Abstract
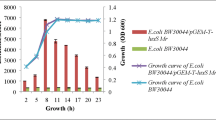
YvqEC is one of the two-component signal transduction systems that may respond to cell envelope stress and enable cells to adjust multiple cellular functions. It consists of a histidine kinase YvqE and a response regulator YvqC. In this study, we separately constructed a single gene mutant ΔyvqE and a double gene mutant ΔyvqEC in Bacillus thuringiensis BMB171 through a homing endonucleases I-SceI mediated markerless gene deletion method. We found that the deletion of either yvqE or yvqEC weakened the resistance of B. thuringiensis against vancomycin. We also identified nine operons that may be involved in the cellular metabolism regulated by YvqC. This study not only enriches our understanding of bacterial resistance mechanisms against vancomycin, but also helps investigate the functions of YvqEC.






Similar content being viewed by others
References
Almarza O, Núñez D, Toledo H (2015) The DNA-binding protein HU has a regulatory role in the acid stress response mechanism in Helicobacter pylori. Helicobacter 20:29–40
Arias CA, Panesso D, McGrath DM, Qin X, Mojica MF, Miller C, Diaz L, Tran TT, Rincon S, Barbu EM, Reyes J, Roh JH, Lobos E, Sodergren E, Pasqualini R, Arap W, Quinn JP, Shamoo Y, Murray BE, Weinstock GM (2011) Genetic basis for in vivo daptomycin resistance in Enterococci. N Engl J Med 365:892–900
Bourret RB, Silversmith RE (2010) Two-component signal transduction. Curr Opin Microbiol 13:113–115
Chiu KC, Lin CJ, Shaw GC (2014) Transcriptional regulation of the l-lactate permease gene lutP by the LutR repressor of Bacillus subtilis RO-NN-1. Microbiology 160:2178–2189
Christodoulou E, Rypniewski WR, Vorgias CR (2003) High-resolution X-ray structure of the DNA-binding protein HU from the hyper-thermophilic Thermotoga maritima and the determinants of its thermostability. Extremophiles 7:111–122
Eldholm V, Gutt B, Johnsborg O, Brückner R, Maurer P, Hakenbeck R, Mascher T, Håvarstein LS (2010) The pneumococcal cell envelope stress-sensing system LiaFSR is activated by murein hydrolases and lipid II-interacting antibiotics. J Bacteriol 192:1761–1773
Endo T, Sasaki N, Tanaka I, Nakata M (2002) Compact form of DNA induced by DNA-binding protein HU. Biochem Biophys Res Commun 290:546–551
Filippova EV, Kuhn ML, Osipiuk J, Kiryukhina O, Joachimiak A, Ballicora MA, Anderson WF (2015) A novel polyamine allosteric site of SpeG from Vibrio cholerae is revealed by its dodecameric structure. J Mol Biol 27:1316–1334
Fritsch F, Mauder N, Williams T, Weiser J, Oberle M, Beier D (2011) The cell envelope stress response mediated by the LiaFSRLm three-component system of Listeria monocytogenes is controlled via the phosphatase activity of the bifunctional histidine kinase LiaSLm. Microbiology 157:373–386
Gao R, Stock AM (2009) Biological Insights from structures of two-component proteins. Annu Rev Microbiol 63:133–154
Hachmann AB, Angert ER, Helmann JD (2009) Genetic analysis of factors affecting susceptibility of Bacillus subtilis to daptomycin. Antimicrob Agents Ch 53:1598–1609
Hancoc L, Perego M (2002) Two-component signal transduction in Enterococcus faecalis. J Bacteriol 184:5819–5825
He J, Shao X, Zheng H, Li M, Wang J, Zhang Q, Li L, Liu Z, Sun M, Wang S, Yu Z (2010) Complete genome sequence of Bacillus thuringiensis mutant strain BMB171. J Bacteriol 192:4074–4075
Hollenbeck BL, Rice LB (2012) Intrinsic and acquired resistance mechanisms in enterococcus. Virulence 3:421–433
Janes BK, Stibitz S (2006) Routine markerless gene replacement in Bacillus anthracis. Infect Immun 74:1949–1953
Johnson JM, Sanford BL, Strom AM, Tadayon SN, Lehman BP, Zirbes AM, Bhattacharyya S, Musier-Forsyth K, Hati S (2013) Multiple pathways promote dynamical coupling between catalytic domains in Escherichia coli prolyl-tRNA synthetase. Biochemistry 52:4399–4412
Jordan S, Hutchings MI, Mascher T (2008) Cell envelope stress response in gram-positive bacteria. FEMS Microbiol Rev 32:107–146
Jordan S, Junker JA, Helmann JD, Mascher T (2006) Regulation of LiaRS-dependent gene expression in Bacillus subtilis: identification of inhibitor proteins, regulator binding sites, and target genes of a conserved cell envelope stress-sensing two-component system. J Bacteriol 188:5153–5166
Jordan S, Rietkötter E, Strauch MA, Kalamorz F, Butcher BG, Helmann JD, Mascher T (2007) LiaRS-dependent gene expression is embedded in transition state regulation in Bacillus subtilis. Microbiology 153:2530–2540
