Abstract
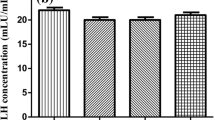
The main objective of this in vivo study was to investigate the effect of different low-level laser therapy (LLLT) doses on polycystic ovary syndrome (PCOS). In the present experimental study, a single dosage of estradiol valerate (EV) was administered to induce PCOS in female rats. After administration of the EV for induction of PCOS, rats were divided into 5 groups (n = 8/group): C group (animals that were not exposed to any form of procedure), PC group (no treatment following EV induction), L1 group (1 J/cm2 LLLT treatment following EV induction), L2 group (2 J/cm2 LLLT treatment following EV induction), L3 group (6 J/cm2 LLLT treatment following EV induction). The results indicated that no significant difference was found in the serum levels of luteinizing hormone (LH), follicle-stimulating hormone (FSH), and progesterone (P4) between the C and L2 groups (p < 0.05). Although the serum levels of testosterone (T) were significantly higher in the C group compared with other groups (p < 0.05), the L2 group was determined to be the closest to the C group. Additionally, the LH, FSH, and T receptor level of the L2 group was closest to the C group. In conclusion, a 2 J/cm2 dosage of LLLT (L2 group) can be considered the most potentially effective treatment of PCOS in the rat. However, more studies are needed to determine the optimal dose of LLLT for the treatment of PCOS.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Hernandez-Medrano JH, Campbell BK, Webb R (2012) Nutritional influences on folli- culogenesis. Reprod Domest Anim 47:274–282. https://doi.org/10.1111/j.1439-0531.2012.02086.x
Chaudhari N, Dawalbhakta M, Nampoothiri L (2018) GnRH dysregulation in polycystic ovarian syndrome (PCOS) is a manifestation of an altered neurotransmitter profile. Reprod Biol Endocrinol 16:1–13. https://doi.org/10.1186/s12958-018-0354-x
Pişkinpaşa S, Yıldız BO (2005) Polikistik over sendromu. Hacettepe Tıp Dergisi 36:168–174
Azziz R (2016) PCOS in 2015: new insights into the genetics of polycystic ovary syndrome. Nat Rev Endocrinol 12(3):74–75. https://doi.org/10.1038/nrendo.2015.230
Noroozzadeh M, Behboudi-Gandevani S, Zadeh-Vakili A, Tehrani FR (2017) Hormone-induced rat model of polycystic ovary syndrome: a systematic review. Life sci 191:259–272. https://doi.org/10.1016/j.lfs.2017.10.020
Singh KB (2005) Persistent estrus rat models of polycystic ovary disease: an update. Fertil steril 84:1228–1234. https://doi.org/10.1016/j.fertnstert.2005.06.013
Tsilchorozidou T, Overton C, Conway GS (2004) The pathophysiology of polycystic ovary syndrome. Clin Endocrinol (Oxf) 60:1–17. https://doi.org/10.1046/j.1365-2265.2003.01842.x
Park CH, Chun S (2016) Association between serum gonadotropin level and insulin resistance- related parameters in Korean women with polycystic ovary syndrome. Obstet Gynecol Sci 59(6):498–505. https://doi.org/10.5468/ogs.2016.59.6.498
Duleba AJ (2012) Medical management of metabolic dysfunction in PCOS. Steroids 77(4):306–311. https://doi.org/10.1016/j.steroids.2011.11.014
Sirmans SM, Pate KA (2014) Epidemiology, diagnosis, and management of polycystic ovary syndrome. Clin Epidemiol 6:1–13. https://doi.org/10.2147/CLEP.S37559
Kabiri N, Mohammad R, Seyed R (2014) Beneficial effects of pioglitazone and metformin in murinr model of polycystic ovaries via improvement of chemerin gene up-regulation. J Pharm Sci 22:39. https://doi.org/10.1186/2008-2231-22-39
Tannys D, Ottawa A (2010) Vancouver. Déclenchement de l’ovulation en présence du SOPK. Directive clinique de la SOGC 242:503–511
