Abstract
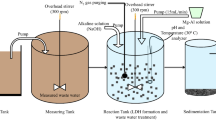
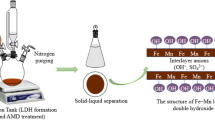
The fate of heavy metals in the environment is significantly influenced by their adsorption onto clay minerals. Herein, we investigate the mechanism of MnO4− and Cd2+ removal by nitrate-intercalated Mg–Al-layered double hydroxides (LDHs) in aqueous solution and optimize the corresponding conditions, showing that these adsorbents can effectively remove Zn, Mn, and Cd from mine wastewater and revealing that adsorbent structure is retained after wastewater treatment. The removal of MnO4− ions from aqueous solution is demonstrated to proceed via their exchange for NO3− ions intercalated within LDHs and is well described by the Freundlich adsorption model, following pseudo-second-order kinetics and featuring chemisorption as the rate-determining step. In contrast, Cd2+ is shown to be removed via the formation of Cd5(OH)8(NO3)2·2H2O (at LDH2) and Cd(NO3)2·Cd(OH)2 (at LDH1).








Similar content being viewed by others
References
Alloway BJ (1995) Heavy metals in soils. Blackie, Glasgow
Bailey SE, Olin TJ, Bricka RM, Adrian DD (1999) A review of potentially low-cost sorbents for heavy metals. Water Res 33:2469–2479
Park M, Choi CL, Seo YJ, Yeo SK, Choi J, Komarneni S, Lee JH (2007) Reaction of Cu2+ and Pb2+ with Mg/Al layered double hydroxide. Appl Clay Sci 37:143–148
Xiao B, Thomas KM (2004) Competitive adsorption of aqueous metal ions on an oxidized nanoporous activated carbon. Langmuir 20:4566–4578
Juang RS, Shao HJ (2002) Effect of pH on competitive adsorption of Cu(II), Ni(II), and Zn(II) from water onto chitosan beads. Adsorption 8:71–78
Trivedi P, Axe L, Dyer J (2001) Adsorption of metal ions onto goethite: single-adsorbate and competitive systems. Colloids Surf A Physicochem Eng Asp 191:107–121
Monzón JD, Pereyra AM, Gonzalez MR, Zerbino RL, Basaldella EI (2018) Mechanical properties and microstructure of cement mortars incorporating Cd-zeolitized fly ash. J Mat Cycles Waste Manag 20(4):2130–2139
Goswamee RL, Sengupta P, Bhattacharyya KG, Dutta DK (1998) Adsorption of Cr(VI) in layered double hydroxides. Appl Clay Sci 13:21–34
You Y, Vance GF, Zhao H (2001) Selenium adsorption on Mg–Al and Zn–Al layered double hydroxides. Appl Clay Sci 20:13–25
Goh KH, Lim TT, Dong Z (2009) Enhanced arsenic removal by hydrothermally treated nanocrystalline Mg/Al layered double hydroxide with nitrate intercalation. Environ Sci Technol 43:2537–2543
Grover K, Komarneni S, Katsuki H (2009) Uptake of arsenite by synthetic layered double hydroxides. Water Res 43:3884–3890
Liu R, Frost RL, Martens WN (2009) Absorption of the selenite anion from aqueous solutions by thermally activated layered double hydroxide. Water Res 43:1323–1329
Wang SL, Liu CH, Wang MK, Chuang YH, Chiang PN (2009) Arsenate adsorption by Mg/Al–NO3 layered double hydroxides with varying the Mg/Al ratio. Appl Clay Sci 43:79–85
Kameda T, Kondo E, Yoshioka T (2014) Preparation of Mg–Al layered double hydroxide doped with Fe2+ and its application to Cr(VI) removal. Sep Purif Technol 122:12–16
Kameda T, Nakamura M, Yoshioka T (2012) Removal of antimonite ions from an aqueous solution by anion exchange with magnesium-aluminum layered double hydroxide and the formation of a brandholzite-like structure. J Environ Sci Health Part A Environ Sci 47:1146–1151
Rahman MT, Kameda T, Kumagai S, Yoshioka T (2017) Adsorption isotherms and kinetics of arsenic removal from aqueous solution by Mg–Al Layered Double Hydroxide Intercalated with Nitrate Ions. React Kinet Mech Catal 120:703–714
Cavani F, Trifiro F, Vaccari A (1991) Hydrotalcite-type anionic clays: preparation, properties and applications. Catal Today 11:173–301
