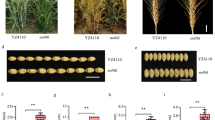
Abstract
Interspecific hybridization can be considered an accelerator of evolution, otherwise a slow process, solely dependent on mutation and recombination. Upon interspecific hybridization, several novel interactions between nuclear and cytoplasmic genomes emerge which provide additional sources of diversity. The magnitude and essence of intergenomic interactions between nuclear and cytoplasmic genomes remain unknown due to the direction of many crosses. This study was conducted to address the role of nuclear-cytoplasmic interactions as a source of variation upon hybridization. Wheat (Triticum aestivum) alloplasmic lines carrying the cytoplasm of Aegilops mutica along with an integrated approach utilizing comparative quantitative trait locus (QTL) and epigenome analysis were used to dissect this interaction. The results indicate that cytoplasmic genomes can modify the magnitude of QTL controlling certain physiological traits such as dry matter weight. Furthermore, methylation profiling analysis detected eight polymorphic regions affected by the cytoplasm type. In general, these results indicate that novel nuclear-cytoplasmic interactions can potentially trigger an epigenetic modification cascade in nuclear genes which eventually change the genetic network controlling physiological traits. These modified genetic networks can serve as new sources of variation to accelerate the evolutionary process. Furthermore, this variation can synthetically be produced by breeders in their programs to develop epigenomic-segregating lines.




Similar content being viewed by others
References
Acosta MC, Premoli AC (2010) Evidence of chloroplast capture in South American Nothofagus (subgenus Nothofagus, Nothofagaceae). Mol Phylogenet Evol 54:235–242. doi:10.1016/j.ympev.2009.08.008
Adams KL, Wendel JF (2005) Novel patterns of gene expression in polyploid plants. Trends Genet 21:539–543. doi:10.1016/j.tig.2005.07.009
Allainguillaume J et al (2009) Rapeseed cytoplasm gives advantage in wild relatives and complicates genetically modified crop biocontainment. New Phytol 183:1201–1211. doi:10.1111/j.1469-8137.2009.02877.x
Barakat MN, Wahba LE, Milad SI (2013) Molecular mapping of QTLs for wheat flag leaf senescence under water-stress. Biol Plant 57:79–84
Bassam BJ, Caetanoanolles G, Gresshoff PM (1991) Fast and sensitive silver staining of DNA in polyacrylamide gels. Anal Biochem 196:80–83. doi:10.1016/0003-2697(91)90120-I
Belmonte MF et al (2013) Comprehensive developmental profiles of gene activity in regions and subregions of the Arabidopsis seed. Proc Natl Acad Sci U S A 110:E435–E444. doi:10.1073/pnas.1222061110
Broman KW, Wu H, Sen S, Churchill GA (2003) R/qtl: QTL mapping in experimental crosses. Bioinformatics 19:889–890. doi:10.1093/bioinformatics/btg112
Budar F, Roux F (2011) The role of organelle genomes in plant adaptation: time to get to work! Plant Signal Behav 6:635–639
Cavanagh CR et al (2013) Genome-wide comparative diversity uncovers multiple targets of selection for improvement in hexaploid wheat landraces and cultivars. Proc Natl Acad Sci U S A 110:8057–8062. doi:10.1073/pnas.1217133110
Chen ZJ (2007) Genetic and epigenetic mechanisms for gene expression and phenotypic variation in plant polyploids. Annu Rev Plant Biol 58:377–406. doi:10.1146/annurev.arplant.58.032806.103835
Childs KL et al (2007) The TIGR plant transcript assemblies database. Nucleic Acids Res 35:D846–D851. doi:10.1093/Nar/Gkl785
