Abstract
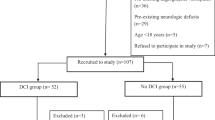
High mobility group box 1 protein (HMGB1) is a prototypical damage associated particle and acts as a key player in aseptic inflammation. HMGB1 appears critical for the crosstalk of a prothrombotic and proinflammatory state that is implicated in mediating and exacerbating ischemic brain injury. The role of HMGB1 in aneurysmal subarachnoid hemorrhage (aSAH) remains to be elucidated. A prospective, single blinded observational study was designed to investigate the role of HMGB1 in aSAH. Serial serum HMGB1 level quantification on admission day 0, 4, 8, and 12 was performed. Primary outcome measures were delayed cerebral ischemia (DCI — new infarction on CT) and poor functional outcome (90-day modified Rankin Scale 4–6). The role of HMGB1 levels for DCI, functional outcome and radiological vasospasm prediction was analyzed. Collectively, 83 aSAH patients were enrolled. Five patients died within 48 h. In 29/78 patients (37.2%), DCI was identified. In multivariable analysis, radiological vasospasm and admission HMGB1 were independent predictors for DCI. Younger age and higher white blood cell count, but not insult burden (World Federation of Neurosurgical Societies scale, modified Fisher scale, intraparenchymal or intraventricular hematoma existence) correlated with admission HMGB1 levels. Serial HMGB1 levels did not differ between patients with or without DCI, poor functional outcome or radiological vasospasm development. Admission serum HMGB1 does not reflect initial insult burden but serves as an independent biomarker predictive of DCI. Further studies are warranted to disentangle the role of HMGB1 surrounding the sequelae of aSAH.




Similar content being viewed by others
Data availability
Data are available upon reasonable request, initiation requires contacting the corresponding author.
Code availability
N/A
Abbreviations
- aSAH :
-
Aneurysmal subarachnoid hemorrhage
- AUC :
-
Area under the curve
- CVS :
-
Clinical vasospasm
- CRP :
-
C-reactive protein
- CTA :
-
CT-angiography
- DAM :
-
Damage associated particle
- DIND :
-
Delayed ischemic neurological deficit
- DSA :
-
Digital subtraction angiography
- DCI :
-
Delayed cerebral ischemia
- EBI :
-
Early brain injury
- HIMOBASH :
-
High mobility group box 1 in aneurysmal subarachnoid hemorrhage
- HMGB1 :
-
High mobility group box 1 protein
- IPH :
-
Intraparenchymal hematoma
- IVH :
-
Intraventricular hemorrhage
- RAGE :
-
Receptor for advanced glycation endproduct
- mRS :
-
Modified Rankin Scale
- NETs :
-
Neutrophil extracellular traps
- RVS :
-
Radiological vasospasm
- TLR :
-
Toll-like-receptor
- TCD :
-
Transcranial doppler studies
- WBC :
-
White blood cell
References
Aida Y, Kamide T, Ishii H, Kitao Y, Uchiyama N, Nakada M, Hori O (2019) Soluble receptor for advanced glycation end products as a biomarker of symptomatic vasospasm in subarachnoid hemorrhage. J Neurosurg 1–9. https://doi.org/10.3171/2019.8.JNS191269
Al-Mufti F, Misiolek KA, Roh D, Alawi A, Bauerschmidt A, Park S, Agarwal S, Meyers PM, Connolly ES, Claassen J, Schmidt JM (2019) White blood cell count improves prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurgery 84:397–403. https://doi.org/10.1093/neuros/nyy045
