Abstract
Purpose
Droxidopa has been approved for the treatment of neurogenic orthostatic hypotension (NOH) under the US Food and Drug Administration accelerated approval program, which warrants confirmatory evidence on long-term efficacy of droxidopa. Hereby, we synthesize evidence from published randomized controlled trials (RCTs) about the safety and efficacy of droxidopa for patients with neurogenic orthostatic hypotension.
Methods
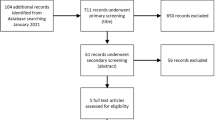
A computer literature search of PubMed, Scopus, Web of Science, and Cochrane Central was conducted using relevant keywords. Records were screened for eligible studies and data were extracted and synthesized using Review Manager version 5.3 for Windows. Subgroup analysis and sensitivity analysis were conducted to investigate long-term durability of droxidopa against placebo.
Results
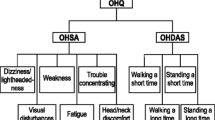
Four RCTs with a total of 485 patients (droxidopa, n = 246; placebo, n = 239) were eligible for the final analysis. The mean difference (MD) of change in the main outcomes from baseline to endpoint favored droxidopa than placebo [Orthostatic Hypotension Questionnaire (OHQ) MD −0.61, P = 0.004; dizziness/lightheadedness score MD −0.83, P = 0.008; and standing systolic blood pressure (SBP) MD 4.09, P = 0.03]. The efficacy of droxidopa decreased gradually after 2 weeks, and its statistical significance was lost after 8 weeks (OHQ score MD −0.18, P = 0.61; dizziness/lightheadedness score MD −0.71, P = 0.11; and standing SBP MD 2.96, P = 0.29). None of the adverse events were significantly higher in the case of droxidopa compared to placebo.
Conclusion
Droxidopa is a safe and effective drug for the short-term management of NOH symptoms. However, current evidence is insufficient to confirm the efficacy of droxidopa for long-term use. Therefore, further studies with increased sample size are needed.






Similar content being viewed by others
References
Kaufmann H (1996) Consensus statement on the definition of orthostatic hypotension, pure autonomic failure and multiple system atrophy. Clin Auton Res 6:125–126
Freeman R, Wieling W, Axelrod FB et al (2011) Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res 21:69–72
Freeman R (2008) Clinical practice. Neurogenic orthostatic hypotension. N Engl J Med 358:615–624. doi:10.1056/NEJMcp074189
Isaacson SH, Skettini J (2014) Neurogenic orthostatic hypotension in Parkinson’s disease: evaluation, management, and emerging role of droxidopa. Vasc Health Risk Manag 10:169
Horowitz DR, Kaufmann H (2001) Autoregulatory cerebral vasodilation occurs during orthostatic hypotension in patients with primary autonomic failure. Clin Auton Res 11:363–367. doi:10.1007/BF02292768
Maule S, Papotti G, Naso D et al (2007) Orthostatic hypotension: evaluation and treatment. Cardiovasc Hematol Disord Drug Targets 7:63–70
Ziegler MG, Lake CR, Kopin IJ (1977) The sympathetic-nervous-system defect in primary orthostatic hypotension. N Engl J Med 296:293–297. doi:10.1056/NEJM197702102960601
Goldstein DS, Sharabi Y (2009) Neurogenic orthostatic hypotension: a pathophysiological approach. Circulation 119:139–146
Ooi WL, Hossain M, Lipsitz LA (2000) The association between orthostatic hypotension and recurrent falls in nursing home residents. Am J Med 108:106–111. doi:10.1016/S0002-9343(99)00425-8
Low PA, Singer W (2008) Management of neurogenic orthostatic hypotension: an update. Lancet Neurol 7:451–458
Mathias CJ, Kimber JR (1999) Postural hypotension: causes, clinical features, investigation, and management. Annu Rev Med 50:317–336
Kaufmann H, Brannan T, Krakoff L et al (1988) Treatment of orthostatic hypotension due to autonomic failure with a peripheral alpha-adrenergic agonist (midodrine). Neurology 38:951–956
McTavish D, Goa KL (1989) Midodrine. Drugs 38:757–777
