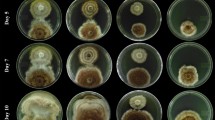
Abstract
Leaf-cutter ants cultivate and feed on the mutualistic fungus, Leucoagaricus gongylophorus, which is threatened by parasitic fungi of the genus Escovopsis. The mechanism of Escovopsis parasitism is poorly understood. Here, we assessed the nature of the antagonism of different Escovopsis species against its host. We also evaluated the potential antagonism of Escovopsioides, a recently described fungal genus from the attine ant environment whose role in the colonies of these insects is unknown. We performed dual-culture assays to assess the interactions between L. gongylophorus and both fungi. We also evaluated the antifungal activity of compounds secreted by the latter on L. gongylophorus growth using crude extracts of Escovopsis spp. and Escovopsioides nivea obtained either in (1) absence or (2) presence of the mutualistic fungus. The physical interaction between these fungi and the mutualistic fungus was examined under scanning electron microscopy (SEM). Escovopsis spp. and E. nivea negatively affected the growth of L. gongylophorus, which was also significantly inhibited by both types of crude extract. These results indicate that Escovopsis spp. and E. nivea produce antifungal metabolites against the mutualistic fungus. SEM showed that Escovopsis spp. and E. nivea maintained physical contact with the mutualistic fungus, though no specialised structures related to mycoparasitism were observed. These results showed that Escovopsis is a destructive mycoparasite that needs physical contact for the death of the mutualistic fungus to occur. Also, our findings suggest that E. nivea is an antagonist of the ant fungal cultivar.



Similar content being viewed by others
References
Augustin JO, Groenewald JZ, Nascimento RJ, Mizubuti ESG, Barreto RW, Elliot SL, Evans HC (2013) Yet more ‘‘weeds’’ in the garden: fungal novelties from nests of leaf-cutting ants. PLoS One 8:e82265. doi:10.1371/journal.pone.0082265
Barnett HL (1964) Mycoparasitism. Mycologia 56:1–19
Birnbaum SL, Gerardo NL (2016) Patterns of specificity of the pathogen Escovopsis across the fungus-growing ant symbiosis. Am Nat 188:52–65. doi:10.1086/686911
Carreiro SC, Pagnocca FC, Bueno OC, Bacci M Jr, Hebling MJA, Silva OA (1997) Yeast associated with nests of the leaf-cutting ant Atta sexdens rubropilosa Forel, 1908. Anton Leeuw Int J G 71:243–248. doi:10.1023/A:1000182108648
Currie CR (2001) A community of ants, fungi, and bacteria: a multilateral approach to studying symbiosis. Annu Rev Microbiol 55:357–380. doi:10.1146/annurev.micro.55.1.357
Currie CR, Stuart AE (2001) Weeding and grooming of pathogens in agriculture by ants. Proc R Soc Lond B 268:1033–1039. doi:10.1098/rspb.2001.1605
Currie CR, Mueller UG, Malloch D (1999) The agricultural pathology of ant fungus gardens. Proc Natl Acad Sci USA 96:7998–8002. doi:10.1073/pnas.96.14.7998
Currie CR, Wong B, Stuart AE, Schultz TR, Rehner SA, Muller UG, Sung GH, Spatafora JW, Straus NA (2003) Ancient tripartite coevolution in the attine ant-microbe symbiosis. Science 299:386–388. doi:10.1126/science.1078155
Elizondo-Wallace DE, Vargas-Ansesio JG, Pinto-Tomás AA (2014) Correlation between virulence and genetic structure of Escovopsis strains from leaf-cutting ant colonies in Costa Rica. Microbiology 160:1727–1736. doi:10.1099/mic.0.073593-0
Folgarait P, Gorosito N, Poulsen M, Currie CR (2011a) Preliminary in vitro insights into the use of natural fungal patoghens of leaf-cutting as biocontrol agents. Curr Microbiol 63:250–258. doi:10.1007/s00284-011-9944-y
Folgarait PJ, Marfetán JA, Cafaro MJ (2011b) Growth and conidiation response of Escovopsis weberi (Ascomycota:Hypocreales) against the fungal cultivar of Acromyrmex lundii (Hymenoptera:Formicidae). Environ Entomol 40:342–349. doi:10.1603/EN10111
Gerardo NM, Mueller UG, Price SL, Currie CR (2004) Exploiting a mutualism: parasite specialization on cultivars within the fungus-growing ant symbiosis. Proc R Soc Lond B 1550:1791–1798. doi:10.1098/rspb.2004.2792
Gerardo NM, Jacobs SR, Currie CR, Mueller UG (2006) Ancient host-pathogen associations maintained by specificity of chemotaxis and antibiosis. PLoS Biol 4:e235. doi:10.1371/journal.pbio.0040235
Jeffries P (1995) Biology and ecology of mycoparasitism. Can J Bot 73:1284–1290
Man TJB, Stajich JE, Kubicek CP, Teiling C, Chenthamara K, Atanasova L, Druzhinina IS, Levenkova N, Birnbaum SSL, Barribeau SM, Bozick BA, Suen G, Currie CR, Gerardo NM (2016) Small genome of the fungus Escovopsis weberi, a specialized disease agent of ant agriculture. Proc Natl Acad Sci USA 113:3567–3572. doi:10.1073/pnas.1518501113
Marfetán JA, Romero AI, Folgarait PJ (2015) Pathogenic interaction between Escovopsis weberi and Leucoagaricus sp.: mechanisms involved and virulence levels. Fungal Ecol 17:52–61. doi:10.1016/j.funeco.2015.04.002