Brown RJ, Kumar A, Dhar R, Sampson TR, Diringer MN (2013) The relationship between delayed infarcts and angiographic vasospasm after aneurysmal subarachnoid hemorrhage. Neurosurgery 72:702–707; discussion 707–708. doi: https://doi.org/10.1227/NEU.0b013e318285c3db
Cahill J, Cahill WJ, Calvert JW, Calvert JH, Zhang JH (2006) Mechanisms of early brain injury after subarachnoid hemorrhage. J Cereb Blood Flow Metab 26:1341–1353. https://doi.org/10.1038/sj.jcbfm.9600283
Chang C-Z, Lin C-L, Wu S-C, Kwan A-L (2014) Purpurogallin, a natural phenol, attenuates high-mobility group box 1 in subarachnoid hemorrhage induced vasospasm in a rat model. Int J Vasc Med 2014:254270. https://doi.org/10.1155/2014/254270
Chang C-Z, Wu S-C, Kwan A-L, Lin C-L (2015) 4’-O-β-D-glucosyl-5-O-methylvisamminol, an active ingredient of Saposhnikovia divaricata, attenuates high-mobility group box 1 and subarachnoid hemorrhage-induced vasospasm in a rat model. Behav Brain Funct 11:28. https://doi.org/10.1186/s12993-015-0074-8
Chang C-Z, Wu S-C, Kwan A-L, Lin C-L (2016) Rhinacanthin-C, A fat-soluble extract from Rhinacanthus nasutus, modulates high-mobility group box 1-related neuro-inflammation and subarachnoid hemorrhage-induced brain apoptosis in a rat model. World Neurosurg 86:349–360. https://doi.org/10.1016/j.wneu.2015.08.071
Chaudhry SR, Güresir A, Stoffel-Wagner B, Fimmers R, Kinfe TM, Dietrich D, Lamprecht A, Vatter H, Güresir E, Muhammad S (2018) Systemic high-mobility group box-1: a novel predictive biomarker for cerebral vasospasm in aneurysmal subarachnoid hemorrhage. Crit Care Med 46:e1023–e1028. https://doi.org/10.1097/CCM.0000000000003319
Chen GY, Nuñez G (2010) Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol 10:826–837. https://doi.org/10.1038/nri2873
Dankbaar JW, Rijsdijk M, van der Schaaf IC, Velthuis BK, Wermer MJH, Rinkel GJE (2009) Relationship between vasospasm, cerebral perfusion, and delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Neuroradiology 51:813–819. https://doi.org/10.1007/s00234-009-0575-y
Dankbaar JW, de Rooij NK, Velthuis BK, Frijns CJM, Rinkel GJE, van der Schaaf IC (2009) Diagnosing delayed cerebral ischemia with different CT modalities in patients with subarachnoid hemorrhage with clinical deterioration. Stroke 40:3493–3498. https://doi.org/10.1161/STROKEAHA.109.559013
Dhar R, Diringer MN (2008) The burden of the systemic inflammatory response predicts vasospasm and outcome after subarachnoid hemorrhage. Neurocrit Care 8:404–412. https://doi.org/10.1007/s12028-008-9054-2
Döring Y, Libby P, Soehnlein O (2020) Neutrophil extracellular traps participate in cardiovascular diseases: recent experimental and clinical insights. Circ Res 126:1228–1241. https://doi.org/10.1161/CIRCRESAHA.120.315931
Dreier JP, Major S, Manning A, Woitzik J, Drenckhahn C, Steinbrink J, Tolias C, Oliveira-Ferreira AI, Fabricius M, Hartings JA, Vajkoczy P, Lauritzen M, Dirnagl U, Bohner G, Strong AJ, COSBID study group (2009) Cortical spreading ischaemia is a novel process involved in ischaemic damage in patients with aneurysmal subarachnoid haemorrhage. Brain 132:1866–1881. https://doi.org/10.1093/brain/awp102
Fuchs TA, Brill A, Duerschmied D, Schatzberg D, Monestier M, Myers DD, Wrobleski SK, Wakefield TW, Hartwig JH, Wagner DD (2010) Extracellular DNA traps promote thrombosis. Proc Natl Acad Sci U S A 107:15880–15885. https://doi.org/10.1073/pnas.1005743107
