Abstract
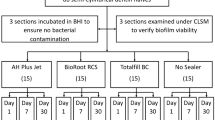
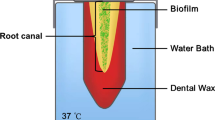
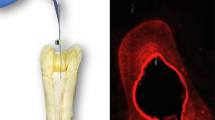
This study aimed to compare the antibacterial effects of different agitation devices on single- and multispecies biofilms in dentin canals using confocal laser scanning microscopy (CLSM). Dentin blocks were prepared from human root dentin. Enterococcus faecalis and multiple species were introduced into the dentinal tubules via centrifugation and incubation. Two infected dentin samples were placed at 8 and 16 mm in a customized model. Samples were randomly divided into eight groups according to the agitation device used: syringe needle irrigation, EndoActivator, passive ultrasonic irrigation (PUI), and EDDY, at 2.5% or 6% NaOCl concentrations. The samples were stained and observed using CLSM. Statistical analysis was performed using an independent sample t test and analysis of variance. Linear models were used to assess the joint impact of the experimental groups on the proportion of biofilms killed. No significant differences were observed between the killing rates of the single- and multispecies biofilms. Both concentrations of NaOCl significantly increased the percentage of dead bacteria compared with the control. Biofilms in dentin tubules was more effectively killed when NaOCl was agitated; however, the difference between PUI and EDDY was not significant. Significantly more bacteria were killed in dentin blocks placed at 8 mm than at 16 mm (p < 0.05). In conclusion, EDDY was as effective as PUI when combined with NaOCl. However, the apical portion, which had a low antimicrobial efficiency, remains a concern. Mechanical instrumentation is incapable of completely eradicating bacteria, and additional research is required to improve the efficacy of root canal disinfection.




Similar content being viewed by others
References
Nair PNR. On the causes of persistent apical periodontitis: a review. Int Endod J. 2006;39:249–81.
Kakehashi S, Stanley HR, Fitzgerald RJ. The effects of surgical exposures of dental pulps in germfree and conventional laboratory rats. J South Calif Dent Assoc. 1966;34:449–51.
Siren EK, Haapasalo MPP, Waltimo TMT, Orstavik D. In vitro antibacterial effect of calcium hydroxide combined with chlorhexidine or iodine potassium iodide on enterococcus faecalis. Eur J Oral Sci. 2004;112:326–31.
Haapasalo M, Shen Y, Qian W, Gao Y. Irrigation in endodontics. Dent Clin North Am. 2010;54:291–312.
Chavez de Paz LE, Dahlen G, Molander A, et al. Bacteria recovered from teeth with apical periodontitis after antimicrobial endodontic treatment. Int Endod J. 2003;36:500–8.
Siqueira JF, Rôças IN, Ricucci D. Biofilms in endodontic infection. Endod Topics. 2010;22:33–49.
Stojicic S, Shen Y, Haapasalo M. Effect of the source of biofilm bacteria, level of biofilm maturation, and type of disinfecting agent on the susceptibility of biofilm bacteria to antibacterial agents. J Endod. 2013;39:473–7.
Peters OA, Peters CI, Schönenberger K, et al. ProTaper rotary root canal preparation: effects of canal anatomy on final shape analysed by micro CT. Int Endod J. 2003;36:86–92.
Rosenfeld EF, James GA, Burch BS. Vital pulp tissue response to sodium hypochlorite. J Endod. 1978;4:140–6.
Du T, Wang Z, Shen Y, et al. Combined antibacterial effect of sodium hypochlorite and root canal sealers against enterococcus faecalis biofilms in dentin canals. J Endod. 2015;41:1294–8.
Ma J, Wang Z, Shen Y, et al. A new noninvasive model to study the effectiveness of dentin disinfection by using confocal laser scanning microscopy. J Endod. 2011;37:1380–5.
Yang Y, Shen Y, Wang Z, et al. Evaluation of the susceptibility of multispecies biofilms in dentinal tubules to disinfecting solutions. J Endod. 2016;42:1246–50.
Liu H, Li H, Zhang L, et al. In vitro evaluation of the antibacterial effect of four root canal sealers on dental biofilms. Clin Oral Investig. 2022. https://doi.org/10.1007/s00784-022-04399-9.
