Abstract
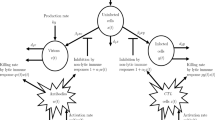
In this paper we consider a mathematical model of HIV-virus dynamics and propose an efficient control strategy to keep the number of HIV virons under a pre-specified level and to reduce the total amount of medications that patients receive. The model considered is a nonlinear third-order model. The third-order model describes dynamics of three most dominant variables: number of healthy white blood cells (T-cells), number of infected T-cells, and number of virus particles. There are two control variables in this model corresponding to two categories of antiviral drugs: reverse transcriptase inhibitors (RTI) and protease inhibitors (PI). The proposed strategy is based on linearization of the nonlinear model at the equilibrium point (steady state). The corresponding controller has two components: the first one that keeps the system state variables at the desired equilibrium (set-point controller) and the second-one that reduces in an optimal way deviations of the system state variables from their desired equilibrium values. The second controller is based on minimization of the square of the error between the actual and desired (equilibrium) values for the linearized system (linear-quadratic optimal controller). The obtained control strategy recommends to HIV researchers and experimentalists that the constant dosages of drugs have to be administrated at all times (set point controller, open-loop controller) and that the variable dosages of drugs have to be administrated on a daily basis (closed-loop controller, feedback controller).

















Similar content being viewed by others
References
Astrom, K., and M. Wittenmark. PID Controllers Theory, Design, and Tuning. Instrument Society of America, 1995.
Banks, H., H. Kwon, J. Toivanen, and H. Tran. A state-dependent Riccati equation-based estimator approach for HIV feedback control. Optimal Control Applications and Method, 27, 93-121, 2006.
Brandt, M. and G. Chen. Feedback control of a biodynamical model of HIV-1. IEEE Transactions on Biomedical Engineering, 48:754-759, 2001. doi:10.1109/10.930900.
Chen, T-C. Linear Systems and Design Oxford University Press, Oxford, United Kingdom, 1999.
Craig, I. and X. Xia. Can HIV/AIDS be controlled. IEEE Control Systems Magazine, 25:80-83, 2005. doi:10.1109/MCS.2005.1388805.
Craig, I. X, Xia, and J. Venter. Introducing HIV/AIDS education into the electrical engineering curriculum at the university of Pretoria. IEEE Transactions on Education, 47:65-73, 2004. doi:10.1109/TE.2003.817620.
Culshaw, R. Review of HIV models: The role of the natural immune response and implications for treatment. J. Biol. Syst., 12:123-135, 2004. doi:10.1142/S0218339004001099.
de Souza, F. Modeling the Dynamics of HIV-1 and CD4 and CD8 lymphocytes. Engineering in Medicine and Biology, 18, 21-24, 1999.
Friedland, B. Advanced Control System Design. Prentice Hall, Englewood Cliffs, 1996.
Ge, S. Z. Tian, and T. Lee. Nonlinear control of dynamic model of HIV-1. IEEE Transactions on Biomedical Engineering, 52:353-361, 2005. doi:10.1109/TBME.2004.840463.
Havlir, D. and D. Richman. Viral dynamics of HIV: Implications for drug development and therapeutic strategies. Annals of Internal Medicine, 124:984-994, 1996.
Ho, D. D, A. U. Neumann, A. S. Perelson, W. Chen, J. M. Leonard, and M. Markowitz. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature, 373:123-126, 1995. doi:10.1038/373123a0.
Jeffrey, A. M., X. Xia, and K. Craig. When to initiate HIV therapy: a control theoretic approach. IEEE Transactions on Biomedical Engineering, 50:1213-1220, 2003. doi:10.1109/TBME.2003.818465.
Khalil, H. Nonlinear Systems. Prentice Hall, Englewood Cliffs, 2002.
Ko, J. W. Kim, and C. Chung. Optimized structural interpretation for HIV therapy and its performance analysis on controllability. IEEE Transactions on Biomedical Engineering, 53:380-386, 2006. doi:10.1109/TBME.2005.869651.
Kremling, A. and J. Saez-Rodriguez. Systems biology—An engineering perspective. Journal of Biotechnology, 129:329-351, 2007. doi:10.1016/j.jbiotec.2007.02.009.
Nowak, M. and R. May. Virus Dynamics: Mathematical Principles of Immunology and Virology. New York, Oxford Press, 2000.
Perelson, A. and P. Nelson. Mathematical analysis of HIV-I dynamics in vivo. SIAM Review, 41:3-44, 1999. doi:10.1137/S0036144598335107.
Perelson, A. S., A. U. Neumann, M. Markowitz, J. M. Leonard, and D. D. Ho. HIV-1 dynamics in vivo: Virion clearance rate, infected cell lifespan and viral generation time. Science, 271: 582-1586, 1996. doi:10.1126/science.271.5255.1582.
Sontag, E. D. New directions in control theory inspired by systems biology. Systems Biology, 1:9-18, 2004. doi:10.1049/sb:20045006.
Sontag, E. D. Molecular system biology and control. European Journal of Control, 11:396-435, 2005. doi:10.3166/ejc.11.396-435.
U.S. Department of Health and Human Services. Guidelines for the use of antiretroviral agents in HIV-infected adults and adolescents. Online http://www.aidsinfo.nih.gov/guidelines, 2003.
Xia, X. Estimation of HIV/AIDS parameters. Automatica, 39: 1983-1988, 2003. doi:10.1016/S0005-1098(03)00220-6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radisavljevic-Gajic, V. Optimal Control of HIV-Virus Dynamics. Ann Biomed Eng 37, 1251–1261 (2009). https://doi.org/10.1007/s10439-009-9672-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-009-9672-7