Abstract
Background
The rate of metachronous recurrence after endoscopic submucosal dissection for early-stage esophageal squamous cell carcinoma and hypopharynx squamous cell carcinoma is as high (10–15%). The acetaldehyde breath test may detect acetaldehyde dehydrogenase 2 gene polymorphisms. Therefore, we evaluated its usefulness in assessing metachronous recurrence in patients with esophageal squamous cell carcinoma and hypopharynx squamous cell carcinoma.
Methods
A total of 76 patients underwent endoscopic submucosal dissection for esophageal squamous cell carcinoma and hypopharynx squamous cell carcinoma and were followed up for at least 3 years (non-recurrence group: 52 patients; recurrence group: 24 patients). The risk factors for carcinogenesis were compared between the recurrence and non-recurrence groups, and the acetaldehyde-to-ethanol ratio was assessed. The cutoff acetaldehyde-to-ethanol ratio that correlated with recurrence was established, and the cumulative recurrence rate was evaluated.
Results
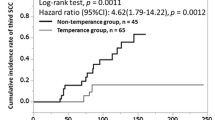
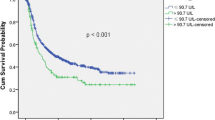
The recurrence group had a higher acetaldehyde-to-ethanol ratio, daily alcohol consumption, and Lugol-voiding lesion grade than the non-recurrence group in the univariate analysis. The cutoff acetaldehyde-to-ethanol ratio for recurrence was 28.1 based on the receiver operating characteristic curve. The multivariate analysis revealed an acetaldehyde-to-ethanol ratio of > 28.1 and a Lugol-voiding lesion grade associated with carcinogenesis. Patients with an acetaldehyde-to-ethanol ratio of ≥ 28.1 had a significantly high recurrence rate using the Kaplan–Meier method.
Conclusions
The acetaldehyde-to-ethanol ratio detected using the acetaldehyde breath test could be a novel biomarker of metachronous recurrence after endoscopic submucosal dissection in patients with esophageal squamous cell carcinoma and hypopharynx squamous cell carcinoma.
Trial registration number
UMIN000040615.




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, [FS], upon reasonable request.
References
Baan R, Straif K, Grosse Y, et al. Carcinogenicity of alcoholic beverages. Lancet Oncol. 2007;8:292–3.
Ohashi S, Miyamoto S, Kikuchi O, et al. Recent advances from basic and clinical studies of esophageal squamous cell carcinoma. Gastroenterology. 2015;149:1700–15.
Katada C, Muto M, Nakayama M, et al. Risk of superficial squamous cell carcinoma developing in the head and neck region in patients with esophageal squamous cell carcinoma. Laryngoscope. 2012;122:1291–6.
Muto M, Minashi K, Yano T, et al. Early detection of superficial squamous cell carcinoma in the head and neck region and esophagus by narrow band imaging: a multicenter randomized controlled trial. J Clin Oncol. 2010;28:1566–72.
Abe S, Hirai Y, Uozumi T, et al. Endoscopic resection of esophageal squamous cell carcinoma: current indications and treatment outcomes. DEN Open. 2022;2: e45.
Kawashima K, Abe S, Koga M, et al. Optimal selection of endoscopic resection in patients with esophageal squamous cell carcinoma: endoscopic mucosal resection versus endoscopic submucosal dissection according to lesion size. Dis Esophagus. 2021. https://doi.org/10.1093/dote/doaa096.
Urabe Y, Hiyama T, Tanaka S, et al. Metachronous multiple esophageal squamous cell carcinomas and Lugol-voiding lesions after endoscopic mucosal resection. Endoscopy. 2009;41:304–9.
Shimizu Y, Tukagoshi H, Fujita M, et al. Metachronous squamous cell carcinoma of the esophagus arising after endoscopic mucosal resection. Gastrointest Endosc. 2001;54:190–4.
Hori K, Okada H, Kawahara Y, et al. Lugol-voiding lesions are an important risk factor for a second primary squamous cell carcinoma in patients with esosphageal cancer or head and neck cancer. Am J Gastroenterol. 2011;106:858–66.
Katada C, Yokoyama T, Yano T, et al. Alcohol consumption and multiple dysplastic lesions increase risk of squamous cell carcinoma in the esophagus, head, and neck. Gastroenterology. 2016;151:860-869.e7.
Secretan B, Straif K, Baan R, et al. A review of human carcinogens–part E: tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol. 2009;10:1033–4.
Yokoyama A, Muramatsu T, Ohmori T, et al. Esophageal cancer and aldehyde dehydrogenase-2 genotypes in Japanese males. Cancer Epidemiol Biomarkers Prev. 1996;5:99–102.
Yokoyama A, Mizukami T, Yokoyama T. Genetic polymorphisms of alcohol dehydrogense-1B and aldehyde dehydrogenase-2, alcohol flushing, mean corpuscular volume, and aerodigestive tract neoplasia in Japanese drinkers. Adv Exp Med Biol. 2015;815:265–79.
Yokoyama A, Omori T, Yokoyama T. Alcohol and aldehyde dehydrogenase polymorphisms and a new strategy for prevention and screening for cancer in the upper aerodigestive tract in East Asians. Keio J Med. 2010;59:115–30.