Jankovic J, Gilden JL, Hiner BC et al (1993) Neurogenic orthostatic hypotension: a double-blind, placebo-controlled study with midodrine. Am J Med 95:38–48. doi:10.1016/0002-9343(93)90230-M
Low PA, Gilden JL, Freeman R et al (1997) Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA 277:1046–1051. doi:10.1001/jama.277.13.1046
Wright R, Kaufmann H, Perera R et al (1998) A double-blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology 51:120–124. doi:10.1212/WNL.51.1.120
Biaggioni I, Robertson D (1987) Endogenous restoration of noradrenaline by precursor therapy in dopamine-beta-hydroxylase deficiency. Lancet 2:1170–1172. doi:10.1016/S0140-6736(87)91317-1
Freeman R, Landsberg L, Young J (1999) The treatment of neurogenic orthostatic hypotension with 3,4-dl-threo-dihydroxyphenylserine: a randomized, placebo-controlled, crossover trial. Neurology 53:2151–2157. doi:10.1212/WNL.53.9.2151
Mathias CJ, Senard JM, Braune S et al (2001) L-threo-dihydroxyphenylserine (l-threo-DOPS; droxidopa) in the management of neurogenic orthostatic hypotension: a multi-national, multi-center, dose-ranging study in multiple system atrophy and pure autonomic failure. Clin Auton Res 11:235–242. doi:10.1007/BF02298955
Kaufmann H, Saadia D, Voustianiouk A et al (2003) Norepinephrine precursor therapy in neurogenic orthostatic hypotension. Circulation 108:724–728. doi:10.1161/01.CIR.0000083721.49847.D7
Kaufmann H, Freeman R, Biaggioni I et al (2014) Droxidopa for neurogenic orthostatic hypotension: a randomized, placebo-controlled, phase 3 trial. Neurology 83:328–335. doi:10.1212/WNL.0000000000000615
Hauser RA, Isaacson S, Lisk JP et al (2014) Droxidopa for the short-term treatment of symptomatic neurogenic orthostatic hypotension in Parkinson’s disease (nOH306B). Neurology 00:1–9. doi:10.1002/mds.26086
Biaggioni I, Freeman R, Mathias CJ et al (2015) Randomized withdrawal study of patients with symptomatic neurogenic orthostatic hypotension responsive to droxidopa. Hypertension 65:101–107. doi:10.1161/HYPERTENSIONAHA.114.04035
Hauser RA, Hewitt LA, Isaacson S (2014) Droxidopa in patients with neurogenic orthostatic hypotension associated with Parkinson’s disease (NOH306A). J Parkinsons Dis 4:57–65. doi:10.3233/JPD-130259
Kaufmann H, Malamut R, Norcliffe-Kaufmann L et al (2012) The Orthostatic Hypotension Questionnaire (OHQ): validation of a novel symptom assessment scale. Clin Auton Res 22:79–90. doi:10.1007/s10286-011-0146-2
Altman DGG, Bland JMM (2005) Standard deviations and standard errors. BMJ 331:903. doi:10.1136/bmj.331.7521.903
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634. doi:10.1136/bmj.316.7129.469
Terrin N, Schmid CH, Lau J, Olkin I (2003) Adjusting for publication bias in the presence of heterogeneity. Stat Med 22:2113–2126. doi:10.1002/sim.1461
Izcovich A, González Malla C, Manzotti M et al (2014) Midodrine for orthostatic hypotension and recurrent reflex syncope: a systematic review. Neurology 83:1170–1177. doi:10.1212/WNL.0000000000000815
Acknowledgments
We would like to thank our colleagues in Medical Research Society (MRS, Kasr Al ainy School of Medicine, Cairo University, Egypt) and Medical Research Group of Egypt (MRGE; the nationwide student-led medical research team of Egypt) for their encouragement. We are grateful to the two anonymous reviewers for their positive inputs to the content of this manuscript. We would like also to thank Manuscriptedit Company for providing professional language editing service.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors confirm no financial or personal relationship with a third party whose interests could be positively or negatively influenced by the article’s content.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Elgebaly, A., Abdelazeim, B., Mattar, O. et al. Meta-analysis of the safety and efficacy of droxidopa for neurogenic orthostatic hypotension. Clin Auton Res 26, 171–180 (2016). https://doi.org/10.1007/s10286-016-0349-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-016-0349-7