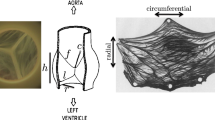
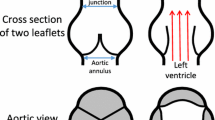
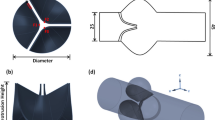
Abstract
The simplified and modified Bernoulli equations break down in estimating the true pressure gradient across the stenotic aortic valve due to their over simplifying assumptions of steady and inviscid conditions as well as the fundamental nature in which aortic valves are different than idealized orifices. Nevertheless, despite having newer models based on time-dependent momentum balance across an orifice, the simplified and modified Bernoulli continue to be the clinical standard because to date, they remain the only models clinically implementable. The objective of this study is to (1) illustrate the fundamental considerations necessary to accurately model the time-dependent instantaneous pressure gradient across a fixed orifice and (2) propose empirical corrections when applying orifice based models to severely stenotic aortic valves. We introduce a general model to predict the time-dependent instantaneous pressure gradient across an orifice that explicitly model the Reynolds number dependence of both the steady and unsteady terms. The accuracy of this general model is assessed with respect to previous models through pulse duplicator experiments on a round orifice model as well as an explanted stenotic surgical aortic valve both with geometric areas of 0.6 cm2 (less than 1 cm2 which is the threshold for stenosis determination) over cardiac outputs of 3 and 5 L/min and heart rates of 60, 90 and 120 bpm. The model and the raw experimental data corresponding to the orifice showed good agreement over a wide range of cardiac outputs and heart rates (R2 exceeding 0.91). The derived average and peak transvalvular pressure gradients also demonstrated good agreement with no significant differences between the respective peaks (p > 0.09). The new model proposed holds promise with its physical and closed form representation of pressure drop, however accurate modeling of the time-variability of the valve area is necessary for the model to be applied on stenotic valves.











Similar content being viewed by others
Abbreviations
- PG:
-
Pressure gradient
- BPS:
-
Beats per second
- bpm:
-
Beats per minute
- HR:
-
Heart rate
- CO:
-
Cardiac output
- EOA:
-
Effective orifice area
- Re:
-
Reynolds number
References
Baumgartner, H., J. Hung, J. Bermejo, J. B. Chambers, A. Evangelista, B. P. Griffin, B. Iung, C. M. Otto, P. A. Pellikka, and M. Quiñones. Echocardiographic assessment of valve stenosis: EAE/ASE recommendations for clinical practice. J. Am. Soc. Echocardiogr. 22(1):1–23, 2009.
Baumgartner, H., T. Stefenelli, J. Niederberger, H. Schima, and G. Maurer. “Overestimation” of catheter gradients by Doppler ultrasound in patients with aortic stenosis: a predictable manifestation of pressure recovery. J. Am. Coll. Cardiol. 33(6):1655–1661, 1999.
Berger, M., R. L. Berdoff, P. E. Gallerstein, and E. Goldberg. Evaluation of aortic stenosis by continuous wave Doppler ultrasound. J. Am. Coll. Cardiol. 3(1):150–156, 1984.
Bermejo, J., J. C. Antoranz, I. G. Burwash, J. Alvarez, M. Moreno, M. A. García-Fernández, and C. M. Otto. In-vivo analysis of the instantaneous transvalvular pressure difference-flow relationship in aortic valve stenosis: implications of unsteady fluid-dynamics for the clinical assessment of disease severity. J. Heart Valve Dis. 11(4):557–566, 2002.
Carabello, B. A. Advances in the Hemodynamic Assessment of Stenotic Cardiac Valves. Amsterdam: Elsevier, 1987.
Chandran, K. B., S. E. Rittgers, and A. P. Yoganathan. Biofluid Mechanics: The Human Circulation. Boca Raton: CRC Press, 2012.
Clark, C. Relation between pressure difference across the aortic valve and left ventricular outflow. Cardiovasc. Res. 12(5):276–287, 1978.
Dasi, L. P., H. A. Simon, P. Sucosky, and A. P. Yoganathan. Fluid mechanics of artificial heart valves. Clin. Exp. Pharmacol. Physiol. 36(2):225–237, 2009.
DeGroff, C. G., R. Shandas, and L. Valdes-Cruz. Analysis of the effect of flow rate on the Doppler continuity equation for stenotic orifice area calculations: a numerical study. Circulation 97(16):1597–1605, 1998.
Fusman, B., D. Faxon, and T. Feldman. Hemodynamic rounds: Transvalvular pressure gradient measurement. Catheter. Cardiovasc. Interv. 53(4):553–561, 2001.
Garcia, D., L. Kadem, D. Savéry, P. Pibarot, and L.-G. Durand. Analytical modeling of the instantaneous maximal transvalvular pressure gradient in aortic stenosis. J. Biomech. 39(16):3036–3044, 2006.
Garcia, D., P. Pibarot, and L.-G. Durand. Analytical modeling of the instantaneous pressure gradient across the aortic valve. J. Biomech. 38(6):1303–1311, 2005.
