Abstract
Introduction
This study was to evaluate the surgical and survival effects of neoadjuvant chemotherapy (NAC) followed by radical hysterectomy (RH) for cervical cancer with stages IB2 to IIB of FIGO 2009 staging.
Methods
From February 2, 2001 to November 11, 2015, 428 patients received NAC followed by RH in a tertiary hospital, in which all the major procedures were performed by one surgeon. Surgical and survival outcomes were evaluated between the NAC and primary RH groups.
Results
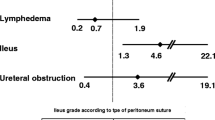
A total of 279 (65.2%) patients received NAC, and the overall clinical and complete pathological response rates were 65.9% and 10.8%, respectively. Compared with primary RH patients, NAC patients had more advanced stages, higher recurrence rate, longer median duration of RH, and more median estimated blood loss. After adjusted with baseline risk factors, no significant differences in progression-free or overall survival were observed between the NAC and primary RH groups. However, the responders to NAC had better survival outcomes.
Conclusions
There were no surgical or survival benefits of NAC for patients with cervical cancer of stages IB2 to IIB except for the responders to NAC.


Similar content being viewed by others
Abbreviations
- LACC:
-
Locally advanced cervical cancer
- NAC:
-
Neoadjuvant chemotherapy
- OS:
-
Overall survival
- PALN:
-
Para-aortic lymph nodes
- PF:
-
Fluorouracil and cisplatin
- PFS:
-
Progression-free survival
- PLN:
-
Pelvic lymph node
- RH:
-
Radical hysterectomy
- TC:
-
Paclitaxel and carboplatin
- TP:
-
Paclitaxel and cisplatin
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30. https://doi.org/10.3322/caac.21332
Chen W, Zheng R, Baade PD et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132. https://doi.org/10.3322/caac.21338
deSouza NM, Soutter WP, Rustin G et al (2004) Use of neoadjuvant chemotherapy prior to radical hysterectomy in cervical cancer: monitoring tumour shrinkage and molecular profile on magnetic resonance and assessment of 3-year outcome. Br J Cancer 90:2326–2331. https://doi.org/10.1038/sj.bjc.6601870
Panici PB, Bellati F, Plotti F et al (2008) Neoadjuvant chemotherapy followed by radical surgery in patients affected by vaginal carcinoma. Gynecol Oncol 111:307–311. https://doi.org/10.1016/j.ygyno.2008.07.005
Katsumata N, Yoshikawa H, Kobayashi H et al (2013) Phase III randomised controlled trial of neoadjuvant chemotherapy plus radical surgery vs radical surgery alone for stages IB2, IIA2, and IIB cervical cancer: a Japan Clinical Oncology Group trial (JCOG 0102). Br J Cancer 108:1957–1963. https://doi.org/10.1038/bjc.2013.179
Yang Z, Chen D, Zhang J et al (2016) The efficacy and safety of neoadjuvant chemotherapy in the treatment of locally advanced cervical cancer: a randomized multicenter study. Gynecol Oncol 141:231–239. https://doi.org/10.1016/j.ygyno.2015.06.027
Kim HS, Sardi JE, Katsumata N et al (2013) Efficacy of neoadjuvant chemotherapy in patients with FIGO stage IB1 to IIA cervical cancer: an international collaborative meta-analysis. Eur J Surg Oncol 39:115–124. https://doi.org/10.1016/j.ejso.2012.09.003
Chemoradiotherapy for Cervical Cancer Meta-Analysis Collaboration (2010) Reducing uncertainties about the effects of chemoradiotherapy for cervical cancer: individual patient data meta-analysis. Cochrane Database Syst Rev 26:CD008285. https://doi.org/10.1002/14651858.cd008285
Minig L, Patrono MG, Romero N et al (2014) Different strategies of treatment for uterine cervical carcinoma stage IB2–IIB. World J Clin Oncol 5:86–92. https://doi.org/10.5306/wjco.v5.i2.86
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Cervical cancer. Version 2.2019—October 12, 2018. https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf. Accessed 15 Dec 2018
Nama V, Angelopoulos G, Twigg J et al (2018) Type II or type III radical hysterectomy compared to chemoradiotherapy as a primary intervention for stage IB2 cervical cancer. Cochrane Database Syst Rev 10:CD011478. https://doi.org/10.1002/14651858.CD011478.pub2
