Abstract
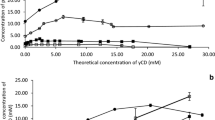
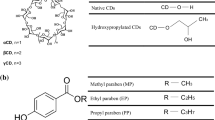
The drawback of some organic pesticides is their low water solubility. Cyclodextrins are used as agents for improving pesticides water solubility. However, the stabilization of the crystalline lattice can reduces to a large extent the solubility of cyclodextrins, especially β-cyclodextrin. Here, searching for the water soluble β-cyclodextrin derivatives, a set of new “host” compounds were prepared by the reaction of β-cyclodextrin with methyl epoxycinnamate. The reactants ratio was varied in to study the substitution degree of the obtained derivatives. The structure was determined by the 1H-NMR and FT IR spectra. The average degree of substitution was determined by integration of the corresponding NMR signals and by elemental analysis. The solubility of four selected pesticides: dimethoate, simazine, linuron and thiram, in water with and without addition of the new modified β-cyclodextrin was measured by ultraviolet spectrophotometry. Our results showed that the modified β-cyclodextrins significantly improve solubility of pesticides.



Similar content being viewed by others
References
Choi JK, Girek T, Shin DH, Lim ST (2002) Structural and physical characterisation of octenylsuccinyl β-cyclodextrin. Carbohyd Polym 49:289–296
Dailey O, Bland J, Trask-Morrell B (1993) Preparation and characterization of cyclodextrin complexes of the insecticides aldicarb and sulprofos. J Agric Food Chem 41:1767–1771
Delgado R, Virgilia A, Garcia-Anton JM, Parente A (1997) Reaction of β-cyclodextrin with N-2, 3-epoxypropylphtalimide. Preparation, characterisation and study of a new substituted cycloheptaamylose. J Inc Phen 28:205–212
Kile DE, Chiou CT (1989) Water solubility enhancements of DDT and trichlorobenzene by some surfactants below and above the critical micelle concentration. Environ Sci Technol 23:832–838
Lezcano M, Al-Soufi W, Novo M, Rodriguez-Nunez E, Vazquez J (2002) Complexation of several benzimidazole-type fungicides with α- and β-cyclodextrins. J Agric Food Chem 50:108–112
Liu L, Guo QX (2002) The driving forces in the inclusion complexation of cyclodextrins. J Inc Phen 42:1–14
Liu Y, Han BH, Li B, Zhang YM, Zhao P, Chen YT, Wada T, Inoue Y (1998) Molecular recognition study on supramolecular systems. Synthesis of modified cyclodextrin and their inclusion complexation thermodynamics with L-tryptophan and some naphthalene derivatives. J Org Chem 63:1444–1454
Luo YC, Zeng QR, Wu G, Luan ZK, Yang RB, Liao BH (2003) Effect of beta-cyclodextrin compounds on the solubilization of three selected pesticides and their toxicity with methyl-parathion to Rana tigrina tadpoles. Bull Environ Contam Toxicol 70:998–1005
Mamba BB, Krause RW, Malefetse TJ, Nxumalo EN (2007) Monofunctionalized cyclodextrin polymers for the removal of organic pollutants from water. Environ Chem Lett 5:79–84
Morillo E, Perez-Martinez J, Gines J (2001) Leaching of 2, 4-D from a soil in the presence of β-cyclodextrin: laboratory columns experiments. Chemosphere 44:1065–1069
Nagai T, Ueda H (1996) Aspects of drug formulation with cyclodextrins. In: Comprehensive supramolecular chemistry, Vol. 3: Cyclodextrins. Elsevier Science, Oxford, pp 441–450
Stutz H, Malissa H (1999) Determination of regularly distributed plant protectants in raw and drinking waters, using a multiresidue method with cyclodextrin-modified micellar electrokinetic chromatography. J AOAC Inter 82:1510–1522
Szejtli J (1982) Cyclodextrins and their inclusion complexes. Akadémiai Kiadó, Budapest
Szejtli J (1985) Cyclodextrins in pesticides. Starch 37:382–386
Vogel A (1978) Textbook of practical organic chemistry, 4th edn. Longman Group Ltd, London, pp 308, 921
Wang XJ, Brusseau M (1993) Solubilization of some low-polarity organic compounds by hydroxypropyl-β-cyclodextrin. Environ Sci Technol 27:2821–2825
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petrović, G., Stojanović, G. & Palić, R. Modified β-cyclodextrins as prospective agents for improving water solubility of organic pesticides. Environ Chem Lett 9, 423–429 (2011). https://doi.org/10.1007/s10311-010-0296-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-010-0296-9