Abstract
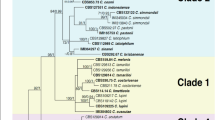
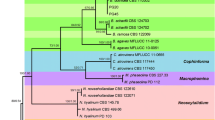
The members of the Colletotrichum gloeosporioides species complex (CGSC), the dominant pathogens of apple bitter rot in Nagano prefecture, Japan, were reidentified and the relationship between the species and fungicide sensitivity was revealed. Based on phylogenetic analysis of the ApMat locus with the neighbor-joining (NJ) method, isolates from apple contained three species of the CGSC; C. fructicola, C. aenigma, C. siamense, and three clades of the CGSC: Clade V, S and K. Colletotrichum fructicola and Clade S dominated in Nagano Prefecture. Isolates of C. siamense, C. aenigma and Clade V, S and K remained sensitive to benomyl and quinone outside inhibitor (QoI) fungicides, while C. fructicola often developed resistance to benomyl and QoI fungicides. These results suggest that the development of fungicide resistance differs among members of the CGSC.


Similar content being viewed by others
References
Akahira T, Hanaoka T (2013) Occurrence of trifloxystrobin resistant strains of Colletotrichum gloeosporioides (Glomerella cingulata), the causal fungus of apple bitter rot in Aomori Prefecture. Jpn J Phytopathol 79:197–198 (Abstract in Japanese)
Alaniz S, Hernández L, Mondino P (2015) Colletotrichum fructicola is the dominant and one of the most aggressive species causing bitter rot of apple in Uruguay. Trop Plant Pathol 40:265–274
Baroncelli R, Sreenivasaprasad S, Thon MR, Sukno SA (2014) First report of apple bitter rot caused by Colletotrichum godetiae in the United Kingdom. Plant Dis 98:1000
Braganca CAD, Damm U, Baroncelli R, Massola Jr NS, Crous PW (2016) Species of the Colletotrichum acutatum complex associated with anthracnose diseases of fruit in Brazil. Fungal Biol 120:547–561
Chethana CS, Chowdappa P, Biju CN, Praveena R, Sujatha AM (2016) Molecular and phenotypic characterization revealed six Colletotrichum species responsible for anthracnose disease of small cardamom in South India. Eur J Plant Pathol 146:465–481
Chung W, Ishii H, Nishimura K, Fukaya M, Yano K, Kajitani Y (2006) Fungicide sensitivity and phylogenetic relationship of anthracnose fungi isolated from various fruit crops in Japan. Plant Dis 90:506–512
Damm U, Cannon PF, Woudenberg JHC, Crous PW (2012) The Colletotrichum acutatum species complex. Stud Mycol 73:37–113
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evol Int J org Evol 39:783–791
Fungicide Resistance Action Committee (2014) Pathogen risk list 2014. Fungicide Resistance Action Committee, CropLife International, Brussells, Belgium. http://www.frac.info/docs/default-source/publications/pathogen-risk/pathogen-risk-list.pdf. Accessed 30 Apr 2017
Gan P, Nakata N, Suzuki T, Shirasu K (2017) Markers to differentiate species of anthracnose fungi identify Colletotrichum fructicola as the predominant virulent species in strawberry plants in Chiba Prefecture of Japan. J Gen Plant Pathol 83:14–22
Gisi U, Sierotzki H, Cook A, McCaffery A (2002) Mechanisms influencing the evolution of resistance to Qo inhibitor fungicides. Pest Manag Sci 58:859–867
Hirayama Y, Asano S, Watanabe K, Sakamoto Y, Ozaki M, Okayama K, Ohki ST, Tojo M (2016) Control of Colletotrichum fructicola on strawberry with a foliar spray of neutral electrolyzed water through an overhead irrigation system. J Gen Plant Pathol 82:186–189
Inada M, Ishii H, Chung W, Yamada T, Yamaguchi J, Furuta A (2008) Occurrence of strobilurin resistant strains of Colletotrichum gloeosporioides (Glomerella cingulata), the causal fungus of strawberry anthracnose. Jpn J Phytopathol 74:114–117 (Japanese with English summary)
Ishii H (2009) Corynespora cassiicola. In: Manual of sensitivity to fungicide of plant pathogens II. Japan Plant Protection Association, Tokyo, pp 69–71
Ishii H, Fraaije BA, Sugiyama T, Noguchi K, Nishimura K, Takeda T, Amano T, Hollomon DW (2001). Occurrence and molecular characterization of strobilurin resistance in cucumber powdery mildew and downy mildew. Phytopathology 91:1166–1171
Ishii H, Fountaine J, Chung WH, Kansako M, Nishimura K, Takahashi K, Oshima M (2009) Characterisation of QoI-resistant field isolates of Botrytis cinerea from citrus and strawberry. Pest Manag Sci 65:916–922
Jayawardena RS, Hyde KD, Damm U, Cai L, Liu M, Li XH, Zhang W, Zhao WS, Yan JY (2016) Notes on currently accepted species of Colletotrichum. Mycosphere 7:1192–1260
Kawai Y, Kondo K, Okazawa K, Miyazawa T (2000) Distribution of apple bitter rot caused by Colletotrichum acutatum in Nagano Prefecture (in Japanese). Annual Report of the Kanto-Tosan Plant Protection. Society 47:79–81
