Abstract
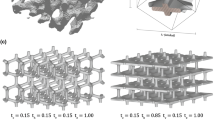
Bone tissue mechanical properties and trabecular microarchitecture are the main factors that determine the biomechanical properties of cancellous bone. Artificial cancellous microstructures, typically described by a reduced number of geometrical parameters, can be designed to obtain a mechanical behavior mimicking that of natural bone. In this work, we assess the ability of the parameterized microstructure introduced by Kowalczyk (Comput Methods Biomech Biomed Eng 9:135–147, 2006. doi:10.1080/10255840600751473) to mimic the elastic response of cancellous bone. Artificial microstructures are compared with actual bone samples in terms of elasticity matrices and their symmetry classes. The capability of the parameterized microstructure to combine the dominant isotropic, hexagonal, tetragonal and orthorhombic symmetry classes in the proportions present in the cancellous bone is shown. Based on this finding, two optimization approaches are devised to find the geometrical parameters of the artificial microstructure that better mimics the elastic response of a target natural bone specimen: a Sequential Quadratic Programming algorithm that minimizes the norm of the difference between the elasticity matrices, and a Pattern Search algorithm that minimizes the difference between the symmetry class decompositions. The pattern search approach is found to produce the best results. The performance of the method is demonstrated via analyses for 146 bone samples.










Similar content being viewed by others
References
Be’ery-Lipperman M, Gefen A (2005) Contribution of muscular weakness to osteoporosis: computational and animal models. Clin Biomech 20:984–997. doi:10.1016/j.clinbiomech.2005.05.018
Bose S, Vahabzadeh S, Bandyopadhyay A (2013) Bone tissue engineering using 3D printing. Mater Today 16:496–504. doi:10.1016/j.mattod.2013.11.017
Brennan O, Kennedy OD, Lee TC et al (2009) Biomechanical properties across trabeculae from the proximal femur of normal and ovariectomised sheep. J Biomech 42:498–503. doi:10.1016/j.jbiomech.2008.11.032
Browaeys JT, Chevrot S (2004) Decomposition of the elastic tensor and geophysical applications. Geophys J Int 159:667–678. doi:10.1111/j.1365-246X.2004.02415.x
Carretta R, Lorenzetti S, Müller R (2013) Towards patient-specific material modeling of trabecular bone post-yield behavior. Int J Numer Method Biomed Eng 29:250–272. doi:10.1002/cnm.2516
Colabella L, Ibarra Pino AA, Ballarre J et al (2017) Calculation of cancellous bone elastic properties with the polarization-based FFT iterative scheme. Int J Numer Method Biomed Eng. doi:10.1002/cnm.2879
Cowin SC (2001) Bone mechanics handbook, 2nd edn. CRC Press, Boca Raton
Cowin SC, Mehrabadi M (1987) On the identification of material symmetry for anisotropic elastic materials. Q J Mech Appl Math 40:451–476. doi:10.1093/qjmam/40.4.451
Dagan D, Be’ery M, Gefen A (2004) Single-trabecula building block for large-scale finite element models of cancellous bone. Med Biol Eng Comput 42:549–556. doi:10.1007/BF02350998
Doube M, Klosowski MM, Arganda-Carreras I et al (2010) BoneJ: free and extensible bone image analysis in ImageJ. Bone 47:1076–1079. doi:10.1016/j.bone.2010.08.023
Fritsch A, Hellmich C (2007) “Universal” microstructural patterns in cortical and trabecular, extracellular and extravascular bone materials: micromechanics-based prediction of anisotropic elasticity. J Theor Biol 244:597–620. doi:10.1016/j.jtbi.2006.09.013
Gill PE, Murray W, Saunders MA, Wright MH (1984) Procedures for optimization problems with a mixture of bounds and general linear constraints. ACM Trans Math Softw 10:282–298. doi:10.1145/1271.1276
Gill PE, Murray W, Wright MH (1991) Numerical linear algebra and optimization. Addison-Wesley Pub. Co, Redwood City
Goda I, Ganghoffer JF (2015a) Identification of couple-stress moduli of vertebral trabecular bone based on the 3D internal architectures. J Mech Behav Biomed Mater 51:99–118. doi:10.1016/j.jmbbm.2015.06.036
Goda I, Ganghoffer JF (2015b) 3D plastic collapse and brittle fracture surface models of trabecular bone from asymptotic homogenization method. Int J Eng Sci 87:58–82. doi:10.1016/j.ijengsci.2014.10.007
Goda I, Assidi M, Ganghoffer JF (2014) A 3D elastic micropolar model of vertebral trabecular bone from lattice homogenization of the bone microstructure. Biomech Model Mechanobiol 13:53–83. doi:10.1007/s10237-013-0486-z