Fujii M, Yan J, Rolland WB, Soejima Y, Caner B, Zhang JH (2013) Early brain injury, an evolving frontier in subarachnoid hemorrhage research. Transl Stroke Res 4:432–446. https://doi.org/10.1007/s12975-013-0257-2
Haruma J, Teshigawara K, Hishikawa T, Wang D, Liu K, Wake H, Mori S, Takahashi HK, Sugiu K, Date I, Nishibori M (2016) Anti-high mobility group box-1 (HMGB1) antibody attenuates delayed cerebral vasospasm and brain injury after subarachnoid hemorrhage in rats. Sci Rep 6:37755. https://doi.org/10.1038/srep37755
Hendrix P, Foreman PM, Harrigan MR, Fisher WS, Vyas NA, Lipsky RH, Lin M, Walters BC, Tubbs RS, Shoja MM, Pittet J-F, Mathru M, Griessenauer CJ (2017) Impact of high-mobility group box 1 polymorphism on delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. World Neurosurg 101:325–330. https://doi.org/10.1016/j.wneu.2017.01.121
Hijdra A, van Gijn J, Nagelkerke NJ, Vermeulen M, van Crevel H (1988) Prediction of delayed cerebral ischemia, rebleeding, and outcome after aneurysmal subarachnoid hemorrhage. Stroke 19:1250–1256. https://doi.org/10.1161/01.str.19.10.1250
Kiiski H, Långsjö J, Tenhunen J, Ala-Peijari M, Huhtala H, Hämäläinen M, Moilanen E, Öhman J, Peltola J (2017) Time-courses of plasma IL-6 and HMGB-1 reflect initial severity of clinical presentation but do not predict poor neurologic outcome following subarachnoid hemorrhage. eNeurologicalSci 6:55–62. https://doi.org/10.1016/j.ensci.2016.11.010
Kim J-B, Sig Choi J, Yu Y-M, Nam K, Piao C-S, Kim S-W, Lee M-H, Han P-L, Park J-S, Lee J-K (2006) HMGB1, a novel cytokine-like mediator linking acute neuronal death and delayed neuroinflammation in the postischemic brain. J Neurosci 26:6413–6421. https://doi.org/10.1523/JNEUROSCI.3815-05.2006
Kim S-W, Lee H, Lee H-K, Kim I-D, Lee J-K (2019) Neutrophil extracellular trap induced by HMGB1 exacerbates damages in the ischemic brain. Acta Neuropathol Commun 7:94. https://doi.org/10.1186/s40478-019-0747-x
Kim S-W, Lee J-K (2020) Role of HMGB1 in the interplay between NETosis and Thrombosis in ischemic stroke: a review. Cells 9:1794. https://doi.org/10.3390/cells9081794
Klune JR, Dhupar R, Cardinal J, Billiar TR, Tsung A (2008) HMGB1: endogenous danger signaling. Mol Med 14:476–484. https://doi.org/10.2119/2008-00034.Klune
Kornblit B, Munthe-Fog L, Madsen HO, Strøm J, Vindeløv L, Garred P (2008) Association of HMGB1 polymorphisms with outcome in patients with systemic inflammatory response syndrome. Crit Care 12:R83. https://doi.org/10.1186/cc6935
Laridan E, Denorme F, Desender L, François O, Andersson T, Deckmyn H, Vanhoorelbeke K, De Meyer SF (2017) Neutrophil extracellular traps in ischemic stroke thrombi. Ann Neurol 82:223–232. https://doi.org/10.1002/ana.24993
Li H, Wu W, Sun Q, Liu M, Li W, Zhang X, Zhou M, Hang C (2014) Expression and cell distribution of receptor for advanced glycation end-products in the rat cortex following experimental subarachnoid hemorrhage. Brain Res 1543:315–323. https://doi.org/10.1016/j.brainres.2013.11.023
Li Y, Sun F, Jing Z, Wang X, Hua X, Wan L (2017) Glycyrrhizic acid exerts anti-inflammatory effect to improve cerebral vasospasm secondary to subarachnoid hemorrhage in a rat model. Neurol Res 39:727–732. https://doi.org/10.1080/01616412.2017.1316903
Macdonald RL (2014) Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol 10:44–58. https://doi.org/10.1038/nrneurol.2013.246
