Summary
Background
Giant liver cysts causing compression symptoms require surgical therapy. Laparoscopy is nowadays considered the first-line approach and has been shown to be non-inferior to open surgery. Ancillary techniques and novel technologies may have the potential to reduce complications rates and improve long-term outcomes.
Methods
The management of a female patient with a giant and symptomatic liver cyst is reported, as is a literature search in PubMed and Scopus spanning the past two decades, with the aim of assessing current evidence regarding procedural details of laparoscopic deroofing.
Results
Wide laparoscopic deroofing of a 21-cm liver cyst arising from segment 6 was safely performed under indocyanine green fluorescence imaging using a combination of ultrasonic energy excision and stapling. A contemporary literature review showed that only 22 of the 35 publications included details of the surgical procedure. Ancillary techniques such as omentopexy, argon plasma coagulation, monopolar radiofrequency device ablation, and ethanol sclerotherapy were rarely used (10.8% of patients). Use of energy devices and/or linear staplers was reported in 22 (62.8%) studies. Indocyanine green fluorescence was reported in 4 (11.4%) studies.
Conclusion
The case report and the literature review show that wide laparoscopic deroofing of giant liver cysts is an effective and relatively simple procedure. Use of emerging technology such as indocyanine green fluorescence imaging can further enhance precision surgery and minimize complications and long-term recurrence rates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
-
Wide laparoscopic deroofing of simple liver cysts in favorable anatomical locations remains the procedure of choice.
-
Combination of ultrasonic dissection and stapling for excision of the cyst wall near the transition zone with liver parenchyma is effective, safe, and timesaving.
-
Near infra-red imaging with indocyanine green may enhance the safety of the procedure.
Introduction
Simple liver cysts (SLC), also known as biliary retention cysts, are benign masses filled with fluid that originate from malformation of the ductal plate during embryologic development, likely from micro-hamartomas or peribiliary glands isolated from the bile ducts. The reported prevalence in the general population is 3–18% [1, 2]. These cysts are generally asymptomatic, have no malignant potential, and only 5–10% of patients are thought to become symptomatic [3]. However, the cyst can increase in size with age, causing compression symptoms such as abdominal pain and distention, nausea, anorexia, early satiety, gastroesophageal reflux, weight loss, jaundice, dyspnea, and inferior vena cava syndrome. Patients can also present with a palpable mass, and/or complications such as intra-cystic bleeding and rupture [3].
In 1968, Lin et al. [4] first described the surgical technique of liver cyst fenestration through a laparotomic approach. With the advent of minimally invasive surgery, laparoscopy has become the standard of treatment and has proven feasible and safe even in patients with giant cysts, at least in anatomically favorable locations [5]. Herein, we describe a case of laparoscopic fenestration performed with ultrasonic dissection and stapling, assisted by indocyanine green (ICG) fluorescence. We also performed a literature review spanning the past two decades to summarize the current status of laparoscopic fenestration and the possible impact of technical details and technology on clinical outcomes.
Case report
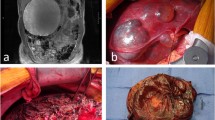
A 63-year-old woman with a body mass index of 24.3 kgm-2 was referred to our hospital for debilitating, worsening abdominal pain and tenderness that started about 6 months prior to admission. No concomitant gastrointestinal symptoms were reported. Her medical history included saphenectomy and carpal tunnel release. At physical examination, only epigastric tenderness was noticed. Abdominal ultrasonography showed a 21-cm hepatic cyst located in the right liver lobe and displacing the right hemidiaphragm and the right kidney. Gallbladder microlithiasis was also present. Magnetic resonance imaging confirmed a well-defined giant hepatic cyst arising from segment 6 with regular thickened walls, small calcifications, some intra-cystic septa, no nodules, and homogenous fluid filling on T1-weighted images; no enhancement after contrast injection was noted (Fig. 1).
A complete blood panel including tumor markers (alfa-fetoprotein, carcinoembryonic antigen [CEA], neurospecific enolase [NSE], carbohydrate antigen [CA] 15‑3, CA 19‑9, CA 125) and serological testing for hydatidosis resulted negative. The diagnosis of simple giant hepatic cyst was eventually confirmed, and the patient agreed to undergo laparoscopic fenestration. Upon pre-admission, she was classified as American Society of Anesthesiologists (ASA) score 1.
