics reveals the dynamic evolution of cytoplasmic incompatibility. Mol Biol Evol 38:2–15. https://doi.org/10.1093/molbev/msaa209
Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, von Haeseler A, Lanfear R (2020) IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37(5):1530–1534. https://doi.org/10.1093/molbev/msaa015
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucl Acids Res 8:4321–4326. https://doi.org/10.1093/nar/8.19.4321
Nakamura Y, Kawai S, Yukuhiro F et al (2009) Prevalence of Cardinium bacteria in planthoppers and spider mites and taxonomic revision of “Candidatus Cardinium hertigii” based on detection of a new Cardinium group from biting midges. Appl Environ Microbiol 75:6757–6763. https://doi.org/10.1128/AEM.01583-09
Nguyen DT, Morrow JL, Spooner-Hart RN, Riegler M (2017) Independent cytoplasmic incompatibility induced by Cardinium and Wolbachia maintains endosymbiont coinfections in haplodiploid thrips populations. Evolution 71:995–1008. https://doi.org/10.1111/evo.13197
Perlmutter JI, Bordenstein SR (2020) Microorganisms in the reproductive tissues of arthropods. Nat Rev Microbiol 18:97–111. https://doi.org/10.1038/s41579-019-0309-z
Perrot-Minnot M-J, Cheval B, Migeon A, Navajas M (2002) Contrasting effects of Wolbachia on cytoplasmic incompatibility and fecundity in the haplodiploid mite Tetranychus urticae: contrasting effects of Wolbachia in a haplodiploid mite. J Evol Biol 15:808–817. https://doi.org/10.1046/j.1420-9101.2002.00446.x
Pina T, Sabater-Muñoz B, Cabedo-López M et al (2020) Molecular characterization of Cardinium, Rickettsia, Spiroplasma and Wolbachia in mite species from citrus orchards. Exp Appl Acarol 81:335–355. https://doi.org/10.1007/s10493-020-00508-z
Pollmann M, Moore LD, Krimmer E et al (2022) Highly transmissible cytoplasmic incompatibility by the extracellular insect symbiont Spiroplasma. iScience 25:104335. https://doi.org/10.1016/j.isci.2022.104335
Rameshgar F, Khajehali J, Nauen R et al (2019) Characterization of abamectin resistance in Iranian populations of European red mite, Panonychus ulmi Koch (Acari: Tetranychidae). Crop Prot 125:104903. https://doi.org/10.1016/j.cropro.2019.104903
Ros VID, Breeuwer JAJ (2009) The effects of, and interactions between, Cardinium and Wolbachia in the doubly infected spider mite Bryobia sarothamni. Heredity 102:413–422
Ros VID, Fleming VM, Feil EJ, Breeuwer JAJ (2012) Diversity and recombination in Wolbachia and Cardinium from Bryobia spider mites. BMC Microbiol 12:S13
Rosenwald LC, Sitvarin MI, White JA (2020) Endosymbiotic Rickettsiella causes cytoplasmic incompatibility in a spider host. Proc R Soc B 287:20201107. https://doi.org/10.1098/rspb.2020.1107
Shropshire JD, Leigh B, Bordenstein SR (2020) Symbiont-mediated cytoplasmic incompatibility: what have we learned in 50 years. Elife 9:e61989. https://doi.org/10.7554/eLife.61989
Smith AH, Łukasik P, O’Connor MP et al (2015) Patterns, causes and consequences of defensive microbiome dynamics across multiple scales. Mol Ecol 24:1135–1149. https://doi.org/10.1111/mec.13095
Stouthamer CM, Kelly SE, Mann E et al (2019) Development of a multi-locus sequence typing system helps reveal the evolution of Cardinium hertigii, a reproductive manipulator symbiont of insects. BMC Microbiol 19:266. https://doi.org/10.1186/s12866-019-1638-9
Takano S, Tuda M, Takasu K et al (2017) Unique clade of alphaproteobacterial endosymbionts induces complete cytoplasmic incompatibility in the coconut beetle. Proc Natl Acad Sci USA 114:6110–6115. https://doi.org/10.1073/pnas.1618094114
Takano S, Gotoh Y, Hayashi T (2021) “Candidatus Mesenet longicola”: novel endosymbionts of Brontispa longissima that induce cytoplasmic incompatibility. Microb Ecol 82:512–522. https://doi.org/10.1007/s00248-021-01686-y
Teixeira L, Ferreira Á, Ashburner M (2008) The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. PLoS Biol 6:e1000002. https://doi.org/10.1371/journal.pbio.1000002
Vacante V (2016) The handbook of mites of economic plants - Identification, bio-ecology and control. CABI, Wallingford
