Abstract
Purpose
To investigate the effectiveness of displacement of submacular hemorrhage (SMH) caused by polypoidal choroidal vasculopathy (PCV) by assessing retinal sensitivity using microperimetry.
Methods
We retrospectively reviewed the medical records of 11 consecutive PCV patients with SMH. All patients underwent vitrectomy, subretinal injection of tissue plasminogen activator, and fluid-air exchange, followed by antivascular endothelial growth factor therapy using a pro re nata regimen. The retinal sensitivity was measured by use of microperimetry before and after surgery.
Results
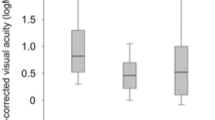
The mean (SD) age of the patients was 74.1 ± 9.4 years. The mean SMH diameter was 6.8 ± 5.2 disc diameters. The best-corrected visual acuity (BCVA), mean retinal sensitivity, and mean number of measure points with a sensitivity ≥10 dB before the surgery were 0.94 ± 0.49, 4.2 ± 4.5 dB, and 15.6 ± 15.1 points, respectively. These had significantly improved 6 months after surgery (0.39 ± 0.37, 15.6 ± 7.3 dB, and 50.9 ± 22.2 points, respectively; P < 0.05 for all outcome measures). The mean number of measure points with an absolute scotoma before surgery had decreased significantly 6 months after surgery (from 40.5 ± 15.0 to 9.4 ± 16.0 points; P < 0.001).
Conclusions
Displacement of SMH effectively improves retinal sensitivity as well as BCVA.


Similar content being viewed by others
References
Uyama M, Wada M, Nagai Y, Matsubara T, Matsunaga H, Fukushima I, et al. Polypoidal choroidal vasculopathy: natural history. Am J Ophthalmol. 2002;133:639–48.
Sho K, Takahashi K, Yamada H, Wada M, Nagai Y, Otsuji T, et al. Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Arch Ophthalmol. 2003;121:1392–6.
Cho JH, Ryoo N-K, Cho KH, Park SJ, Park KH, Woo SJ. Incidence rate of massive submacular hemorrhage and its risk factors in polypoidal choroidal vasculopathy. Am J Ophthalmol. 2016;169:79–88.
Ohji M, Saito Y, Hayashi A, Lewis JM, Tano Y. Pneumatic displacement of subretinal hemorrhage without tissue plasminogen activator. Arch Ophthalmol. 1998;116:1326–32.
Hesse L, Schmidt J, Kroll P. Management of acute submacular hemorrhage using recombinant tissue plasminogen activator and gas. Graefes Arch Clin Exp Ophthalmol. 1999;237:273–7.
Haupert CL, McCuen BW, Jaffe GJ, Steuer ER, Cox TA, Toth CA, et al. Pars plana vitrectomy, subretinal injection of tissue plasminogen activator, and fluid-gas exchange for displacement of thick submacular hemorrhage in age-related macular degeneration. Am J Ophthalmol. 2001;131:208–15.
Fine HF, Iranmanesh R, Del Priore LV, Barile GR, Chang LK, Chang S, et al. Surgical outcomes after massive subretinal hemorrhage secondary to age-related macular degeneration. Retina. 2010;30:1588–94.
Kimura S, Morizane Y, Hosokawa M, Shiode Y, Kawata T, Doi S, et al. Submacular hemorrhage in polypoidal choroidal vasculopathy treated by vitrectomy and subretinal tissue plasminogen activator. Am J Ophthalmol. 2015;159:683–9.
van Zeeburg EJT, van Meurs JC. Literature review of recombinant tissue plasminogen activator used for recent-onset submacular hemorrhage displacement in age-related macular degeneration. Ophthalmologica. 2013;229:1–14.
Steel DHW, Sandhu SS. Submacular haemorrhages associated with neovascular age-related macular degeneration. Br J Ophthalmol. 2011;95:1051–7.
Toth CA, Morse LS, Hjelmeland LM, Landers MB. Fibrin directs early retinal damage after experimental subretinal hemorrhage. Arch Ophthalmol. 1991;109:723–9.
Benner JD, Morse LS, Toth CA, Landers MB, Hjelmeland LM. Evaluation of a commercial recombinant tissue-type plasminogen activator preparation in the subretinal space of the cat. Arch Ophthalmol. 1991;109:1731–6.
Hillenkamp J, Surguch V, Framme C, Gabel V-P, Sachs HG. Management of submacular hemorrhage with intravitreal versus subretinal injection of recombinant tissue plasminogen activator. Graefes Arch Clin Exp Ophthalmol. 2010;248:5–11.
Vujosevic S, Midena E, Pilotto E, Radin PP, Chiesa L, Cavarzeran F. Diabetic macular edema: correlation between microperimetry and optical coherence tomography findings. Invest Ophthalmol Vis Sci. 2006;47:3044–51.
Pranger F, Michels S, Geitzenauer W, Schmidt-Erfurth U, Simader C. Changes in retinal sensitivity in patients with neovascular age-related macular degeneration after systemic bevacizumab (Avastin) therapy. Retina. 2008;28:682–8.
Parravano M, Oddone F, Tedeschi M, Chiaravalloti A, Perillo L, Boccassini B, et al. Retinal functional changes measured by microperimetry in neovascular age-related macular degeneration treated with ranibizumab. Retina. 2010;30:1017–24.