On the day of the procedure, about 4 h before induction of anesthesia, 2.5 mg of ICG (Verdye, 5 mg/ml injection, A.P.M. Srl, Trezzano sul Naviglio, Milan, Italy) was administered intravenously. The patient was placed in the supine position with arms extended and legs apart. Pneumoperitoneum was established by the Veress needle technique, and the abdomen was entered using a 12-mm optical trocar inserted into the left hypochondrium. Three additional operative trocars (one 12 mm and two 5 mm) were inserted. Peritoneal lavage for cytological analysis was performed. The cyst was punctured and approximately 2.4 L of serous fluid was suctioned and sent for cytological examination. Further opening of the cyst wall with a monopolar cautery allowed inspection of the inner surface of the cyst, and no visible nodules or bile leakage were noticed. Wide deroofing was then performed using a combination of harmonic scalpel (Ultracision, Ethicon Endosurgery, Norderstedt, Germany) and Tri-Staple EndoGIA (US Surgical, Norwalk, CT; Reload 45.0 mm and 60.0 mm). Indocyanine green fluorescence imaging helped to identify the liver edge close to the cyst wall and guided the choice of the surgical device. Given the significant thickness of the cyst wall near the liver parenchyma, we preferred to use the linear stapler. After the deroofing procedure had been completed, we confirmed that there was no bleeding from the staple lines and no ICG leakage from the inner surface of the cyst. Finally, simultaneous retrograde cholecystectomy was performed (Fig. 2). The resected cyst and the gallbladder were inserted into an extraction bag and removed from the left lateral port site. The procedure lasted 130 min, and the estimated blood loss was < 50 mL. The postoperative course was uneventful, and the patient was discharged home on postoperative day 3 in good general condition with a mean VAS score of 3. Histopathological examination confirmed a SLC composed by cuboidal epithelium and fibrous stromal tissue. Results of peritoneal cytology were negative. At 1‑year follow-up the patient is completely asymptomatic and abdominal ultrasound show no recurrence.
a Laparoscopic inspection of the cyst. b Laparoscopic suction of serous cystic fluid. c Resection of the thinner part of the cystic wall with harmonic scalpel. d Resection of the thicker cystic wall with an endoscopic linear stapler. e Retrograde cholecystectomy. f Final indocyanine green fluorescence imaging showing no evidence of bile leakage
Discussion
Simple liver cysts are often asymptomatic and have no malignant potential. However, symptoms from large cysts can arise and surgical therapy is warranted in these patients. Although there is no clear cut-off for defining a giant liver cyst, it has been reported that cysts > 10 cm in diameter are more likely to cause compression-related symptoms [6]. Cyst contents are usually serous, but proteinaceous material from previous intra-cystic hemorrhage may be present. Massive hemorrhage and/or spontaneous rupture of SLC has also been described and may require an emergency surgical procedure [3].
Treatment modalities for SLC range from percutaneous needle aspiration and sclerotherapy with ethanol injection to open or laparoscopic fenestration. Liver resection is usually performed in the setting of polycystic liver disease or when biliary cystoadenoma or carcinoma is suspected [1].
Laparoscopic deroofing, first described by Z’Graggen et al. [7] in 1991, has gradually become the surgical therapy of choice, especially for cysts located in the ventral and left lateral locations. Kisiel et al. [8] reported the long-term outcomes including symptom relief and quality of life in 48 patients operated in a single hepatobiliary unit between 2000 and 2012. There was one (2%) bile leak and no mortality. These patients were followed for a median of 62 (22–173) months and 60.4% were followed up for over 5 years. Of 46 patients who had initial symptom relief, 37 (80%) reported long-term symptomatic benefit and 2 underwent redo surgery with open fenestration and omentopexy. In a large meta-analysis including 62 studies with a total of 1314 patients, Bernts et al. [2] reported symptomatic relief in 90.2% and symptomatic recurrence in 9.6% of the patients, with a reintervention rate of 7.1%. The postoperative complication rate was 10.8% and the major complications rate 3.3%, with a procedure-related mortality of 1%.