Bosron WF, Li TK. Genetic polymorphism of human liver alcohol and aldehyde dehydrogenases, and their relationship to alcohol metabolism and alcoholism. Hepatology. 1986;6:502–10.
Crabb DW, Edenberg HJ, Bosron WF, Li TK. Genotypes for aldehyde dehydrogenase deficiency and alcohol sensitivity. The inactive ALDH2(2) allele is dominant. J Clin Invest. 1989;83:314–6.
Lai CL, Yao CT, Chau GY, et al. Dominance of the inactive Asian variant over activity and protein contents of mitochondrial aldehyde dehydrogenase 2 in human liver. Alcohol Clin Exp Res. 2014;38:44–50.
Gross ER, Zambelli VO, Small BA, et al. A personalized medicine approach for Asian Americans with the aldehyde dehydrogenase 2*2 variant. Annu Rev Pharmacol Toxicol. 2015;55:107–27.
Boyle P, Autier P, Bartelink H, et al. European code against cancer and scientific justification: third version (2003). Ann Oncol Third Vers. 2003;14:973–1005.
Aoyama I, Ohashi S, Amanuma Y, et al. Establishment of a quick and highly accurate breath test for ALDH2 genotyping. Clin Transl Gastroenterol. 2017;8: e96.
Sasaki F, Kanmura S, Oda K, et al. Acetaldehyde breath test as a cancer risk marker in patients with esophageal and hypopharyngeal squamous cell carcinoma. PLoS ONE. 2021;16: e0251457.
Yokoyama T, Yokoyama A, Kumagai Y, et al. Health risk appraisal models for mass screening of esophageal cancer in Japanese men. Cancer Epidemiol Biomarkers Prev. 2008;17:2846–54.
Lachenmeier DW, Sohnius EM. The role of acetaldehyde outside ethanol metabolism in the carcinogenicity of alcoholic beverages: evidence from a large chemical survey. Food Chem Toxicol. 2008;46:2903–11.
Hanada M, Koda H, Onaga K, et al. Portable oral malodor analyzer using highly sensitive In2O3 gas sensor combined with a simple gas chromatography system. Anal Chim Acta. 2003;475:27–35.
Ishihara R, Iishi H, Uedo N, et al. Comparison of EMR and endoscopic submucosal dissection for en bloc resection of early esophageal cancers in Japan. Gastrointest Endosc. 2008;68:1066–72.
Muto M, Katada C, Yokoyama T, et al. Field effect of alcohol, cigarette smoking, and their cessation on the development of multiple dysplastic lesions and squamous cell carcinoma: a long-term multicenter cohort study. Gastro Hep Adv. 2022;1:265–76.
Matsuno K, Ishihara R, Nakagawa K, et al. Endoscopic findings corresponding to multiple Lugol-voiding lesions in the esophageal background mucosa. J Gastroenterol Hepatol. 2019;34:390–6.
Ono S, Kawada K, Dohi O, et al. Linked color imaging focused on neoplasm detection in the upper gastrointestinal tract: a randomized trial. Ann Intern Med. 2021;174:18–24.
Maeda H, Sasaki F, Kabayama M, et al. Efficacy of endoscopic ultrasonography in evaluating tumor thickness in patients with superficial pharyngeal carcinoma: a pilot study. Auris Nasus Larynx. 2022;49:495–503.
Väkeväinen S, Tillonen J, Agarwal DP, Srivastava N, Salaspuro M. High salivary acetaldehyde after a moderate dose of alcohol in ALDH2-deficient subjects: strong evidence for the local carcinogenic action of acetaldehyde. Alcohol Clin Exp Res. 2000;24:873–7.
Yokoi A, Maruyama T, Yamanaka R, et al. Relationship between acetaldehyde concentration in mouth air and tongue coating volume. J Appl Oral Sci. 2015;23:64–70.
Acknowledgements
The authors would like to thank Ms. Yuko Morinaga-Nakamura for her technical assistance.
Author information
Authors and Affiliations
Contributions
Conceptualization – FS. Data curation—SM, FS. Formal analysis – KO. Funding acquisition – AI. Investigation—KO, HY, HM. Methodology – SK. Project administration—KO, AI. Resources – SA. Software – SM. Supervision – SH. Validation—ST, AT. Visualization – KK. Writing—original draft – FS. Writing—review & editing—FS, SM, KO, HY, HM, AT, SA, KK, ST, SH, SK, AI.
Corresponding author
Ethics declarations
Conflict of interest
Fumisato Sasaki, Seiichi Mawatari, Kohei Oda, Hiroki Yano, Hidehito Maeda, Akihito Tanaka, Shiho Arima, Kotaro Kumagai, Shiroh Tanoue, Shinichi Hashimoto, Shuji Kanmura, and Akio Ido declare that they have no conflict of interest.
Ethical Statement
This study was approved by the Research Ethics Committee of Kagoshima University (no. 28–137). A written informed consent was obtained from all participants. The procedures were performed in accordance with the 1964 Declaration of Helsinki and its later amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sasaki, F., Mawatari, S., Oda, K. et al. Usefulness of the aldehyde breath test for predicting metachronous recurrence in patients with esophageal squamous cell carcinoma and hypopharyngeal squamous cell carcinoma. Esophagus 20, 749–756 (2023). https://doi.org/10.1007/s10388-023-01024-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-023-01024-w