Meirelles LA, Solomon SE, Jr Bacci, Wright AM, Mueller UG, Rodrigues A (2015) Shared Escovopsis parasites between leaf-cutting and non-leaf-cutting ants in the higher attine fungus-growing ant symbiosis. R Soc Open Sci 2:150257. doi:10.1098/rsos.150257
Möller A (1893) Die Pilzgärten einiger südamerikanischer Ameisen. Bot Mitt Tropen 6:1–127
Pagnocca FC, Silva OA, Hebling-Beraldo MJ, Bueno OC, Fernandes JB, Vieira PC (1990) Toxicity of sesame extracts to the symbiotic fungus of leaf-cutting ants. Bull Entomol Res 80:349–352
Pagnocca FC, Masiulionis VE, Rodrigues A (2012) Specialized fungal parasites and opportunistic fungi in gardens of attine ants. Psyche 12:1–9. doi:10.1155/2012/905109
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Reis BMS, Silva A, Alvarez MR, Oliveira TB, Rodrigues A (2015) Fungal communities in gardens of the leafcutter ant Atta cephalotes in forest and cabruca agrosystems of southern Bahia State (Brazil). Fungal Biol 119:1170–1178. doi:10.1016/j.funbio.2015.09.001
Reynolds HT, Currie CR (2004) Pathogenicity of Escovopsis weberi: the parasite of the attine ant-microbe symbiosis directly consumes the ant-cultivated fungus. Mycologia 96:955–959
Rodrigues A, Pagnocca FC, Bacci M Jr, Hebling MJA, Bueno OC, Pfenning LH (2005) Variability of non-mutualist fungi associated with Atta sexdens rubropilosa nests. Folia Microbiol 50:421–425
Rodrigues A, Bacci M Jr, Mueller UG, Ortiz A, Pagnocca FC (2008) Microfungal “weeds” in the leafcutter ant symbiosis. Microb Ecol 56:604–614. doi:10.1007/s00248-008-9380-0
Rodrigues A, Cable RN, Mueller UG, Bacci M Jr, Pagnocca FC (2009) Antagonistic interactions between garden yeast and microfungal garden pathogens of leaf-cutting ants. Anton Leeuw Int J G 96:331–342. doi:10.1007/s10482-009-9350-7
Santos AV, Dillon RJ, Dillon VM, Reynolds SE, Samuels RJ (2004) Occurrence of the antibiotic producing bacterium Burkholderia sp. in colonies of the leaf-cutting ant Atta sexdens rubropilosa. FEMS Microbiol Lett 239:319–323. doi:10.1016/j.femsle.2004.09.005
Savoie JM, Mata G, Billette C (1998) Extracellular laccase production during hyphal interactions between Trichoderma sp. and shiitake, Lentinula edodes. Appl Microbiol Biotechnol 49:589–593. doi:10.1007/s002530051218
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to imageJ: 25 years of image analysis. Nat Methods 9:671–675. doi:10.1038/nmeth.2089
Schultz TR, Brady SG (2008) Major evolutionary transitions in ant agriculture. Proc Natl Acad Sci USA 105:5435–5440. doi:10.1073/pnas.0711024105
Sen R, Ishak HD, Estrada D, Dowd SE, Hong E, Mueller UG (2009) Generalized antifungal activity and 454-screening of Pseudonocardia and Amycolatopsis bacteria in nests of fungus-growing ants. Proc Natl Acad Sci USA 106:17805–17810. doi:10.1073/pnas.0904827106
Silva A, Rodrigues A, Bacci M Jr, Pagnocca FC, Bueno OC (2006) Susceptibility of the ant-cultivated fungus Leucoagaricus gongylophorus (Agaricales: Basidiomycota) towards microfungi. Mycopathologia 162:115–119. doi:10.1007/s11046-006-0037-6
Suen G, Scott JJ, Aylward FO, Adams SM, Tringe SG, Pinto-Tomás AA, Foster CE, Pauly M, Weimer PJ, Barry KW, Goodwin LA, Bouffard P, Li L, Osterberger J, Harkins TT, Slater SC, Donohue TJ, Currie CR (2010) An insect herbivore microbiome with high plant biomass-degrading capacity. PLoS Genet 6:e1001129. doi:10.1371/journal.pgen.1001129
Tsujiyama S, Minami M (2005) Production of phenol-oxidizing enzymes in the interaction between white-rot fungi. Mycoscience 46:268–271. doi:10.1007/s10267-005-0243-y
Weber NA (1972) Gardening ants, the Attines. American Philosophical Society, Philadelphia
Acknowledgements
The authors are grateful to “FAPESP - Fundação de Amparo a Pesquisa do Estado de São Paulo” for financial support (Grant 2011/16765-0) and “CAPES - Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” for providing a scholarship to SSVH and the PNPD Grant 1455/2008 to TRA. We also thank Dr. Fernando C. Pagnocca (CEIS/UNESP, Rio Claro) for providing strain FF2006 of the mutualistic fungus used in this study, Dr. Christian Jost (University of Toulouse, France) for helping with statistical analysis, and Quimi Vidaurre Montoya (UNESP, Rio Claro) and Antônio Teruyoshi Yabuki (UNESP, Rio Claro) for technical assistance. We are in debt to three anonymous reviewers that provided constructive comments on this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Varanda-Haifig, S.S., Albarici, T.R., Nunes, P.H. et al. Nature of the interactions between hypocrealean fungi and the mutualistic fungus of leaf-cutter ants. Antonie van Leeuwenhoek 110, 593–605 (2017). https://doi.org/10.1007/s10482-016-0826-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-016-0826-y