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kuck KH (2007) QoI fungicides: resistance mechanisms and its practical importance. In: Ohkawa H, Miyagawa H, Lee PW (eds) Pesticide chemistry; Crop protection, public health, environmental safety. Wiley-VCH, Weinheim, pp 275–283
Liu F, Weir B S, Damm U, Crous P W, Wang Y, Liu B, Wang M, Zhang M, Cai L (2015) Unravelling Colletotrichum species associated with Camellia: employing ApMat and GS loci to resolve species in the C. gloeosporioides complex. Persoonia 35:63–86
Liu F, Wang M, Damm U, Crous PW, Cai L (2016) Species boundaries in plant pathogenic fungi: a Colletotrichum case study. BMC Evol Biol 16:81
Munir M, Amsden B, Dixon E, Vaillancourt L, Ward Gauthier NA (2016) Characterization of Colletotrichum species causing bitter rot of apple in Kentucky orchards. Plant Dis 100:2194–2203
Peres NAR, Souza NL, Peever TL, Timmer LW (2004) Benomyl sensitivity of isolates of Colletotrichum acutatum and C. gloeosporioides from citrus. Plant Dis 88:125–130
Prihastuti H, Cai L, Chen H, McKenzie EHC, Hyde KD (2009) Characterization of Colletotrichum species associated with coffee berries in northern Thailand. Fungal Divers 39:89–109
Saitou N, Nei M (1987) The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sato T (1997) Characters and identification of a plurivorus anthracnose fungus, Colletotrichum acutatum (in Japanese with English summary). Proc Assoc Pl Protec Shikoku 32:1–19
Sato T, Moriwaki J (2013) Molecular re-identification of strains in NIAS Genebank belonging to phylogenetic groups A2 and A4 of the Colletotrichum acutatum species complex. Microbiol Cult Coll 29:13–23
Sato T, Uematsu S, Iijima Y, Koganezawa H (1998) Occurrence of apple bitter rot by grayish colony form of Colletotrichum acutatum in Japan and pathogenicity to apple fruits of C. acutatum and Glomerella cingulata isolated from other plants (in Japanese with English summary). Trans Mycol Soc Jpn 39:35–44
Sato T, Moriwaki J, Misawa T (2013) Molecular re-identification of strains of the Colletotrichum acutatum species complex deposited in the NIAS Genebank and morphological characteristics of its member species. JARQ 47:295–305
Sato T, Aoki T, Nemoto H (2016) Molecular re-identification of MAFF (NIAS Genebank) strains belonging to the Colletotrichum gloeosporioides species complex (abstract in Japanese). Microb Resour Syst 32:77–78
Sato T, Moriwaki J, Aoki T, Nemoto H (2017) Update of pathogen names of several anthracnose based on molecular re-identification of NIAS Genebank strains belonging to the Colletotrichum gloeosporioides species complex (abstract in Japanese). Jpn J Phytopathol 83:43–44
Sharma G, Kumar N, Weir BS, Hyde K, Shenoy BS (2013) The ApMat marker can resolve Colletotrichum species: a case study with Mangifera indica. Fungal Divers 61:117–138
Sharma G, Pinnaka AK, Shenoy BD (2015) Resolving the Colletotrichum siamense species complex using ApMat marker. Fungal Divers 71:247–264
Silva D, Várzea P, Cai L, Paulo O, Batista D (2012) Application of the Apn2/MAT locus to improve the systematics of the Colletotrichum gloeosporioides complex: an example from coffee (Coffea spp.) hosts. Mycologia 104:396–409
Tamura T, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30:2725–2729
Thompson J, Higgins D, Gibson T (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Velho A, Alaniz S, Casanova L, Mondino P, Stadnik M (2015) New insights into the characterization of Colletotrichum species associated with apple diseases in southern Brazil and Uruguay. Fungal Biol 119:229–244
Wang W, Fu D, Zhang R, Sun G (2015) Etiology of apple leaf spot caused by Colletotrichum spp. (in Chinese with English summary). Mycosystema 34:13–25
Weir BS, Johnston PR, Damm U (2012) The Colletotrichum gloeosporioides species complex. Stud Mycol 73:115–180
Wenneker K, Pham KTK, Lemmers MEC, de Boer FA, van der Lans AM, van Leeuwen PJ, Hollinger TC (2016) First report of Colletotrichum godetiae causing bitter rot on ‘Golden Delicious’ apples in the Netherlands. Plant Dis 100:218
Yokosawa S, Sato T, Kondo K (2015) Identification of the Colletotrichum gloeosporioides species complex members isolated from apple and grape in Nagano (abstract in Japanese). Jpn J Phytopathol 81:206
Acknowledgements
We thank Mr. Yasuhiko Iwanami and the Nagano Prefecture Agricultural Extension Center for help in material collections; Mr. Kazuya Minemura, Ms. Akiko Yokota and Ms. Mariko Katsuyama for technical assistance; and Mr. Shuichi Kato for helpful suggestions in this study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yokosawa, S., Eguchi, N., Kondo, Ki. et al. Phylogenetic relationship and fungicide sensitivity of members of the Colletotrichum gloeosporioides species complex from apple. J Gen Plant Pathol 83, 291–298 (2017). https://doi.org/10.1007/s10327-017-0732-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-017-0732-9