A search of English-language case reports or case series published in PubMed and Scopus identified 35 publications, with a total of 148 patients treated by laparoscopic deroofing of large hepatic cysts. We restricted our search to January 2000 to December 2022. We included studies reporting on both conventional and single-port laparoscopy (Table 1).
Studies reporting procedures performed via open laparotomy or robotic surgery and/or associated with liver resections for polycystic liver disease (PLD) were excluded. Pediatric patients were also excluded. According to Murad et al. [44], we assigned a high, medium, or low quality score to each study according to clinical relevance, and reported surgical details and length of follow-up.
Based on the above criteria, one third of the studies (28.5%) were considered to be of high quality. Single-port laparoscopy was performed in 9 studies, for a total of 28 patients (18.9%). Simple liver cysts were more common in women, with a male to female ratio of 1:3.2. Patient age ranged between 47 and 88 years, with a mean age at diagnosis of 72 years. Hepatic cysts ranged in size from 5 to 28 cm and were slightly more frequent in the right (n = 42) than in the left liver segments (n = 37). The majority (78%) of patients had a single liver cyst. Seven studies reported a total of 12 patients with PLD [10, 12, 20, 26, 31, 38, 41]. Overall, 7 (4.7%) patients presented with acute abdomen and underwent emergency surgery for spontaneous cyst rupture, intra-cystic bleeding, or infection [11, 29, 30, 32, 41, 42]. Conversion to laparotomy was reported in only 1 patient (1/149, 0.6%) and was due to a challenging hepatic mobilization during excision of a single hepatic cyst located in segment 8 [10].
Surprisingly, only 22 publications included details of the surgical procedure and devices used to complete the fenestration. In a minority of patients (10.8%), laparoscopic deroofing was combined with omentopexy (n = 8), argon plasma coagulation (n = 4), or was preceded by ethanol sclerotherapy (n = 4). To date, there is no clear consensus in the literature regarding the real efficacy of these ancillary techniques in terms of the recurrence rate, and the evidence to support their use is limited. In addition, they are not free from complications such as cauterization of a major vascular structure or iatrogenic cholangitis due to the sclerosing agent [2].
Regarding the surgical device, the harmonic scalpel (UltracisionTM) was used in 13 centers and a vessel-sealing system (LigaSureTM) in 8. Both devices are considered effective and safe in liver surgery [45, 46].
On the other hand, we found only 3 publications reporting use of a stapling device during laparoscopic deroofing [13, 27, 35]. Stapling devices have gained popularity in laparoscopic liver surgery and are generally employed for major vessel division and less frequently for parenchymal transection [47, 48]. Some authors have used an endoscopic linear stapler to minimize any possibility of blood loss or bile leaks during excision of a thick cyst wall [27]. Interestingly, Umemura et al. [35] used a linear stapler to prevent bile leakage after injecting ICG through an endoscopic nasobiliary drainage. We used the harmonic scalpel to initially excise the cyst wall but then preferred to complete the deroofing with an endoGIA stapler due to the thickness of the cyst wall near the liver parenchyma.
For years, it has been known that intravenously administered ICG is excreted into the bile [49]. In this review, use of ICG was reported in only four studies, all published after 2016. In the case reported by Tanaka et al. [31], intravenous ICG allowed identification of an unexpected bile leakage, which could not be identified in the initial laparoscopic view. However, there is a lack of agreement on the timing of ICG administration. During laparoscopic cholecystectomy, ICG is administered between 0.5 h and 24 h before the procedure. In 3 patients undergoing laparoscopic deroofing [31, 36, 40], ICG was injected intravenously 1 h before surgery, after induction of anesthesia, and 24 h before surgery (followed by an intraoperative boost). We decided to administer ICG 4 h before induction of anesthesia, and there was no need for an intraoperative boost. Indocyanine green fluorescence helps to recognize the correct boundaries to liver parenchyma and to assess the real thickness of the cyst wall edges. Recent consensus guidelines on the use of ICG during open and laparoscopic hepatobiliary surgery state that intraoperative fluorescence imaging improves anatomical visualization, has the potential to guide surgical dissection, and is capable of augmenting safety, efficiency, and outcomes [49].