Kwon T, Tsigdinos GA, Pinnavaia TJ (1988) Pillaring of layered double hydroxides (LDHs) by polyoxometalate anions. J Am Chem Soc 110(11):3653
Kameda T, Takeuchi H, Yoshioka T (2010) Kinetics of uptake of Cu2+ and Cd2+ by Mg–Al layered double hydroxides intercalated with citrate, malate, and tartrate. Coll Surf A 355:172–177
Kameda T, Saito S, Umetsu Y (2005) Mg–Al layered double hydroxide intercalated with ethylene-diaminetetraacetate anion: synthesis and application to the uptake of heavy metal ions from an aqueous solution. Sep Purif Technol 47:20–26
Shan R, Yan L, Yang K, Hao Y, Du B (2015) Adsorption of Cd(II) by Mg–Al–CO3 and magnetic Fe3O4/Mg–Al–CO3 layered double hydroxides: kinetic, isothermal, thermodynamic and mechanistic studies. J Hazard Mater 299:42–49
Oliver SRJ (2009) Cationic inorganic materials for anionic pollutant trapping and catalysis. Chem Soc Rev 38:1868–1881
Rahman MT, Kameda T, Kumagai S, Yoshioka T (2018) Effectiveness of Mg–Al Layered Double Hydroxide for heavy metal removal from mine wastewater and sludge volume reduction. Int J Environ Sci Technol 15:263–272
Komarneni S, Kozai N, Roy R (1998) Novel function for anionic clays: selective transition metal cation uptake by diadochy. J Mat Chem 8:1329–1331
Liang X, Zang Y, Xu Y, Tan X, Hou W, Wang L, Sun Y (2013) Sorption of metal cations on layered double hydroxides. Coll Surf A 433:122–131
Bagherifam S, Komarneni S, Lakzian A, Fotovat A, Khorasani R, Huang W, Ma J, Wang Y (2014) Evaluation of Zn–Al–SO4 layered double hydroxide for the removal of arsenite and arsenate from a simulated soil solution: isotherms and kinetics. Appl Clay Sci 95:119–125
Freundlich H (1926) Adsorption. J Phys Chem 7:57–64
Abou-Mesalam M (2004) Applications of inorganic ion exchangers: II-adsorption of some heavy metal ions from their aqueous waste solution using synthetic iron (III) titanate. Adsorption 10:87–92
Lagergren S (1898) About the theory of so-called adsoption of soluble substances. K. Sven. Vetenskapsakad. Handl Band 24:1–39
Ho YS, Mckay G (1998) Sorption of dye from aqueous solution by peat. Chem Eng J 70:115–124
Bénard P, Louër M, Louër D (1991) Solving the crystal structure of cd5(oh)8(no3)2·2h2o from powder diffraction data. A comparison with single crystal data. Powder Diffr 6(1):10–15
JCPDS card 40-1460
JCPDS card 23-103
Ye J, Cheng B, Wageh S, Al-Ghamdi AA, Yu J (2016) Flexible Mg–Al layered double hydroxide supported Pt on Al foil for use in room-temperature catalytic decomposition of formaldehyde. RSC Adv 6:34280–34287
Zhang DE, Pan XD, Zhu H, Li SZ, Xu GY, Zhang XB, Ying AL, Tong ZW (2008) A simple method to synthesize cadmium hydroxide nanobelts. Nanoscale Res Lett 3:284–288
Chubar N, Gerda V, Megantari O, Micˇušik M, Omastova M, Heister K, Man P, Fraissard J (2013) Applications versus properties of Mg–Al layered double hydroxides provided by their syntheses methods: alkoxide and alkoxide-free sol–gel syntheses and hydrothermal precipitation. Chem Eng J 234:284–299
Richetta M, Digiamberardino L, Mattoccia A, Medaglia PG, Montanari R, Pizzoferrato R, Scarpellini D, Varone A, Kaciulis S, Mezzi A, Soltania P, Orsinid A (2016) Surface spectroscopy and structural analysis of nanostructured multifunctional (Zn, Al) layered double hydroxides. Surf Interface Anal 48:514–518
Acknowledgements
The authors acknowledge financial support from the Japan Oil, Gas and Metals National Corporation (JOGMEC).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rahman, M.T., Kameda, T., Miura, T. et al. Removal of Mn and Cd contained in mine wastewater by Mg–Al-layered double hydroxides. J Mater Cycles Waste Manag 21, 1232–1241 (2019). https://doi.org/10.1007/s10163-019-00875-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-019-00875-9