Ozdemir H, Karacorlu M, Senturk F, Karacorlu SA, Uysal O. Microperimetric changes after intravitreal bevacizumab injection for exudative age-related macular degeneration. Acta Ophthalmol. 2010;90:71–5.
Crossland M, Jackson M-L, Seiple WH. Microperimetry: a review of fundus related perimetry. Optom Rep. 2012;2:11–5.
Cho HJ, Kim CG, Yoo SJ, Cho SW, Lee DW, Kim JW, et al. Retinal functional changes measured by microperimetry in neovascular age-related macular degeneration treated with ranibizumab. Am J Ophthalmol. 2013;155:118–26.
Imamura Y, Engelbert M, Iida T, Freund KB, Yannuzzi LA. Polypoidal choroidal vasculopathy: a review. Surv Ophthalmol. 2010;55:501–15.
Treatment of Age-Related Macular Degeneration With Photodynamic Therapy. (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials–TAP report. Arch Ophthalmol. 1999;117:1329–45.
Sato S, Hirooka K, Baba T, Tenkumo K, Nitta E, Shiraga F. Correlation between the ganglion cell inner plexiform layer thickness measured with Cirrus HD-OCT and macular visual field sensitivity measured with microperimetry. Invest Ophthalmol Vis Sci. 2012;54:3046–51.
Wu Z, Jung CJ, Ayton LN, Luu CD, Guymer RH. Test–retest repeatability of microperimetry at the border of deep scotomas. Invest Ophthalmol Vis Sci. 2015;56:2606–11.
Koh A, Lee WK, Chen L-J, Chen S-J, Hashad Y, Kim H, et al. EVEREST study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina. 2012;32:1453–64.
Scupola A, Coscas G, Soubrane G, Balestrazzi E. Natural history of macular subretinal hemorrhage in age-related macular degeneration. Ophthalmologica. 1999;213:97–102.
Okanouchi T, Toshima S, Kimura S, Morizane Y, Shiraga F. Novel technique for subretinal injection using local removal of the internal limiting membrane. Retina. 2016;36:1035–8.
Treumer F, Roider J, Hillenkamp J. Long-term outcome of subretinal coapplication of rtPA and bevacizumab followed by repeated intravitreal anti-VEGF injections for neovascular AMD with submacular haemorrhage. Br J Ophthalmol. 2012;96:708–13.
Kim JH, Kim JW, Lee TG, Lew YJ. Treatment outcomes in eyes with polypoidal choroidal vasculopathy with poor baseline visual acuity. J Ocul Pharmacol Ther. 2015;31:241–7.
Chang YS, Kim JH, Kim JW, Lee TG, Kim CG, Cho SW. Polypoidal choroidal vasculopathy in patients aged less than 50 years: characteristics and 6-month treatment outcome. Graefes Arch Clin Exp Ophthalmol. 2016;254:1–7.
Lin T-C, Hwang D-K, Lee F-L, Chen S-J. Visual prognosis of massive submacular hemorrhage in polypoidal choroidal vasculopathy with or without combination treatment. J Chin Med Assoc. 2016;79:159–65.
Lee SS, Ghosn C, Yu Z, Zacharias LC, Kao H, Lanni C, et al. Vitreous VEGF clearance is increased after vitrectomy. Invest Ophthalmol Vis Sci. 2010;51:2135–8.
Kakinoki M, Sawada O, Sawada T, Saishin Y, Kawamura H, Ohji M. Effect of vitrectomy on aqueous VEGF concentration and pharmacokinetics of bevacizumab in macaque monkeys. Invest Ophthalmol Vis Sci. 2012;53:5877–80.
Christoforidis JB, Williams MM, Wang J, Jiang A, Pratt C, Abdel-Rasoul M, et al. Anatomic and pharmacokinetic properties of intravitreal bevacizumab and ranibizumab after vitrectomy and lensectomy. Retina. 2013;33:946–52.
Mizutani T, Yasukawa T, Ito Y, Takase A, Hirano Y, Yoshida M, et al. Pneumatic displacement of submacular hemorrhage with or without tissue plasminogen activator. Graefes Arch Clin Exp Ophthalmol. 2011;249:1153–7.
Saito M, Kano M, Itagaki K, Oguchi Y, Sekiryu T. Retinal pigment epithelium tear after intravitreal aflibercept injection. Clin Ophthalmol. 2013;7:1287–9.
Ersoz MG, Karacorlu M, Arf S, Sayman Muslubas I, Hocaoglu M. Retinal pigment epithelium tears: classification, pathogenesis, predictors, and management. Surv Ophthalmol. 2017;62:493–505.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
S. Kimura, None; Y. Morizane, None; R. Matoba, None; M. Hosokawa, None; Y. Shiode, None; M. Hirano, None; S. Doi, None; S. Toshima, None; K. Takahashi, None; M. Hosogi, None; A. Fujiwara, None; F. Shiraga, None.
About this article
Cite this article
Kimura, S., Morizane, Y., Matoba, R. et al. Retinal sensitivity after displacement of submacular hemorrhage due to polypoidal choroidal vasculopathy: effectiveness and safety of subretinal tissue plasminogen activator. Jpn J Ophthalmol 61, 472–478 (2017). https://doi.org/10.1007/s10384-017-0530-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-017-0530-0