Vala F, Van Opijnen T, Breeuwer JAJ, Sabelis MW (2003) Genetic conflicts over sex ratio: mite-endosymbiont interactions. Am Nat 161:254–266. https://doi.org/10.1086/345856
Vautrin E, Vavre F (2009) Interactions between vertically transmitted symbionts: cooperation or conflict? Trends Microbiol 17:95–99. https://doi.org/10.1016/j.tim.2008.12.002
Vavre F, Fleury F, Varaldi J et al (2000) Evidence for female mortality in Wolbachia -mediated cytoplasmic incompatibility in haplodiploid insects: epidemiologic and evolutionary consequences. Evolution 54:191–200. https://doi.org/10.1111/j.0014-3820.2000.tb00019.x
Vavre F, Dedeine F, Quillon M et al (2001) Within-species diversity of Wolbachia-induced cytoplasmic incompatibility in haplodiploid insects. Evolution 55:1710–1714. https://doi.org/10.1111/j.0014-3820.2001.tb00691.x
Wang Y-B, Ren F-R, Yao Y-L et al (2020) Intracellular symbionts drive sex ratio in the whitefly by facilitating fertilization and provisioning of B vitamins. ISME J 14:2923–2935. https://doi.org/10.1038/s41396-020-0717-0
Watts T, Haselkorn TS, Moran NA, Markow TA (2009) Variable incidence of Spiroplasma infections in natural populations of Drosophila Species. PLoS ONE 4:e5703. https://doi.org/10.1371/journal.pone.0005703
Weeks AR, Stouthamer R (2004) Increased fecundity associated with infection by a cytophaga-like intracellular bacterium in the predatory mite, Metaseiulus occidentalis. Proc Royal Soc B 271:S193–S195. https://doi.org/10.1098/rsbl.2003.0137
Weeks AR, Velten R, Stouthamer R (2003) Incidence of a new sex–ratio–distorting endosymbiotic bacterium among arthropods. Proc R Soc Lond B 270:1857–1865. https://doi.org/10.1098/rspb.2003.2425
Weeks AR, Turelli M, Harcombe WR et al (2007) From parasite to mutualist: rapid evolution of Wolbachia in natural populations of Drosophila. PLoS Biol 5:e114. https://doi.org/10.1371/journal.pbio.0050114
White JA, Kelly SE, Perlman SJ, Hunter MS (2009) Cytoplasmic incompatibility in the parasitic wasp Encarsia inaron: disentangling the roles of Cardinium and Wolbachia symbionts. Heredity 102:483–489. https://doi.org/10.1038/hdy.2009.5
WmL P (1970) Occurrence and transmission of a virus disease of the EUROPEAN red mite, Panonychus ulmi. Can Entomol 102:305–321. https://doi.org/10.4039/Ent102305-3
Wybouw N, Mortier F, Bonte D (2022) Interacting host modifier systems control Wolbachia-induced cytoplasmic incompatibility in a haplodiploid mite. Evol Lett 6:255–265. https://doi.org/10.1002/evl3.282
Wybouw N, Van Reempts E, Zarka J et al (2023) Egg provisioning explains the penetrance of symbiont-mediated sex allocation distortion in haplodiploids. Heredity. https://doi.org/10.1038/s41437-023-00638-1
Yaghoobi R, Khajehali J, Alavijeh ES et al (2021) Fenpyroximate resistance in Iranian populations of the European red mite Panonychus ulmi (Acari: Tetranychidae). Exp Appl Acarol 83:69–79. https://doi.org/10.1007/s10493-020-00569-0
Zhang Y-K, Chen Y-T, Yang K et al (2016) Screening of spider mites (Acari: Tetranychidae) for reproductive endosymbionts reveals links between co-infection and evolutionary history. Sci Rep 6:27900. https://doi.org/10.1038/srep27900
Zhu L-Y, Zhang K-J, Zhang Y-K et al (2012) Wolbachia strengthens Cardinium-induced cytoplasmic incompatibility in the spider mite Tetranychus piercei McGregor. Curr Microbiol 65:516–523. https://doi.org/10.1007/s00284-012-0190-8
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
NHG, NP, and JK performed the experiments. NHG, NP, and NW analyzed the data. NW wrote the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haghshenas-Gorgabi, N., Poorjavd, N., Khajehali, J. et al. Cardinium symbionts are pervasive in Iranian populations of the spider mite Panonychus ulmi despite inducing an infection cost and no demonstrable reproductive phenotypes when Wolbachia is a symbiotic partner. Exp Appl Acarol 91, 369–380 (2023). https://doi.org/10.1007/s10493-023-00840-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-023-00840-0