Finally, from our literature search, only 5 patients presented with a symptomatic recurrence during a follow-up ranging from 2 to 72 months from the index operation, and all required laparoscopic revision [10, 14, 25]. Limitations of this review are that some studies do not describe the extent of deroofing, a short follow-up may have underestimated recurrence rates, and postoperative imaging may have been undertaken only in patients with recurrent symptoms.
Conclusion
Our case report and literature review show that wide laparoscopic deroofing of SLC is an effective and relatively simple procedure. The concerns that fenestration via a laparoscopic approach may be inferior to open surgery are probably outdated. Versatility in the use of modern and emerging technology is recommended to minimize the incidence of perioperative complications and recurrences. It is reasonable and timesaving to use staplers as an adjunct to ultrasonic excision of thick cystic walls. Indocyanine green fluorescence with a single intravenous bolus prior to the laparoscopic procedure may further enhance the safety of the procedure.
References
Shimizu T, Yoshioka M, Kaneya Y, et al. Management of simple hepatic cyst. J Nippon Med Sch. 2022;89(1):2–8.
Bernts LHP, Echternach SG, Kievit W, et al. Clinical response after laparoscopic fenestration of symptomatic hepatic cysts : a systematic review and meta-analysis. Surg Endosc. 2019;33(3):691–704. https://doi.org/10.1007/s00464-018-6490-8.
Shah PC. Narrative review of laparoscopic management of hepatic cysts. Laparosc Surg. 2021;5:19. https://doi.org/10.21037/ls-20-36.
Lin T, Chen C, Wang S. Treatment of non-parasitic cystic disease of the liver : a new approach to therapy with polycystic liver. Ann Surg. 1968; https://doi.org/10.1097/00000658-196811000-00022.
Gomez A, Wisneski AD, Luu HY, et al. Contemporary management of hepatic cyst disease: techniques and outcomes at a tertiary hepatobiliary center. J Gastrointest Surg. 2021;25:77–84. https://doi.org/10.1007/s11605-020.
Sato T, Imai M, Hayashi K, et al. Giant hepatic cyst with septal structure: diagnosis and management. Int J Hepatol. 2013; https://doi.org/10.1155/2013/981975.
Z’graggen K, Metzger A, Klaiber C. Symptomatic simple cysts of the liver: treatment by laparoscopic surgery. Surg Endosc. 1991;5(4):224–5.
Kisiel A, Chb MB, Vass DG, et al. Long-term patient-reported outcomes after laparoscopic fenestration of symptomatic liver cysts. Surg Laparosc Endosc Percutan Tech. 2017;27:e80–e2.
Eriguchi N, Nagashima JUN, Uchida S, et al. Treatments of non-parasitic hepatic cysts. Kurume Med J. 2001;48:193–5.
Fiamingo P, Tedeschi U, Veroux M, et al. Laparoscopic treatment of simple hepatic cysts and polycystic liver disease. Surg Endosc. 2003;17:623–6. https://doi.org/10.1007/s00464-002-9088-z.
Cheung FKY, Lee KF, John W, et al. Emergency laparoscopic unroofing of a ruptured hepatic cyst. JSLS. 2005;9:497–9.
Antonini-Canterin F, De Biasio M, Baldessin F, et al. Re-expansion unilateral pulmonary oedema after surgical drainage of a giant hepatic cyst: a case report. J Cardiovasc Med. 2007;8:188–91.
Mekeel KL, Moss AA, Reddy KS, et al. Laparoscopic fenestration of giant hepatic cysts. Surg Laparosc Endosc Percutan Tech. 2008;18:511–3.
Donati M, Stavrou GA, Wellmann A, et al. Laparoscopic deroofing of hepatic cysts: the most effective treatment option. Clin Ter. 2010;161(4):345–8.
Sasaki K, Watanabe G, Matsuda M, et al. Original method of transumbilical single-incision laparoscopic deroofing for liver cyst. J Hepatobiliary Pancreat Sci. 2010;17:733–4. https://doi.org/10.1007/s00534-010-0279-z.
Mantke R, Wicht S. Single-port liver cyst fenestration combined with completely reusable instruments. Surg Laparosc Endosc Percutan Tech. 2010;20:e28–e30.
