Abstract
Background
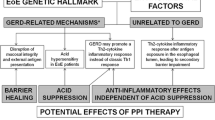
Eosinophilic esophagitis (EoE) is a chronic allergic disease with esophageal symptoms and intraepithelial eosinophil infiltration. Effects of potassium-competitive acid blockers (P-CABs) on EoE have not been elucidated. We aimed to examine and compare the effects of P-CABs and PPIs on symptomatic, endoscopic, and histological responses of patients with EoE.
Methods
We analyzed 118 EoE patients who received PPI or P-CAB therapy with rabeprazole 10 mg (RPZ10, N = 22), rabeprazole 20 mg (RPZ20, N = 34), esomeprazole 20 mg (EPZ20, N = 25), or vonoprazan 20 mg (VPZ20, N = 33). We evaluated symptomatic responses by classifying the patients into three groups: complete relief, partial relief, and no change. Endoscopic responses were evaluated using the endoscopic reference score (EREFS) following PPI or P-CAB therapy. Histological responses were evaluated by determining eosinophil counts in esophageal biopsy samples and classifying the patients into two groups: complete remission [0/1 eosinophil/high-power field (eos/HPF)] and remission (< 15 eos/HPF).
Results
There were no differences among the therapy groups in terms of clinical characteristics, endoscopic findings, and histological findings of the patients before treatment. The rate of complete relief in clinical symptoms was 54.5% in the RPZ10 group, 64.7% in the RPZ20 group, 72.0% in the EPZ20 group, and 75.7% in the VPZ20 group. There were no significant differences in the therapeutic effect among the therapy groups. Similarly, endoscopic and histological complete remission rates were not significantly different among the therapy groups.
Conclusions
Vonoprazan showed similar efficacy to PPIs in EoE.


Similar content being viewed by others
References
Dellon ES, Liacouras CA, Molina-Infante J, et al. Updated International Consensus Diagnostic Criteria for Eosinophilic Esophagitis: Proceedings of the AGREE Conference. Gastroenterology. 2018;155(4):1022–33.
Fujiwara Y, Hashimoto A, Uemura R, et al. Optimal biopsy protocol to evaluate histological effectiveness of proton pump inhibitor therapy in patients with eosinophilic esophagitis. Digestion. 2018;8:1–8. https://doi.org/10.1159/000494253.
Hruz P, Straumann A, Bussmann C, et al. Escalating incidence of eosinophilic esophagitis: a 20-year prospective, population-based study in Olten County, Switzerland. J Allergy Clin Immunol. 2011;128:1349–50.e5.
Furuta GT, Katzka DA. Eosinophilic esophagitis. N Engl J Med. 2015;373(17):1640–8.
van Rhijn BD, van Ree R, Versteeg SA, et al. Birch pollen sensitization with cross-reactivity to food allergens predominates in adults with eosinophilic esophagitis. Allergy. 2013;68(11):1475–81.
Sayej WN, Patel R, Baker RD, et al. Treatment with high-dose proton pump inhibitors helps distinguish eosinophilic esophagitis from noneosinophilic esophagitis. J Pediatr Gastroenterol Nutr. 2009;49(4):393–9.
Moawad FJ, Schoepfer AM, Safroneeva E, et al. Eosinophilic oesophagitis and proton pump inhibitor-responsive oesophageal eosinophilia have similar clinical, endoscopic and histological findings. Aliment Pharmacol Ther. 2014;39(6):603–8.
Sakurai Y, Mori Y, Okamoto H, et al. Acid-inhibitory effects of vonoprazan 20 mg compared with esomeprazole 20 mg or rabeprazole 10 mg in healthy adult male subjects—a randomised open-label cross-over study. Aliment Pharmacol Ther. 2015;42(6):719–30.
Ishimura N, Ishihara S, Kinoshita Y. Sustained acid suppression by potassium-competitive acid blocker (P-CAB) may be an attractive treatment candidate for patients with eosinophilic esophagitis. Am J Gastroenterol. 2016;111(8):1203–4.
Kagami T, Sahara S, Ichikawa H, et al. Potent acid inhibition by vonoprazan in comparison with esomeprazole, with reference to CYP2C19 genotype. Aliment Pharmacol Ther. 2016;43(10):1048–59.
Lucendo AJ, Molina-Infante J, Arias Á, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United European Gastroenterol J. 2017;5:335–58.
Hirano I, Moy N, Heckman MG, et al. Endoscopic assessment of the oesophageal features of eosinophilic oesophagitis: validation of a novel classification and grading system. Gut. 2013;62(4):489–95.
Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80.
Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969;1:87–97.19.
Matsushita T, Maruyama R, Ishikawa N, et al. The number and distribution of eosinophils in the adult human gastrointestinal tract: a study and comparison of racial and environmental factors. Am J Surg Pathol. 2015;39(4):521–7.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Hashimoto A, Sugawa T, Iwakura N, et al. The predictive factors of responsiveness to proton pump inhibitor therapy for eosinophilic esophagitis. Gastrointest Disord. 2019;1(1):220–30.
Tanaka F, Fukumoto S, Morisaki T, et al. Obesity and hiatal hernia may be non-allergic risk factors for esophageal eosinophilia in Japanese adults. Esophagus. 2019;16(3):309–15.
Lafourcade C, Sobo K, Kieffer-Jaquinod S, et al. Regulation of the V-ATPase along the endocytic pathway occurs through reversible subunit association and membrane localization. PLoS ONE. 2008;3(7):e2758.
Suzuki M, Mori M, Miura S, et al. Omeprazole attenuates oxygen-derived free radical production from human neutrophils. Free Radic Biol Med. 1996;21(5):727–31.
Handa O, Yoshida N, Fujita N, et al. Molecular mechanisms involved in anti-inflammatory effects of proton pump inhibitors. Inflamm Res. 2006;55(11):476–80.
Cheng E, Zhang X, Huo X, et al. Omeprazole blocks eotaxin-3 expression by oesophageal squamous cells from patients with eosinophilic oesophagitis and GORD. Gut. 2013;62(6):824–32.
Zhang X, Cheng E, Huo X, et al. Omeprazole blocks STAT6 binding to the eotaxin-3 promoter in eosinophilic esophagitis cells. PLoS ONE. 2012;7(11):e50037.
Ma C, van Rhijn BD, Jairath V, et al. Heterogeneity in clinical, endoscopic, and histologic outcome measures and placebo response rates in clinical trials of eosinophilic esophagitis: a systematic review. Clin Gastroenterol Hepatol. 2018;16(11):1714–1729.e3.
Author information
Authors and Affiliations
Contributions
Data collection was performed by TK. Data analysis and manuscript writing were performed by TK and FT. This study was designed by YF. YF, AS, YN, KO, SH, NK, KT, YN, TT, and TW revised the manuscript critically for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical statement
The study protocol was in accordance with the Declaration of Helsinki and was approved by the institutional review board of the Osaka City University Graduate School of Medicine (September 2018, Protocol number 4141). The need for informed consent has been waived by the institutional review board of the Osaka City University Graduate School of Medicine. We have disclosed the information about this study on our home page on the Internet and the patients had the opportunity to opt-out.
Conflict of interest
All authors have declared no conflict of interest with regard to this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kuzumoto, T., Tanaka, F., Sawada, A. et al. Vonoprazan shows efficacy similar to that of proton pump inhibitors with respect to symptomatic, endoscopic, and histological responses in patients with eosinophilic esophagitis. Esophagus 18, 372–379 (2021). https://doi.org/10.1007/s10388-020-00783-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-020-00783-0