Macdonald RL, Schweizer TA (2017) Spontaneous subarachnoid haemorrhage. The Lancet 389:655–666. https://doi.org/10.1016/S0140-6736(16)30668-7
Martinod K, Wagner DD (2014) Thrombosis: tangled up in NETs. Blood 123:2768–2776. https://doi.org/10.1182/blood-2013-10-463646
Muhammad S, Barakat W, Stoyanov S, Murikinati S, Yang H, Tracey KJ, Bendszus M, Rossetti G, Nawroth PP, Bierhaus A, Schwaninger M (2008) The HMGB1 receptor RAGE mediates ischemic brain damage. J Neurosci 28:12023–12031. https://doi.org/10.1523/JNEUROSCI.2435-08.2008
Rabinstein AA, Friedman JA, Weigand SD, McClelland RL, Fulgham JR, Manno EM, Atkinson JLD, Wijdicks EFM (2004) Predictors of cerebral infarction in aneurysmal subarachnoid hemorrhage. Stroke 35:1862–1866. https://doi.org/10.1161/01.STR.0000133132.76983.8e
de Rooij NK, Rinkel GJE, Dankbaar JW, Frijns CJM (2013) Delayed cerebral ischemia after subarachnoid hemorrhage: a systematic review of clinical, laboratory, and radiological predictors. Stroke 44:43–54. https://doi.org/10.1161/STROKEAHA.112.674291
Roquer J, Cuadrado-Godia E, Guimaraens L, Conesa G, Rodríguez-Campello A, Capellades J, García-Arnillas MP, Fernández-Candil JL, Avellaneda-Gómez C, Giralt-Steinhauer E, Jiménez-Conde J, Soriano-Tárraga C, Villalba-Martínez G, Vivanco-Hidalgo RM, Vivas E, Ois A (2020) Short- and long-term outcome of patients with aneurysmal subarachnoid hemorrhage. Neurology 95:e1819–e1829. https://doi.org/10.1212/WNL.0000000000010618
Saand AR, Yu F, Chen J, Chou SH-Y (2019) Systemic inflammation in hemorrhagic strokes — a novel neurological sign and therapeutic target? J Cereb Blood Flow Metab 39:959–988. https://doi.org/10.1177/0271678X19841443
Schatlo B, Fung C, Stienen MN, Fathi AR, Fandino J, Smoll NR, Zumofen D, Daniel RT, Burkhardt J-K, Bervini D, Marbacher S, Reinert M, Alonzo D, Ahlborn P, Mendes Pereira V, Roethlisberger M, Seule M, Kerkeni H, Remonda L, Weyerbrock A, Woernle K, Venier A, Perren F, Sailer M, Robert T, Rohde V, Schöni D, Goldberg J, Nevzati E, Diepers M, Gralla J, Z’Graggen W, Starnoni D, Woernle C, Maldaner N, Kulcsar Z, Mostaguir K, Maduri R, Eisenring C, Bernays R, Ferrari A, Dan-Ura H, Finkenstädt S, Gasche Y, Sarrafzadeh A, Jakob SM, Corniola M, Baumann F, Regli L, Levivier M, Hildebrandt G, Landolt H, Mariani L, Guzman R, Beck J, Raabe A, Keller E, Bijlenga P, Schaller K (2021) Incidence and outcome of aneurysmal subarachnoid hemorrhage: the Swiss Study on Subarachnoid Hemorrhage (Swiss SOS). Stroke 52(344):347. https://doi.org/10.1161/STROKEAHA.120.029538
Stienen MN, Visser-Meily JM, Schweizer TA, Hänggi D, Macdonald RL, Vergouwen MDI, Unruptured Intracranial Aneurysms and SAH CDE Project Investigators (2019) Prioritization and timing of outcomes and endpoints after aneurysmal subarachnoid hemorrhage in clinical trials and observational studies: proposal of a multidisciplinary research group. Neurocrit Care 30(102):113. https://doi.org/10.1007/s12028-019-00737-0
Sun Q, Wu W, Hu Y-C, Li H, Zhang D, Li S, Li W, Li W-D, Ma B, Zhu J-H, Zhou M-L, Hang C-H (2014) Early release of high-mobility group box 1 (HMGB1) from neurons in experimental subarachnoid hemorrhage in vivo and in vitro. J Neuroinflammation 11:106. https://doi.org/10.1186/1742-2094-11-106
Tian X, Sun L, Feng D, Sun Q, Dou Y, Liu C, Zhou F, Li H, Shen H, Wang Z, Chen G (2017) HMGB1 promotes neurovascular remodeling via Rage in the late phase of subarachnoid hemorrhage. Brain Res 1670:135–145. https://doi.org/10.1016/j.brainres.2017.06.001
