Abstract
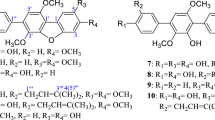
A screening was performed using nine marine-derived fungi as biocatalysts and the natural products (−)-ambrox® (1), (−)-sclareol (2), and (+)-sclareolide (3) in order to select the microorganisms able to catalyze the biooxidation of these compounds. It was observed that only the Aspergillus sydowii CBMAI 934, Botryosphaeria sp., Eutypella sp., and Xylaria sp. presented active oxidoreductases and catalyzed the regioselective hydroxylation in the natural products. The hydroxylated metabolites obtained were 1β-hydroxy-ambrox (1a) (14 %, A. sydowii CBMAI 934); 3β-hydroxy-ambrox (1b) (17 %, Botryosphaeria sp.; 11 %, Eutypella sp.); 3β-hydroxy-sclareol (2a) (31 %, Xylaria sp.; 69 %, Botryosphaeria sp.; 55 %, Eutypella sp.); 18-hydroxy-sclareol (2b) (10 %, Xylaria sp.); and 3β-hydroxy-sclareolide (3a) (34 %, Botryosphaeria sp.; 7 %, Eutypella sp.). This is the first report of biohydroxylation of (−)-ambrox® (1), (−)-sclareol (2), and (+)-sclareolide (3) by whole mycelia of marine-derived fungi.





Similar content being viewed by others
References
Abraham WR (1994) Microbial hydroxylation of sclareol. Phytochemistry 36:1421–1424
Allendes JA, Bustos DA, Pacciaroni ADV, Sosa VE, Bustos DA (2011) Microbial functionalization of (−)-ambroxide by filamentous fungi. Biocatal Biotransfor 29:83–86
Aranda G, Lallemand JY, Mammoumi A, Azerad R (1991) Microbial hydroxylation of sclareol by Mucor plumbeus. Tetrahedron Lett 32:1783–1786
Ata A, Conci LJ, Betteridge J, Orhan I, Sener B (2007) Novel microbial transformations of sclareolide. Chem Pharm Bull 55:118–123
Atta-ur-Rahman FA, Choudhary M (1997) I. Microbial transformation of sclareolide. J Nat Prod 60:1038–1040
Cano A, Ramírez-Apan MT, Delgado G (2011) Biotransformation of sclareolide by filamentous fungi: cytotoxic evaluations of the derivatives. J Braz Chem Soc 22:1177–1182
Choudhary MI, Musharraf SG, Sami A, Rahman A (2004) Microbial transformation of sesquiterpenes (−)-ambrox and (+)-sclareolide. Helv Chim Acta 87:2685–2694
De Carvalho CCCR, Da Fonseca MMR (2006) Biotransformation of terpenes. Biotechnol Adv 24:134–142
Debashish G, Malay S, Barindra S, Joydeep M (2005) Marine enzymes. Adv Biochem Eng Biotechnol 96:189–218
Díez D, Sanchez JM, Rodilla JM, Rocha PM, Mendes RS, Paulino C, Marcos IS, Basabe P, Urones JG (2005) Microbial hydroxylation of sclareol by Rhizopus stolonifer. Molecules 10:1005–1009
Feng Z, Nenkep VN, Yun K, Zhang D, Choi HD, Kang JS, Son BW (2010) Biotransformation of bioactive (−)-mellein by a marine isolate of bacterium Stappia sp. J Microbiol Biotechnol 20:985–987
Hanson JR, Truneh A (1996) The biotransformation of ambrox and sclareolide by Cephalosporium aphidicola. Photochemistry 42:1021–1023
Hanson JR, Hitchcock PB, Nasir H, Truneh A (1994) The biotransformation of the diterpenoid, sclareol, by Cephalosporium aphidicola. Photochemistry 36:903–906
Klemke C, Kehraus S, Wright AD, Konig GM (2004) New secondary metabolites from the marine endophytic fungus Apiospora montagnei. J Nat Prod 67:1058–1063
Kohlmeyer J (1974) Higher fungi as parasites and symbionts of algae. VeröffInst Meeresforsch Bremenh 5:339–356
Koshimura M, Utsukihara T, Kawamoto M, Saito M, Horiuchi CA, Kuniyoshi M (2009) Biotransformation of bromo-sesquiterpenes by marine fungi. Photochemistry 70:2023–2026
Kouzi SA, McChesney JD (1991) Microbial models of mammalian metabolism: fungal metabolism of the diterpenesclareol by Cunningahmella species. J Nat Prod 54:483–490
Nasib A, Musharraf SG, Hussain S, Khan S, Anjum S, Ali S, Rahman A, Choudhary MI (2006) Biotransformation of (−)-ambrox by cell suspension cultures of Actinidia deliciosa. J Nat Prod 69:957–959
Osterhage C, Konig GM, Holler U, Wright AD (2002) Rare sesquiterpenes from the algicolous fungus Drechslera dematioidea. J Nat Prod 65:306–313
Rovirosa J, Astudillo L, Sanchez I, Palacios Y, San-Martin A (1989) Antimicrobial activity of halogenated sesquiterpenes of Laurencia claviformis from Easter Island. Bol de la Sociedad Chilena de Quimica 34:147–152
San-Martín A, Rovirosa J, Astudillo L, Sepúlveda B, Ruiz D, San-Martín C (2008) Biotransformation of the marine sesquiterpene pacifenol by a facultative marine fungus. Nat Prod Res 22:1627–1632
Sarkar S, Pramanik A, Mitra A, Mukherjee J (2010) Bioprocessing data for the production of marine enzymes. Mar Drugs 8:1323–1372
Tenius BSM, Schroeder EK, Achenato RA (2000) Ambergris: perfume e síntese. Quim Nova 23:225–230
Trincone A (2010) Potential biocatalysts originating from sea environments. J Mol Catal B Enzym 66:241–256
Trincone A (2011) Marine biocatalysts: enzymatic features and applications. Mar Drugs 9:478–499
Zhang C, Kim S-K (2010) Research and application of marine microbial enzymes: status and prospects. Mar Drugs 8:1920–1934
Acknowledgments
A. L. M. Porto gratefully acknowledges Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP) for their financial support. M. P. Martins thanks CAPES for the scholarships (Sandwich). The authors wish to thank Professor Roberto G. S. Berlinck for donating the marine-derived fungi and also wish to thank Professor Timothy John Brockson (Universidade Federal de São Carlos) for optical rotation measurements. The authors also thank Sylvie Cordial and Claudine Servy for the discussions and suggestions during the development of work. The English language was reviewed by Timothy Roberts, MSc., a native English speaker.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 9457 kb)
Rights and permissions
About this article
Cite this article
Martins, M.P., Ouazzani, J., Arcile, G. et al. Biohydroxylation of (−)-Ambrox®, (−)-Sclareol, and (+)-Sclareolide by Whole Cells of Brazilian Marine-Derived Fungi. Mar Biotechnol 17, 211–218 (2015). https://doi.org/10.1007/s10126-015-9610-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-015-9610-7