Kashiwagi H, Kumagai K, Nozue M. Single incision laparoscopic surgery for a life-threatening cyst of liver. Tokai J Exp Clin Med. 2011;36(1):13–6.
Fischer PE, Voeller GR. Laparoscopic management of a giant hepatic cyst with fibrin glue fixation of the omentum. Surg Laparosc Endosc Percutan Tech. 2011;21:e273–e4.
Lalezari S, Coates M, Sorour N. Rare disease A 77-year-old female with a giant hepatic cyst. BMJ Case Rep. 2012; https://doi.org/10.1136/bcr.05.2011.4218.
Dapri G, Barabino M, Carnevali P, et al. Single-access transumbilical laparoscopic unroofing of a giant hepatic cyst using reusable instruments. JSLS. 2012; https://doi.org/10.4293/108680812X13427982377300.
Imamura H, Kawashita Y, Koga N, et al. A large hepatic cyst with obstructive jaundice successfully treated with single-incision laparoscopic deroofing. Case Rep Gastroenterol. 2013;7(3):503–10.
Gocho T, Misawa T, Suzuki F, et al. Single-incision laparoscopic surgery for giant hepatic cyst. Asian J Endosc Surg. 2013;6(3):237–40. https://doi.org/10.1111/ases.12021.
Wu S, Li Y, Tian Y, et al. Single-incision laparoscopic surgery versus standard laparoscopic surgery for unroofing of hepatic cysts. JSLS. 2014; https://doi.org/10.4293/108680813X13753907291512.
Iimuro Y, Okada T, Kondo Y, et al. Analysis of unique liver volume restoration after laparoscopic fenestration of liver cysts. Asian J Endosc Surg. 2014;7:124–32.
Lee D, Cho JY, Han H, et al. Laparoscopic treatment of hepatic cysts located in the posterosuperior segments of the liver. Ann Surg Treat Res. 2014; https://doi.org/10.4174/astr.2014.86.5.23.
Riccio E, Santangelo M, Scotti A, et al. A strange epigastric pain. Clin Kidney J. 2014; https://doi.org/10.1093/ckj/sfu088.
Igami T, Tsuchiya T, Ebata T, et al. Application of a z-shaped umbilical incision and a saline-cooled radiofrequency device to single-incision laparoscopic surgery for a huge liver cyst: report of a case. Int Surg. 2015;100:1098–103. https://doi.org/10.9738/INTSURG-D-14-00198.1.
Willems K, Monsivais S, Vassaur H, et al. Single-incision laparoscopic management of a giant hepatic cyst. J Surg Case Rep. 2015;2015(7):rjv73.
Shimada S, Hara Y, Wada N, et al. Spontaneously ruptured hepatic cyst treated with laparoscopic deroofing and cystobiliary communication closure: a case report. Asian J Endosc Surg. 2016;9:208–10.
Imaoka Y, Ohira M, Kobayashi T, et al. Elective laparoscopic deroofing to treat the spontaneous rupture of a large simple liver cyst: a case report. Surg Case Rep. 2016;2(1):148. https://doi.org/10.1186/s40792-016-0275-x.
Tanaka M, Inoue Y, Mise Y, et al. Laparoscopic deroofing for polycystic liver disease using laparoscopic fusion indocyanine green fluorescence imaging. Surg Endosc. 2016;30(6):2620–3. https://doi.org/10.1007/s00464-015-4526-x.
Marques A, Camarneiro R, Silva R, et al. Laparoscopic deroofing of a ruptured hepatic cyst presenting as an acute abdomen. J Surg Case Rep. 2019;2019(2):rjy347. https://doi.org/10.1093/jscr/rjy347.
Carvalho GL, Góes GHB, Furtado RHM, et al. Resection of giant hepatic cyst by hybrid minilaparoscopy. J Minim Access Surg. 2018;15(2):167–9. https://doi.org/10.4103/jmas.JMAS_68_18.
Clancy C, Gibbons C, Ridgway PF. Surgical management of a giant hepatic cyst with suspicious radiological features. Ann R Coll Surg Engl. 2019;101(3):e76–e8. https://doi.org/10.1308/rcsann.2018.0206.