Gupta S, Maheshwari A, Parab P et al (2018) Neoadjuvant chemotherapy followed by radical surgery versus concomitant chemotherapy and radiotherapy in patients with stage IB2, IIA, or IIB squamous cervical cancer: a randomized controlled trial. J Clin Oncol 36:1548–1555. https://doi.org/10.1200/JCO.2017.75.9985
Cibula D, Abu-Rustum NR, Benedetti-Panici P et al (2011) New classification system of radical hysterectomy: emphasis on a three-dimensional anatomic template for parametrial resection. Gynecol Oncol 122:264–268. https://doi.org/10.1016/j.ygyno.2011.04.029
Querleu D, Morrow CP (2008) Classification of radical hysterectomy. Lancet Oncol 9:297–303. https://doi.org/10.1016/s1470-2045(08)70074-3
Li L, Ma S, Tan X et al (2019) The urodynamics and survival outcomes of different methods of dissecting the inferior hypogastric plexus in laparoscopic nerve-sparing radical hysterectomy of type C: a randomized controlled study. Ann Surg Oncol 26:1560. https://doi.org/10.1245/s10434-019-07228-8
Pecorelli S, Zigliani L, Odicino F (2009) Revised FIGO staging for carcinoma of the cervix. Int J Gynecol Obstet 105:107–108. https://doi.org/10.1016/j.ijgo.2009.02.009
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Testa AC, Moro F, Pasciuto T et al (2018) PRospective Imaging of CErvical cancer and neoadjuvant treatment (PRICE) study: role of ultrasound to assess residual tumor in locally advanced cervical cancer patients undergoing chemoradiation and radical surgery. Ultrasound Obstet Gynecol 52:110–118. https://doi.org/10.1002/uog.18953
Obermair A, Gebski V, Frumovitz M et al (2008) A phase III randomized clinical trial comparing laparoscopic or robotic radical hysterectomy with abdominal radical hysterectomy in patients with early stage cervical cancer. J Minim Invasive Gynecol 15:584–588. https://doi.org/10.1016/j.jmig.2008.06.013
Common Terminology Criteria for Adverse Events (CTCAE) v4.03. National Cancer Institute. https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm#ctc_50. Accessed 15 Dec 2018
Shimada M, Nagao S, Fujiwara K et al (2016) Neoadjuvant chemotherapy with docetaxel and carboplatin followed by radical hysterectomy for stage IB2, IIA2, and IIB patients with non-squamous cell carcinoma of the uterine cervix. Int J Clin Oncol 21:1128–1135. https://doi.org/10.1007/s10147-016-1010-0
Gadducci A, Landoni F, Cosio S et al (2018) Neoadjuvant platinum-based chemotherapy followed by radical hysterectomy for stage Ib2–IIb adenocarcinoma of the uterine cervix—an Italian Multicenter Retrospective Study. Anticancer Res 38:3627–3634. https://doi.org/10.21873/anticanres.12637
Tsubamoto H, Maeda H, Kanazawa R et al (2013) Phase II trial on neoadjuvant intravenous and trans-uterine arterial chemotherapy for locally advanced bulky cervical adenocarcinoma. Gynecol Oncol 129:129–134. https://doi.org/10.1016/j.ygyno.2013.01.007
Junker P, Puppe J, Thangarajah F et al (2018) Neoadjuvant therapy of cervical carcinoma with the angiogenesis inhibitor bevacizumab: a single-centre analysis. Geburtshilfe Frauenheilkd 78:768–774. https://doi.org/10.1055/a-0641-5588
Rydzewska L, Tierney J, Vale CL et al (2012) Neoadjuvant chemotherapy plus surgery versus surgery for cervical cancer. Cochrane Database Syst Rev 12:CD007406. https://doi.org/10.1002/14651858.CD007406.pub3
Mossa B, Mossa S, Corosu L et al (2010) Follow-up in a long-term randomized trial with neoadjuvant chemotherapy for squamous cell cervical carcinoma. Eur J Gynaecol Oncol 31:497–503
Kokka F, Bryant A, Brockbank E et al (2015) Hysterectomy with radiotherapy or chemotherapy or both for women with locally advanced cervical cancer. Cochrane Database Syst Rev 4:CD010260. https://doi.org/10.1002/14651858.CD010260.pub2
Wen H, Wu X, Li Z et al (2012) A prospective randomized controlled study on multiple neoadjuvant treatments for patients with stage IB2 to IIA cervical cancer. Int J Gynecol Cancer 22:296–302. https://doi.org/10.1097/IGC.0b013e31823610a1