Venereau E, De Leo F, Mezzapelle R, Careccia G, Musco G, Bianchi ME (2016) HMGB1 as biomarker and drug target. Pharmacol Res 111:534–544. https://doi.org/10.1016/j.phrs.2016.06.031
Vergouwen MDI, Vermeulen M, Coert BA, Stroes ESG, Roos YBWEM (2008) Microthrombosis after aneurysmal subarachnoid hemorrhage: an additional explanation for delayed cerebral ischemia. J Cereb Blood Flow Metab 28:1761–1770. https://doi.org/10.1038/jcbfm.2008.74
Vergouwen MDI, Vermeulen M, van Gijn J, Rinkel GJE, Wijdicks EF, Muizelaar JP, Mendelow AD, Juvela S, Yonas H, Terbrugge KG, Macdonald RL, Diringer MN, Broderick JP, Dreier JP, Roos YBWEM (2010) Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke 41:2391–2395. https://doi.org/10.1161/STROKEAHA.110.589275
Haoliang Xu, Chanannait P, Lizhen M, Francesco V, Dale P (2011) Abstract 10081: inhibition of RAGE-related pathway prevents subarachnoid hemorrhage (sah)-associated neuroinflammation and neurologic dysfunction in rats. Circulation 124:A10081–A10081. https://doi.org/10.1161/circ.124.suppl_21.A10081
Zhu X-D, Chen J-S, Zhou F, Liu Q-C, Chen G, Zhang J-M (2012) Relationship between plasma high mobility group box-1 protein levels and clinical outcomes of aneurysmal subarachnoid hemorrhage. J Neuroinflammation 9:194. https://doi.org/10.1186/1742-2094-9-194
Acknowledgements
The authors highly appreciate the generous support of Wolfgang Thomann, and the technical support of Birgit Beilmann-Höh and Tim Schmidt. The authors would like to specifically thank the nursing staff of the neurosurgical intensive and intermediate care units (NC-01, NC-05) for their strong support of the study. The authors appreciate funding support from the Homburger Forschungsförderung (HOMFOR 2018) and the Dr. Theiss Research Award 2017.
Funding
This study was financially supported by intramural research grants to PH obtained from the Homburger Forschungsförderung (HOMFOR 2018) and the Dr. Theiss Research Award 2017.
Author information
Authors and Affiliations
Contributions
SH and PH performed data analysis, discussed results, created figures, and wrote the manuscript. SH, SS, PH collected patient data and blood samples. JG analyzed samples and provided technical supervision. CG, AS, JG, JO provided technical and administrative support. PH designed the study. JO and PH provided clinical supervision. All authors reviewed and edited the manuscript for content.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the ethics committee of the Saarland Medical Association 118/17. The study was performed in accordance with the Declaration of Helsinki. All patients or their legal representatives gave informed consent. All patients or their legal representatives gave informed consent.
Consent for publication
N/A, no identifiable information.
Conflict of interest
None of the authors have any conflicts of interests regarding the submitted work.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hemmer, S., Senger, S., Griessenauer, C.J. et al. Admission serum high mobility group box 1 (HMGB1) protein predicts delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurg Rev 45, 807–817 (2022). https://doi.org/10.1007/s10143-021-01607-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-021-01607-0