Gross T, Pahr DH, Zysset PK (2013) Morphology–elasticity relationships using decreasing fabric information of human trabecular bone from three major anatomical locations. Biomech Model Mechanobiol 12:793–800. doi:10.1007/s10237-012-0443-2
Helgason B, Perilli E, Schileo E et al (2008) Mathematical relationships between bone density and mechanical properties: a literature review. Clin Biomech 23:135–146. doi:10.1016/j.clinbiomech.2007.08.024
Hollister SJ (2005) Porous scaffold design for tissue engineering. Nat Mater 4:518–524. doi:10.1038/nmat1421
Kabel J, Odgaard A, van Rietbergen B, Huiskes R (1999a) Connectivity and the elastic properties of cancellous bone. Bone 24:115–120. doi:10.1016/S8756-3282(98)00164-1
Kabel J, van Rietbergen B, Odgaard A, Huiskes R (1999b) Constitutive relationships of fabric, density, and elastic properties in cancellous bone architecture. Bone 25:481–486. doi:10.1016/S8756-3282(99)00190-8
Keaveny TM, Guo XE, Wachtel EF et al (1994) Trabecular bone exhibits fully linear elastic behavior and yields at low strains. J Biomech. doi:10.1016/0021-9290(94)90053-1
Keaveny TM, Morgan EF, Niebur GL, Yeh OC (2001) Biomechanics of trabecular bone. Annu Rev Biomed Eng 3:307–333. doi:10.1146/annurev.bioeng.3.1.307
Kowalczyk P (2006) Orthotropic properties of cancellous bone modelled as parameterized cellular material. Comput Methods Biomech Biomed Eng 9:135–147. doi:10.1080/10255840600751473
Kowalczyk P (2010) Simulation of orthotropic microstructure remodelling of cancellous bone. J Biomech 43:563–569. doi:10.1016/j.jbiomech.2009.09.045
Maquer G, Musy SN, Wandel J et al (2015) Bone volume fraction and fabric anisotropy are better determinants of trabecular bone stiffness than other morphological variables. J Bone Miner Res 30:1000–1008. doi:10.1002/jbmr.2437
Oftadeh R, Perez-Viloria M, Villa-Camacho JC et al (2015) Biomechanics and mechanobiology of trabecular bone: a review. J Biomech Eng 137:10802. doi:10.1115/1.4029176
Oliver WC, Pharr GM (1992) An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J Mater Res 7:1564–1583
Parkinson IH, Fazzalari NL (2013) Characterisation of trabecular bone structure. In: Silva MJ (ed) Skeletal aging and osteoporosis: biomechanics and mechanobiology. Springer, Berlin, pp 31–51
Sansalone V, Naili S, Bousson V et al (2010) Determination of the heterogeneous anisotropic elastic properties of human femoral bone: from nanoscopic to organ scale. J Biomech 43:1857–1863. doi:10.1016/j.jbiomech.2010.03.034
Sansalone V, Bousson V, Naili S, Bergot C (2012) Anatomical distribution of the degree of mineralization of bone tissue in human femoral neck: impact on biomechanical properties. Bone 50:876–884. doi:10.1016/j.bone.2011.12.020
Stauber M, Müller R (2006a) Age-related changes in trabecular bone microstructures: global and local morphometry. Osteoporos Int 17:616–626. doi:10.1007/s00198-005-0025-6
Stauber M, Müller R (2006b) Volumetric spatial decomposition of trabecular bone into rods and plates—a new method for local bone morphometry. Bone 38:475–484. doi:10.1016/j.bone.2005.09.019
van Rietbergen B, Ito K (2015) A survey of micro-finite element analysis for clinical assessment of bone strength: the first decade. J Biomech 48:832–841. doi:10.1016/j.jbiomech.2014.12.024
van Rietbergen B, Müller R, Ulrich D et al (1999) Tissue stresses and strain in trabeculae of a canine proximal femur can be quantified from computer reconstructions. J Biomech 32:165–173. doi:10.1016/S0021-9290(98)00150-X
Walker AM, Wookey J (2012) MSAT—a new toolkit for the analysis of elastic and seismic anisotropy. Comput Geosci 49:81–90. doi:10.1016/j.cageo.2012.05.031
Wang X, Xu S, Zhou S et al (2016) Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: a review. Biomaterials 83:127–141. doi:10.1016/j.biomaterials.2016.01.012
Yang G, Kabel J, Van Rietbergen B et al (1998) The anisotropic Hooke’s law for cancellous bone and wood. J Elast 53:125–146. doi:10.1023/A:1007575322693
Acknowledgements
This work has been supported by projects PIRSES-GA2009_246977 “Numerical Simulation in Technical Sciences” of the Marie Curie Actions FP7-PEOPLE-2009-IRSES of the European Union and by the PICS project “Modeling and Simulation in Multidisciplinary Engineering” MoSiMe funded in the framework of the CAFCI call by CONICET (Argentina) and CNRS (France). This project has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (Grant agreement No. 682001, project ERC Consolidator Grant 2015 BoneImplant).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Appendices