Kuroda M, Kuwahara-Arai K, Hiramatsu K (2000) Identification of the up- and down-regulated genes in vancomycin-resistant Staphylococcus aureus strains Mu3 and Mu50 by cDNA differential hybridization method. Biochem Biophys Res Commun 269:485–490
Kuroda M, Kuroda H, Oshima T, Takeuchi F, Mori H, Hiramatsu K (2003) Two-component system VraSR positively modulates the regulation of cell-wall biosynthesis pathway in Staphylococcus aureus. Mol Microbiol 49:807–821
Kwun MJ, Novotna G, Hesketh AR, Hill L, Hong H (2013) In vivo studies suggest that induction of VanS-dependent vancomycin resistance requires binding of the drug to d-Ala-d-Ala termini in the peptidoglycan cell wall. Antimicrob Agents Ch 57:4470–4480
Li L, Shao Z, Yu Z (2000a) Transformation of Bacillus thuringiensis recipient BMB171 by electroporation. Wei Sheng Wu Xue Tong Bao 27:331–334 (In Chinese)
Li L, Yang C, Liu Z, Li F, Yu Z (2000b) Screening of acrystalliferous mutants from Bacillus thuringiensis and their transformation properties. Wei Sheng Wu Xue Bao 40:85–90 (In Chinese)
Martínez B, Zomer AL, Rodríguez A, Kok J, Kuipers OP (2007) Cell envelope stress induced by the bacteriocin Lcn972 is sensed by the Lactococcal two-component system CesSR. Mol Microbiol 64:473–486
Mascher T (2006) Intramembrane-sensing histidine kinases: a new family of cell envelope stress sensors in Firmicutes bacteria. FEMS Microbiol Lett 264:133–144
Mascher T, Margulis NG, Wang T, Ye RW, Helmann JD (2003) Cell wall stress responses in Bacillus subtilis: the regulatory network of the bacitracin stimulon. Mol Microbiol 50:1591–1604
Mascher T, Zimmer SL, Smith TA, Helmann JD (2004) Antibiotic-inducible promoter regulated by the cell envelope stress-sensing two-component system LiaRS of Bacillus subtilis. Antimicrob Agents Ch 48:2888–2896
Maughan H, Galeano B, Nicholson WL (2004) Novel rpoB mutations conferring rifampin resistance on Bacillus subtilis: global effects on growth, competence, sporulation, and germination. J Bacteriol 186:2481–2486
Moeller R, Vlašić I, Reitz G, Nicholson WL (2012) Role of altered rpoB alleles in Bacillus subtilis sporulation and spore resistance to heat, hydrogen peroxide, formaldehyde, and glutaraldehyde. Arch Microbiol 194:759–767
Su J, Bao P, Bai T, Deng L, Wu H, Liu F, He J (2013) CotA, a multicopper oxidase from Bacillus pumilus WH4, exhibits manganese-oxidase activity. PLoS One 8:e60573
Su J, Deng L, Huang L, Guo S, Liu F, He J (2014) Catalytic oxidation of manganese (II) by multicopper oxidase CueO and characterization of the biogenic Mn oxide. Water Res 56:304–313
Suntharalingam P, Senadheera MD, Mair RW, Lévesque CM, Cvitkovitch DG (2009) The LiaFSR system regulates the cell envelope stress response in Streptococcus mutans. J Bacteriol 191:2973–2984
Szurmant H, Hoch JA (2010) Interaction fidelity in two-component signaling. Curr Opin Microbiol 13:190–197
Wang J, Mei H, Qian H, Tang Q, Liu X, Yu Z, He J (2013a) Expression profile and regulation of spore and parasporal crystal formation-associated genes in Bacillus thuringiensis. J Proteome Res 12:5487–5501
Wang J, Mei H, Zheng C, Qian H, Cui C, Fu Y, Su J, Liu Z, Yu Z, He J (2013b) The metabolic regulation of sporulation and parasporal crystal formation in Bacillus thuringiensis revealed by transcriptomics and proteomics. Mol Cell Proteomics 12:1363–1376
Wecke T, Zühlke D, Mäder U, Jordan S, Voigt B, Pelzer S, Labischinski H, Homuth G, Hecker M, Mascher T (2009) Daptomycin versus Friulimicin B: in-depth profiling of Bacillus subtilis cell envelope stress responses. Antimicrob Agents Ch 53:1619–1623
Wolf D, Kalamorz F, Wecke T, Juszczak A, Mäder U, Homuth G, Jordan S, Kirstein J, Hoppert M, Voigt B, Hecker M, Mascher T (2010) In-depth profiling of the liaR response of Bacillus subtilis. J Bacteriol 192:4680–4693
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant 31270105) and the National High-tech R&D Program of China (863 Program, Grant 2011AA10A205).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, S., Hu, Y., Fan, Q. et al. Two-component system YvqEC-dependent bacterial resistance against vancomycin in Bacillus thuringiensis . Antonie van Leeuwenhoek 108, 365–376 (2015). https://doi.org/10.1007/s10482-015-0489-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0489-0