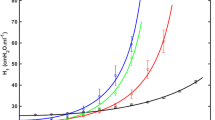
Abstract
Acute respiratory distress syndrome (ARDS) and ventilator-induced lung injury (VILI) are heterogeneous conditions. The spatiotemporal evolution of these heterogeneities is complex, and it is difficult to elucidate the mechanisms driving its progression. Through previous quantitative analyses, we explored the distributions of cellular injury and neutrophil infiltration in experimental VILI and discovered that VILI progression is characterized by both the formation of new injury in quasi-random locations and the expansion of existing injury clusters. Distributions of neutrophil infiltration do not correlate with cell injury progression and suggest a systemic response. To further examine the dynamics of VILI, we have developed a novel computational model that simulates damage (cellular injury progression and neutrophil infiltration) using a stochastic approach. Optimization of the model parameters to fit experimental data reveals that the range and strength of interdependence between existing and new damaged regions both increase as mechanical ventilation patterns become more injurious. The interdependence of cellular injury can be attributed to mechanical tethering forces, while the interdependence of neutrophils is likely due to longer-range cell signaling pathways.











Similar content being viewed by others
References
Ware, L. B., and M. A. Matthay. The acute respiratory distress syndrome. N. Engl. J. Med. 342(18):1334–1349, 2000.
Force, A. D. T., et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 307(23):2526–2533, 2012.
Andrade, M. C., et al. Applying positive end-expiratory pressure during mechanical ventilation causes pulmonary redox imbalance and inflammation in rats. Shock. 50(5):572–578, 2018.
Choudhury, S., et al. Mechanisms of early pulmonary neutrophil sequestration in ventilator-induced lung injury in mice. Am. J. Physiol. Lung. Cell Mol. Physiol. 287(5):L902–L910, 2004.
Seah, A. S., et al. Quantifying the roles of tidal volume and PEEP in the pathogenesis of ventilator-induced lung injury. Ann. Biomed. Eng. 39(5):1505–1516, 2011.
Slutsky, A. S. Lung injury caused by mechanical ventilation. Chest. 116(Suppl. 1):9S-15S, 1999.
Slutsky, A. S., and Y. Imai. Ventilator-induced lung injury, cytokines, PEEP, and mortality: implications for practice and for clinical trials. Intens. Care Med. 29(8):1218–1221, 2003.
Taskar, V., et al. Surfactant dysfunction makes lungs vulnerable to repetitive collapse and reexpansion. Am. J. Respir. Crit. Care Med. 155:313–320, 1997.
Villar, J., et al. The ALIEN study: incidence and outcome of acute respiratory distress syndrome in the era of lung protective ventilation. Intens. Care Med. 37(12):1932–1941, 2011.
Kaczka, D. W., et al. Quantifying mechanical heterogeneity in canine acute lung injury: impact of mean airway pressure. Anesthesiology. 103(2):306–317, 2005.
Sinclair, S. E., N. L. Polissar, and W. A. Altemeier. Spatial distribution of sequential ventilation during mechanical ventilation of the uninjured lung: an argument for cyclical airway collapse and expansion. BMC Pulm. Med. 10:25, 2010.
Fagerberg, A., et al. Electrical impedence tomography and heterogeneity of pulmonary perfusion and ventilation in porcine acute lung injury. Acta Anaesthesiol. Scand. 53(10):1300–1309, 2009.
Porra, L., et al. Quantitative imaging of regional aerosol deposition, lung ventilation and morphology by synchrotron radiation CT. Sci. Rep. 8(1):3519, 2018.
Stahr, C. S., et al. Quantification of heterogeneity in lung disease with image-based pulmonary function testing. Sci. Rep. 6:29438, 2016.
Gattinoni, L., and A. Pesenti. The concept of “baby lung.” Intens. Care Med. 31(6):776–784, 2005.
Perlman, C. E., D. J. Lederer, and J. Bhattacharya. Micromechanics of alveolar edema. Am. J. Respir. Cell Mol. Biol. 44(1):34–39, 2011.
Costa, E. L., et al. Mild endotoxemia during mechanical ventilation produces spatially heterogeneous pulmonary neutrophilic inflammation in sheep. Anesthesiology. 112(3):658–669, 2010.
Gajic, O., et al. Ventilator-induced cell wounding and repair in the intact lung. Am. J. Respir. Crit Care Med. 167(8):1057–1063, 2003.
Le, A., et al. Alveolar cell apoptosis is dependent on p38 MAP kinase-mediated activation of xanthine oxidoreductase in ventilator-induced lung injury. J. Appl. Physiol. 105(4):1282–1290, 2008.
Makena, P. S., et al. Deletion of apoptosis signal-regulating kinase-1 prevents ventilator-induced lung injury in mice. Am. J. Respir. Cell Mol. Biol. 46(4):461–469, 2012.
Otto, C.M., et al., Spatial and temporal heterogeneity of ventilator-associated lung injury after surfactant depletion. J. Appl. Physiol. (1985), 2008. 104(5): p. 1485-94.