Umemura A, Nitta H, Suto T, et al. Indocyanine green fluorescence imaging via endoscopic nasal biliary drainage during laparoscopic deroofing of liver cysts. J Minim Access Surg. 2021;17(1):131–4. https://doi.org/10.4103/jmas.JMAS_26_20.
Tanioka N, Maeda H, Shimizu S, et al. Indocyanine green fluorescence-guided laparoscopic deroofing of a liver cyst: a case report. Asian J Endosc Surg. 2022;15(2):359–62. https://doi.org/10.1111/ases.12999.
Le TV, Truong T, Ngo TNM, et al. A case of hepatic cyst-induced internal jugular venous thrombosis. CASE (Phila). 2021;5(3):170–2. https://doi.org/10.1016/j.case.2021.02.007.
Brusic A, Oo J, Stella DL, et al. Giant haemorrhagic hepatic cyst with flame-like morphology in a patient with polycystic kidney and liver disease. Ultrasound. 2021;29(3):187–92. https://doi.org/10.1177/1742271X20987263.
Nakabayashi K, Murakami M, Hata S, et al. Giant hepatic cyst: a possible cause of inferior vena cava syndrome. Intern Med. 2021;60(13):2081–4. https://doi.org/10.2169/internalmedicine.6474-20.
Shimagaki T, Itoh S, Toshida K, et al. Prevention of bile duct injury using indocyanine green fluorescence in laparoscopic liver cyst fenestration for giant liver cyst: a case report. J Surg Case Rep. 2022;2022(10):479. https://doi.org/10.1093/jscr/rjac479.
Kenzaka T, Sato Y, Nishisaki H. Giant infected hepatic cyst causing exclusion pancreatitis: a case report. World J Clin Cases. 2022;10(7):2294–300. https://doi.org/10.12998/wjcc.v10.i7.2294.
Chogahara I, Oshita A, Nakahara H, et al. Spontaneous rupture of hemorrhagic hepatic cyst: two case reports. Surg Case Rep. 2022;8(1):30. https://doi.org/10.1186/s40792-022-01382-0.
Calabrese GV, Waku M, Mazzarulli F, et al. Laparoscopic deroofing in large hepatic cyst with alcohol sclerotherapy. Analysis of three cases. Ann Ital Chir. 2022;93:224–8.
Murad MH, Sultan S, Haffar S, et al. Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med. 2018;23(2):60–3. https://doi.org/10.1136/bmjebm-2017-110853.
Sugo H, Mikami Y, Matsumoto F, et al. Hepatic resection using the harmonic scalpel. Surg Today. 2000;30(10):959–62. https://doi.org/10.1007/s005950070055.
Lesurtel M, Belghiti J. Open hepatic parenchymal transection using ultrasonic dissection and bipolar coagulation. HPB (Oxford). 2008;10(4):265–70. https://doi.org/10.1080/13651820802167961.
Mehrabi A, Hoffmann K, Nagel AJ, et al. Technical aspects of stapled hepatectomy in liver surgery: how we do it. J Gastrointest Surg. 2019;23(6):1232–9. https://doi.org/10.1007/s11605-019-04159-3.
Yao DB, Wu SD. Application of stapling devices in liver surgery: current status and future prospects. World J Gastroenterol. 2016;22(31):7091–8. https://doi.org/10.3748/wjg.v22.i31.7091.
Wang X, Teh CSC, Ishizawa T, et al. Consensus guidelines for the use of fluorescence imaging in hepatobiliary surgery. Ann Surg. 2021;274(1):97–106. https://doi.org/10.1097/SLA.0000000000004718.
Funding
Open access funding provided by Università degli Studi di Milano within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Kersik, L. Galassi, G. Colombo and L. Bonavina declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants or on human tissue were in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. Internal review board approval: HSD 2023-011.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kersik, A., Galassi, L., Colombo, G. et al. Laparoscopic deroofing of simple liver cysts: do ancillary techniques, surgical devices, and indocyanine green improve outcomes?. Eur Surg 55, 100–106 (2023). https://doi.org/10.1007/s10353-023-00798-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-023-00798-8