Development Core Team R (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Dobrovolskaya O, Martinek P, Voylokov AV, Korzun V, Roder MS, Borner A (2009) Microsatellite mapping of genes that determine supernumerary spikelets in wheat (T. aestivum) and rye (S. cereale). Theor Appl Genet 119:867–874. doi:10.1007/s00122-009-1095-1
Fey V et al (2005) Retrograde plastid redox signals in the expression of nuclear genes for chloroplast proteins of Arabidopsis thaliana. J Biol Chem 280:17572–17572
Gehring M (2013) Genomic imprinting: insights from plants. Annu Rev Genet 47:187–208
He GM et al (2010) Global epigenetic and transcriptional trends among two rice subspecies and their reciprocal hybrids. Plant Cell 22:17–33. doi:10.1105/tpc.109.072041
Hegarty MJ, Hiscock SJ (2005) Hybrid speciation in plants: new insights from molecular studies. New Phytol 165:411–423. doi:10.1111/j.1469-8137.2004.01253.x
Jackson S, Chen ZJ (2010) Genomic and expression plasticity of polyploidy. Curr Opin Plant Biol 13:153–159. doi:10.1016/j.pbi.2009.11.004
Kohler C, Hennig L, Spillane C, Pien S, Gruissem W, Grossniklaus U (2003) The polycomb-group protein MEDEA regulates seed development by controlling expression of the MADS-box gene PHERES1. Genes Dev 17:1540–1553. doi:10.1101/Gad.257403
Lee KP, Kim C, Landgraf F, Apel K (2007) EXECUTER1- and EXECUTER2-dependent transfer of stress-related signals from the plastid to the nucleus of Arabidopsis thaliana. Proc Natl Acad Sci U S A 104:10270–10275. doi:10.1073/pnas.0702061104
Leinonen PH, Remington DL, Leppala J, Savolainen O (2013) Genetic basis of local adaptation and flowering time variation in Arabidopsis lyrata. Mol Ecol 22:709–723. doi:10.1111/j.1365-294X.2012.05678.x
Levin DA (2003) The cytoplasmic factor in plant speciation. Syst Bot 28:5–11
Maan SS (1977) Cytoplasmic homology between Aegilops-Mutica-Boiss and Aegilops-Ovata L. Euphytica 26:601–613. doi:10.1007/Bf00021685
Maan SS (1991) Nucleo-cytoplasmic genetics of wheat. In: Sasakuma T, Kinoshita (eds) Proc. Int. Symp. Nuclear and Organeller Genetics of Wheat. Hokkaido University, Sapporo, Japan, pp. 175–194
Maan SS (1992) A Gene for embryo-endosperm compatibility and seed viability in alloplasmic Triticum turgidum. Genome 35:772–779
Madlung A et al (2005) Genomic changes in synthetic Arabidopsis polyploids. Plant J 41:221–230. doi:10.1111/j.1365-313X.2004.02297.x
Mallet J (2005) Hybridization as an invasion of the genome. Trends Ecol Evol 20:229–237. doi:10.1016/j.tree.2005.02.010
Mallet J (2007) Hybrid speciation. Nature 446:279–283. doi:10.1038/Nature05706
Mcclintock B (1984) The significance of responses of the genome to challenge. Science 226:792–801. doi:10.1126/science.15739260
Nagaharu U (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilisation. Jpn J Bot 389-452
Najeb Barakat M, Al-Doss AA, Elshafei AA, Moustafa KA (2011) Identification of new microsatellite marker linked to the grain filling rate as indicator for heat tolerance genes in F2 wheat population. Aust J Crop Sci 5(2):104–105
Peng ZS, Yen C, Yang JL (1998) Chromosomal location of genes for supernumerary spikelet in bread wheat. Euphytica 103:109–114
Rand DM, Haney RA, Fry AJ (2004) Cytonuclear coevolution: the genomics of cooperation. Trends Ecol Evol 19:645–653. doi:10.1016/j.tree.2004.10.003
Rhoads DM, Subbaiah CC (2007) Mitochondrial retrograde regulation in plants. Mitochondrion 7:177–194. doi:10.1016/j.mito.2007.01.002