Gu L, Kim JR, Ling J, et al. Review of contemporary irrigant agitation techniques and devices. J Endod. 2009;35:791–804.
Ruddle CJ. Endodontic disinfection: tsunami irrigation. Saudi Endod J. 2015;5:1–12.
Caron G. Cleaning efficiency of the apical millimeters of curved canals using three different modalities of irrigant activation: an SEM study. Paris: Paris VII University; 2007.
Neuhaus KW, Liebi M, Stauffacher S, et al. Antibacterial efficacy of a new sonic irrigation device for root canal disinfection. J Endod. 2016;42:1799–803.
Ahmad M, Pitt Ford TR, Crum LA. Ultrasonic debridement of root canals: acoustic streaming and its possible role. J Endod. 1987;13:490–9.
Peciuliene V, Balciuniene I, Eriksen HM, et al. Isolation of Enterococcus faecalis in previously root-filled canals in a Lithuanian population. J Endod. 2000;26:593–5.
Conde AJ, Estevez R, Loroño G, Valencia de Pablo Ó, Rossi-Fedele G, Cisneros R. Effect of sonic and ultrasonic activation on organic tissue dissolution from simulated grooves in root canals using sodium hypochlorite and EDTA. Int Endod J. 2017;50:976–82.
Donnermeyer D, Wyrsch H, Bürklein S, Schäfer E. Removal of calcium hydroxide from artificial grooves in straight root canals: sonic activation using EDDY versus passive ultrasonic irrigation and XPendo Finisher. J Endod. 2019;45:322–6.
Plotino G, Colangeli M, Özyürek T, et al. Evaluation of smear layer and debris removal by stepwise intraoperative activation (SIA) of sodium hypochlorite. Clin Oral Investig. 2021;25:237–45.
Zeng C, Everett J, Sidow S, et al. In vitro evaluation of efficacy of two endodontic sonic-powered irrigant agitation systems in killing single-species intracanal biofilms. J Dent. 2021;115: 103859. https://doi.org/10.1016/j.jdent.2021.103859.
Matsuo T, Shirakami T, Ozaki K, et al. An immunohistological study of the localization of bacteria invading root pulpal walls of teeth with periapical lesions. J Endod. 2003;29:194–200.
Haapasalo M, Ørstavik D. In vitro infection and disinfection of dentinal tubules. J Dent Res. 1987;66:1375–9.
Siqueira JF Jr, Rôças IN, Lopes HP. Patterns of microbial colonization in primary root canal infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;93:174–8.
Qian Z, Sagers RD, Pitt WG. Investigation of the mechanism of the bioacoustic effect. J Biomed Mater Res. 1999;44:198–205.
Shen Y, Stojicic S, Qian W, Olsen I, Haapasalo M. The synergistic antimicrobial effect by mechanical agitation and two chlorhexidine preparations on biofilm bacteria. J Endod. 2010;36:100–4.
Swimberghe RCD, De Clercq A, De Moor RJG, et al. Efficacy of sonically, ultrasonically and laser-activated irrigation in removing a biofilm-mimicking hydrogel from an isthmus model. Int Endod J. 2019;52:515–23.
Wang Z, Shen Y, Haapasalo M. Dynamics of dissolution, killing, and inhibition of dental plaque biofilm. Front Microbiol. 2020;11:964. https://doi.org/10.3389/fmicb.2020.00964.
Wang Z, Shen Y, Ma J, et al. The effect of detergents on the antibacterial activity of disinfecting solutions in dentin. J Endod. 2012;38:948–53.
Wang Z, Shen Y, Haapasalo M. Effect of smear layer against disinfection protocols on Enterococcus faecalis–infected dentin. J Endod. 2013;39:1395–400.
Acknowledgements
This study was supported by the Canadian Academy of Endodontics and the authors deny any conflicts of interest related to this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Al-Zuhair, H., Su, Z., Liu, H. et al. Antimicrobial effects of agitational irrigation on single- and multispecies biofilms in dentin canals. Odontology 111, 49–56 (2023). https://doi.org/10.1007/s10266-022-00719-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-022-00719-3