Shoji T, Takatori E, Furutake Y et al (2016) Phase II clinical study of neoadjuvant chemotherapy with CDDP/CPT-11 regimen in combination with radical hysterectomy for cervical cancer with a bulky mass. Int J Clin Oncol 21:1120–1127. https://doi.org/10.1007/s10147-016-1008-7
Mahmoud O, Einstein MH (2018) Which patients with cervical squamous cell carcinoma might benefit from neoadjuvant chemotherapy? J Clin Oncol 36:1543–1547. https://doi.org/10.1200/JCO.2017.77.3416
Marchetti C, De Felice F, Di Pinto A et al (2018) Survival nomograms after curative neoadjuvant chemotherapy and radical surgery for stage IB2–IIIB cervical cancer. Cancer Res Treat 50:768–776. https://doi.org/10.4143/crt.2017.141
Pergialiotis V, Rodolakis A, Christakis D et al (2013) Laparoscopically assisted vaginal radical hysterectomy: systematic review of the literature. J Minim Invasive Gynecol 20:745–753. https://doi.org/10.1016/j.jmig.2013.04.021
Bentivegna E, Maulard A, Pautier P et al (2016) Fertility results and pregnancy outcomes after conservative treatment of cervical cancer: a systematic review of the literature. Fertil Steril 106:1195–1211. https://doi.org/10.1016/j.fertnstert.2016.06.032
Plante M (2015) Bulky early-stage cervical cancer (2–4 cm lesions): upfront radical trachelectomy or neoadjuvant chemotherapy followed by fertility-preserving surgery: which is the best option? Int J Gynecol Cancer 25:722–728. https://doi.org/10.1097/IGC.0000000000000410
Li J, Wang LJ, Zhang BZ et al (2011) Neoadjuvant chemotherapy with paclitaxel plus platinum for invasive cervical cancer in pregnancy: two case report and literature review. Arch Gynecol Obstet 284:779–783. https://doi.org/10.1007/s00404-011-1943-5
Fruscio R, Villa A, Chiari S et al (2012) Delivery delay with neoadjuvant chemotherapy for cervical cancer patients during pregnancy: a series of nine cases and literature review. Gynecol Oncol 126:192–197. https://doi.org/10.1016/j.ygyno.2012.04.027
Tang J, Tang Y, Yang J et al (2012) Chemoradiation and adjuvant chemotherapy in advanced cervical adenocarcinoma. Gynecol Oncol 125:297–302. https://doi.org/10.1016/j.ygyno.2012.01.033
Lan M, Chen C, Huang Y et al (2017) Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in nasopharyngeal carcinoma patients with cervical nodal necrosis. Sci Rep 7:42624. https://doi.org/10.1038/srep42624
Ferrandina G, Palluzzi E, Gallotta V et al (2018) Neo-adjuvant platinum-based chemotherapy followed by chemoradiation and radical surgery in locally advanced cervical cancer (Lacc) patients: a phase II study. Eur J Surg Oncol 44:1062–1068. https://doi.org/10.1016/j.ejso.2018.04.019
Ferrandina G, Corrado G, Vitrano G et al (2018) Dose-dense paclitaxel/carboplatin as neo-adjuvant chemotherapy followed by radical surgery in locally advanced cervical cancer: a prospective phase II study. Cancer Chemother Pharmacol 83:431. https://doi.org/10.1007/s00280-018-3742-1
Ramirez PT, Frumovitz M, Pareja R et al (2018) Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med 379:1895–1904. https://doi.org/10.1056/NEJMoa1806395
Melamed A, Margul DJ, Chen L et al (2018) Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med 379:1905–1914. https://doi.org/10.1056/NEJMoa1804923
Funding
This work was supported by the Chinese Academy of Medical Sciences Initiative for Innovative Medicine (no. CAMS-2017-I2M-1-002). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
LL: assistant surgeon, protocol/project development, data management, data analysis, and manuscript drafting; MW: major surgeon and manuscript editing; SM, XT, SZ: assistant surgeons and data management.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Li, L., Wu, M., Ma, S. et al. Neoadjuvant chemotherapy followed by radical hysterectomy for stage IB2-to-IIB cervical cancer: a retrospective cohort study. Int J Clin Oncol 24, 1440–1448 (2019). https://doi.org/10.1007/s10147-019-01510-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-019-01510-1