Sinclair, S. E., et al. Positive end-expiratory pressure alters the severity and spatial heterogeneity of ventilator-induced lung injury: an argument for cyclical airway collapse. J. Crit. Care. 24(2):206–211, 2009.
Wellman, T. J., et al. Effect of local tidal lung strain on inflammation in normal and lipopolysaccharide-exposed sheep. Crit. Care Med. 42(7):e491-500, 2014.
Motta-Ribeiro, G., et al., Spatial Heterogeneity of Lung Strain and Aeration and Regional Inflammation During Early Lung Injury Assessed with PET/CT. Acad Radiol, 2018.
Paula, L. F., T. J. Wellman, T. Winkler, P. M. Spieth, A. Güldner, J. G. Venegas, M. Gama de Abreu, A. R. Carvalho, and M. F. Vidal Melo. Regional tidal lung strain in mechanically ventilated normal lungs. J. Appl. Physiol. 121(6):1335–47, 2016.
Yen, S., et al., The Link between Regional Tidal Stretch and Lung Injury during Mechanical Ventilation. Am. J. Respir. Cell Mol. Biol. 0(ja): p. null.
Ma, B. and J.H. Bates, Continuum vs. spring network models of airway-parenchymal interdependence. J. Appl. Physiol. (1985), 2012. 113(1): p. 124-9.
Ma, B., B. Breen, and J. H. Bates. Influence of parenchymal heterogeneity on airway-parenchymal interdependence. Respir. Physiol. Neurobiol. 188(2):94–101, 2013.
Ma, B., B. J. Smith, and J. H. Bates. Resistance to alveolar shape change limits range of force propagation in lung parenchyma. Respir. Physiol. Neurobiol. 211:22–28, 2015.
Ma, B. S., and J. H. T. Bates. Mechanical interactions between adjacent airways in the lung. J. Appl. Physiol. 116(6):628–634, 2014.
Mattson, C.L., et al., Spatiotemporal distribution of cellular injury and leukocytes during the progression of ventilator-induced lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2022; 323(3):281–96
Tsuno, K., et al. Histopathologic pulmonary changes from mechanical ventilation at high peak airway pressures. Am. Rev. Respir. Dis. 143(5):1115–1120, 1991.
Villar, J., et al. Experimental ventilator-induced lung injury: exacerbation by positive end-expiratory pressure. Anesthesiology. 110(6):1341–1347, 2009.
Hirsch, J., et al. Double impact of cigarette smoke and mechanical ventilation on the alveolar epithelial type II cell. Crit. Care. 18(2):R50, 2014.
Mattson, C., et al., Spatiotemporal Distribution of Cellular Injury and Leukocytes during the Progression of Ventilator-Induced Lung Injury: Online Supplement. 2022: figshare.
Blanca, M. J., et al. Non-normal data: is ANOVA still a valid option? Psicothema. 29(4):552–557, 2017.
Makiyama, A. M., et al. Stress concentration around an atelectatic region: a finite element model. Respir. Physiol. Neurobiol. 201:101–110, 2014.
Albert, R. K., et al. Is progression of pulmonary fibrosis due to ventilation-induced lung injury? Am. J. Respir. Crit. Care Med. 200(2):140–151, 2019.
Reynolds, A., et al. Cellular automata modeling of pulmonary inflammation. Mol. Cell Biomech. 9(2):141–156, 2012.
Ibrahim, I. B. M., O. V. S. Sarma, and R. M. Pidaparti. Simulation of healing threshold in strain-induced inflammation through a discrete informatics model. IEEE J. Biomed. Health Inform. 22(3):935–941, 2018.
Wilson, M. R., and M. Takata. Inflammatory mechanisms of ventilator-induced lung injury: a time to stop and think? Anaesthesia. 68(2):175–178, 2013.
Acknowledgements
This work was supported by the National Institutes of Health Grants R01 HL151630 (“Predicting and Preventing Ventilator-Induced Lung Injury”; to B.J.S.) and F31 HL149268 (“Spatiotemporal evolution of lung injury during ventilator-induced lung injury”; to C.L.M.); and National Science Foundation Grant 2225554 (‘”RECODE: Defining Environmental Design Criteria for Directed Differentiation of Type 1 from Type 2 Lung Alveolar Epithelial Cells,” to B.J.S and C.L.M.). This work used the computing resources at the Center for Computational Mathematics, University of Colorado Denver, including the Alderaan cluster, supported by the National Science Foundation award OAC-2019089.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Additional information
Associate Editor Merryn Tawhai oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 6275 kb)
Supplementary file2 (MP4 6356 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mattson, C.L., Smith, B.J. Modeling Ventilator-Induced Lung Injury and Neutrophil Infiltration to Infer Injury Interdependence. Ann Biomed Eng 51, 2837–2852 (2023). https://doi.org/10.1007/s10439-023-03346-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-023-03346-3