Appendix
1.1 Human samples
Figure 11 depicts the residual \(\mathcal {R}_{1}\) and the symmetry class errors (18) of the SQP optimizations as functions of BV/TV. Figure 11a shows that \(\mathcal {R}_{1}\) and its dispersion diminish sharply with BV/TV, from \(0.5\lesssim \mathcal {R}_{1}\lesssim 3\) for \(\hbox {BV}/\hbox {TV}<7.5\% \) to \(0.1\lesssim \mathcal {R}_{1}\lesssim 0.6\) for \(\hbox {BV}/\hbox {TV}>32\% \). The relaxation of the \(\hbox {BV}/\hbox {TV}_\mathrm{tol}\) constrain does not result in significant improvements in \(\mathcal {R}_{1}\) (results not reported here for analyses performed for \(\hbox {BV}/\hbox {TV}_\mathrm{tol}=10\% \) exhibit the same behavior). Results in Fig. 11b allow to observe that \(c_\mathrm{iso}\) and \(c_\mathrm{hex}\) are systematically over and underestimated, respectively; their mean errors are \(\overline{e_{c_\mathrm{iso}}}=0.20\) and \(\overline{e_{c_\mathrm{hex}}}=-0.74\). Errors tend to decrease with BV/TV, the only exception is that of the tetragonal symmetry, which can attain values over the range \(-5<e_{c_\mathrm{tet}}<12\). Mean relative errors for the tetragonal and the orthorhombic symmetry classes are \(\overline{e_{c_\mathrm{tet}}}=1.74\) and \(\overline{e_{c_\mathrm{ort}}}=0.18\), respectively.
The PS optimization resulted in a mean value for the residual \(\overline{\mathcal {R}_{2}}=0.06\) with standard deviation \(\hbox {SD}_{\mathcal {R}_{2}}=0.09\). Like for the SQP approach, the relaxation of the \(\hbox {BV}/\hbox {TV}_\mathrm{tol}\) constrain did not result in significant improvements for \(\mathcal {R}_{2}\). In addition to the error for the elasticity matrices shown in Figs. 9. Figure 12 presents the errors (18) for the symmetry classes. Figure 12 allows to observe that mean errors for the symmetry classes are much lower than those of the SQP approach in Fig. 11b: \(\overline{e_{c_\mathrm{iso}}}=0.06\), \(\overline{e_{c_\mathrm{hex}}}=-0.13\), \(\overline{e_{c_\mathrm{tet}}}=-0.06\) and \(\overline{e_{c_\mathrm{ort}}}=-0.02\); standard deviations are \((\hbox {SD}_{e_\mathrm{iso}}=0.11)\), \((\hbox {SD}_{e_\mathrm{hex}}=0.27)\), \((\hbox {SD}_{e_\mathrm{tet}}=1.67)\) and \((\hbox {SD}_{e_\mathrm {ort}}=0.18)\). Like for the SQP optimization, \(e_{c_\mathrm{tet}}\) presents the largest dispersion and there are tendencies to overestimate \(c_\mathrm{iso}\) and to underestimate \(c_\mathrm{hex}\). The high dispersion of \(e_{c_\mathrm{tet}}\) is consequence of the small relative contribution of the tetragonal symmetry to the elasticity matrix, which is always \(c_\mathrm{tet}<0.05\), see Table 3 and Fig. 1.
Bovine Samples
Target elastic matrices of the bovine femoral samples
The PS optimization of each sample was run ten times using different seeds. For each case, the best two outcomes (these are, the solutions that achieve the lowest values of the objective function) were compared to assess the repeatability of the solutions. It was found that for Samples #2 and #4, the residuals of the best two solutions are almost coincident (they differ less than 1%), while the resultant values for the geometrical parameters coincide within 2%. For samples #1 and #3, the lowest two residuals differ in around 12% and the geometrical parameters have discrepancies of up to 80%. It is interesting to note that the parameter \(t_{e}\), the one which governs microstructure orthotropy, showed a remarkable repeatability, it presented discrepancies within 0.2% for the analyses of Samples #1, #2, #3 and #4. In contrast, for Sample #5, residuals of the best two solutions differ in nearly 90% and the geometrical parameters up to 70%; the best performance is for \(t_{e}\), which presents a discrepancy discrepancy of 13%. The behavior for \(t_{e}\) could be explored as a mean for the refinement of the optimization procedure.
Finally, the resultant elastic matrices for the mimetic parameterized microstructures are:
Rights and permissions
About this article
Cite this article
Colabella, L., Cisilino, A.P., Häiat, G. et al. Mimetization of the elastic properties of cancellous bone via a parameterized cellular material. Biomech Model Mechanobiol 16, 1485–1502 (2017). https://doi.org/10.1007/s10237-017-0901-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-017-0901-y