Hatoum, H., J. Dollery, S. M. Lilly, J. A. Crestanello, and L. P. Dasi. Implantation depth and rotational orientation effect on valve-in-valve hemodynamics and sinus flow. Ann. Thorac. Surg. 106:70–78, 2018.
Hatoum, H., F. Heim, and L. P. Dasi. Stented valve dynamic behavior induced by polyester fiber leaflet material in transcatheter aortic valve devices. J. Mech. Behav. Biomed. Mater. 86:232–239, 2018.
Hatoum, H., P. Maureira, and L. P. Dasi. A turbulence in vitro assessment of On-X and St Jude medical prostheses. J. Thorac. Cardiovasc. Surg. 2019. https://doi.org/10.1016/j.jtcvs.2019.02.046.
Hatoum, H., B. L. Moore, and L. P. Dasi. On the significance of systolic flow waveform on aortic valve energy loss. Ann. Biomed. Eng. 46:2102–2111, 2018.
Hatoum, H., A. Yousefi, S. Lilly, P. Maureira, J. Crestanello, and L. P. Dasi. An in-vitro evaluation of turbulence after transcatheter aortic valve implantation. J. Thorac. Cardiovasc. Surg. 156:1837–1848, 2018.
Kheradvar, A., E. M. Groves, A. Falahatpisheh, M. K. Mofrad, S. Hamed Alavi, R. Tranquillo, L. P. Dasi, C. A. Simmons, K. Jane Grande-Allen, C. J. Goergen, F. Baaijens, S. H. Little, S. Canic, and B. Griffith. Emerging trends in heart valve engineering: Part IV. Computational modeling and experimental studies. Ann. Biomed. Eng. 43(10):2314–2333, 2015.
LaNieve, H. L. Entrance Effects in Non-Newtonian Pipe Flow. Knoxville: University of Tennessee, 1963.
Levine, R. A., and E. Schwammenthal. Stenosis is in the eye of the observer: impact of pressure recovery on assessing aortic valve area. J. Am. Coll. Cardiol. 41(3):443–445, 2003.
Otto, C. M. Valvular aortic stenosis: disease severity and timing of intervention. J. Am. Coll. Cardiol. 47(11):2141–2151, 2006.
Padala, M., E. L. Sarin, P. Willis, V. Babaliaros, P. Block, R. A. Guyton, and V. H. Thourani. An engineering review of transcatheter aortic valve technologies. Cardiovasc. Eng. Technol. 1(1):77–87, 2010.
Pibarot, P., D. Garcia, and J. G. Dumesnil. Energy loss index in aortic stenosis: from fluid mechanics concept to clinical application. Am. Heart Assoc. 127:1101–1104, 2013.
Sotiropoulos, F., T. B. Le, and A. Gilmanov. Fluid mechanics of heart valves and their replacements. Ann. Rev. Fluid Mech. 48:259–283, 2016.
Thaden, J. J., V. T. Nkomo, K. J. Lee, and J. K. Oh. Doppler imaging in aortic stenosis: the importance of the nonapical imaging windows to determine severity in a contemporary cohort. J. Am. Soc. Echocardiogr. 28(7):780–785, 2015.
VanAuker, M. D., M. Chandra, J. Shirani, and J. A. Strom. Jet eccentricity: a misleading source of agreement between Doppler/catheter pressure gradients in aortic stenosis. J. Am. Soc. Echocardiogr. 14(9):853–862, 2001.
White, F. M. Fluid Mechanics, McGraw-Hill Series in Mechanical Engineering. Boston: McGraw-Hill International Editions, 1998.
Young, D. F. Fluid-mechanics of arterial stenoses. J. Biomech. Eng. Trans. ASME 101(3):157–175, 1979.
Conflict of interest
Dr. Crestanello reports having grants from Medtronic, Boston Scientific and St. Jude in addition to being part of Medtronic advisory board. Dr. Dasi reports having patent applications filed on novel polymeric valves, vortex generators and superhydrophobic/omniphobic surfaces. No other conflicts were reported.
Funding
The research done was partly supported by National Institutes of Health (NIH) under Award Number R01HL119824 and the American Heart Association (AHA) under Award Number 19POST34380804.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Dan Elson oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A: Non-dimensional Analysis
Appendix A: Non-dimensional Analysis
From non-dimensional analysis, we choose to select 6 different variables \(\left( {\Delta P, \rho , V, D, \nu , \frac{\text{d}}{{{\text{d}}t}}\left( {\frac{{\rho V^{2} }}{2}} \right)} \right)\) where \(\nu\) is the kinematic viscosity.
The π groups obtained can be summarized:
Stemming from Eq. (4), the relationship between the π groups can be written as follows:
Rights and permissions
About this article
Cite this article
Hatoum, H., Mo, XM., Crestanello, J.A. et al. Modeling of the Instantaneous Transvalvular Pressure Gradient in Aortic Stenosis. Ann Biomed Eng 47, 1748–1763 (2019). https://doi.org/10.1007/s10439-019-02275-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02275-4