Sall T, Jakobsson M, Lind-Hallden C, Hallden C (2003) Chloroplast DNA indicates a single origin of the allotetraplold Arabidopsis suecica. J Evol Biol 16:1019–1029. doi:10.1046/j.1420-9101.2003.00554.x
Santamaria RD et al (2014) MSH1-induced non-genetic variation provides a source of phenotypic diversity in sorghum bicolor. Plos One 9:e108407. doi:10.1371/journal.pone.0108407
Soltis DE, Soltis PS (1999) Polyploidy: recurrent formation and genome evolution. Trends Ecol Evol 14:348–352. doi:10.1016/S0169-5347(99)01638-9
Soltis PS, Soltis DE (2009) The role of hybridization in plant speciation. Annu Rev Plant Biol 60:561–588. doi:10.1146/annurev.arplant.043008.092039
Strand A, Asami T, Alonso J, Ecker JR, Chory J (2003) Chloroplast to nucleus communication triggered by accumulation of Mg-protoporphyrinIX. Nature 421:79–83. doi:10.1038/Nature01204
Thanh PT, Vladutu CI, Kianian SF, Thanh PT, Ishii T, Nitta M, Nasuda S, Mori N (2013) Molecular genetic analysis of domestication traits in emmer wheat. I: Map construction and QTL analysis using an F2 pupulation. Bio Biotech Equip 27(2):3627–3637
Tsunewaki K (2009) Plasmon analysis in the Triticum-Aegilops complex breeding. Science 59:455–470
Tsunewaki K, Wang GZ, Matsuoka Y (1996) Plasmon analysis of Triticum (wheat) and Aegilops .1. Production of alloplasmic common wheats and their fertilities. Genes Genet Syst 71:293–311. doi:10.1266/Ggs.71.293
Virdi KS et al (2015) Arabidopsis MSH1 mutation alters the epigenome and produces heritable changes in plant growth. Nat Commun 6:6386. doi:10.1038/Ncomms7386
Wang S, Basten CJ, Zeng ZB (2012) Windows QTL Cartographer 2.5. Department of Statistics, North Carolina State University, Raleigh
Wendel JF, Cronn RC (2003) Polyploidy and the evolutionary history of cotton. Adv Agron 78:139–186. doi:10.1016/S0065-2113(02)78004-8
Wilkinson PA, Winfield MO, Barker GLA, Allen AM, Burridge A, Coghill JA, Edwards KJ (2012) CerealsDB 2.0: an integrated resource for plant breeders and scientists. BMC Bioinformatics 13:219. doi:10.1186/1471-2105-13-219
Woodson JD, Chory J (2008) Coordination of gene expression between organellar and nuclear genomes. Nat Rev Genet 9:383–395. doi:10.1038/Nrg2348
Yang X et al (2015) MSH1-derived epigenetic breeding potential in tomato. Plant Physiol. doi:10.1104/pp.15.00075
Acknowledgments
We would like to thank Professor S.S. Maan and Professor K. Tsunewaki for the decades of dedicated effort in the development of alloplasmic wheat lines and providing them for use in this study. We are also grateful for all the assistance received from members of the Wheat Germplasm Enhancement and the Hard Red Spring Wheat programs at NDSU. This research was supported by NSF-IOS 1361554 to S.F.K. and Monsanto Beachell-Borlaug International Scholars program supporting A.S. in his graduate studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Disclaimers
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Expression analysis of detected polymorphic methylated genes. Blue bars indicate the expression level of genes in alloplasmic condition relative to euplasmic expression level (red bars). For more information, refer to Table S1. (PDF 163 kb)
Table S1
Sequences of primers used for expression analysis (DOCX 10 kb)
Rights and permissions
About this article
Cite this article
Soltani, A., Kumar, A., Mergoum, M. et al. Novel nuclear-cytoplasmic interaction in wheat (Triticum aestivum) induces vigorous plants. Funct Integr Genomics 16, 171–182 (2016). https://doi.org/10.1007/s10142-016-0475-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-016-0475-2