Stang M, Wysowski DK, Butler-Jones D (1999) Incidence of lactic acidosis in metformin users. Diabetes Care 22:925–927
Gruber DM, Huber JC (1999) Mortality associated with oral contraceptive use: 25-year follow-up of a cohort of 46,000 women from Royal College of General Practitioners’s Oral Contraception Study. Analysis and comment on the BMJ study of the long-term risk of using oral contraceptives. Gynakol Geburtshilfliche Rundsch 39:231–232
Thomson RL, Spedding S, Buckley JD (2012) Vitamin D in the aetiology and management of polycystic ovary syndrome. Clin Endocrinol 77:343–350. https://doi.org/10.1111/j.1365-2265.2012.04434.x
Mvondo MA, Mzemdem Tsoplfack FI, Awounfack CF, Njamen D (2020) The leaf aqueous extract of Myrianthus arboreus P. Beauv. (Cecropiaceae) improved letrozole-induced polycystic ovarian syndrome associated conditions and infertility in female Wistar rats. BMC Complement Med Ther 20:1–13. https://doi.org/10.1186/s12906-020-03070-8
Ruh AC, Frigo L, Cavalcanti M, Svidnicki P, Vicari VN, Lopes-Martins RAB, Leal Junior ECP, De Isla N, Diomede F, Trubiani O, Favero GM (2018) Laser photobiomodulation in pressure ulcer healing of human diabetic patients: gene expression analysis of inflammatory biochemical markers. Laser. Med Sci 33:165e171
Hamblin MR, Agrawal T, de Sousa M (2016) Handbook of low-level laser therapy. CRC Press
Oubiña G, Pascuali N, Scotti L, Di Pietro M, La Spina FA, Buffone MG, Parborell F, Higuera J, Abramovich D (2018) Low level laser therapy (LLLT) modulates ovarian function in mature female mice. Prog Biophys Mol Biol 145:10–18. https://doi.org/10.1016/j.pbiomolbio.2018.11.010
Okur S, Okumuş Z (2023) Effects of low-level laser therapy and therapeutic ultrasound on Freund’s complete adjuvant-induced knee arthritis model in rats. Arch Rheumatol 38(1):32
Matic M, Lazetic B, Poljacki M, Duran V, Ivkov-Simic M (2003) Low level laser irradiation and its effect on repair processes in the skin. Med. Pregl 56:137–141. https://doi.org/10.2298/mpns0304137m
Huang YY, Chenet AC, Carroll JD, Hamblin RM (2009) Biphasic dose response in low level light therapy. Dose Response 7(4):358–383
Hamblin MR, Demidova TN (2006) Mechanisms of low level light therapy. Mechanisms for low-light therapy 6140:614001
Tessaro I, Modina SC, Franciosi F, Sivelli G, Terzaghi L, Lodde V, Luciano AM (2015) Effect of oral administration of low-dose follicle stimulating hormone on hyperandrogenized mice as a model of polycystic ovary syndrome. J Ovarian Res 8:64. https://doi.org/10.1186/s13048-015-0192-9
Alves ED, Bonfá ALO, Pigatto GR, Anselmo-Franci JA, Achcar JA, Parizotto NA, Montrezor LH (2019) Photobiomodulation can improve ovarian activity in polycystic ovary syndrome-induced rats. J Photochem Photobiol B: Biol 194:6–13. https://doi.org/10.1016/j.jphotobiol.2019.03.006
Szukiewicz D, Uilenbroek JT (1998) Polycystic ovary syndrome--searching for an animal model. J Med 29(5-6):259–275
Montrezor LH, Carvalho D, Dias MB, Anselmo-Franci JA, Bícego KC, Gargaglioni LH (2015) Hypoxic and hypercapnic ventilatory responses in rats with polycystic ovaries. Respir Physiol Neurobiol 217:17–24. https://doi.org/10.1016/j.resp.2015.06.009
Pereira VM, Honorato-Sampaio K, Martins AS, Reis FM, Reis AM (2014) Downregulation of natriuretic peptide system and increased steroidogenesis in rat polycystic ovary. Peptides 60:80–85. https://doi.org/10.1016/j.peptides.2014.07.027
Uchiyama F, Jikyo T, Takeda R, Ogata M (2014) Lepidium meyenii (Maca) enhances the serum levels of luteinising hormone in female rats. J Ethnopharmacol 151(2):897–902
Hirano S (2012) Western blot analysis. Nanotoxicity: methods and protocols, pp 87–97
Brawer JR, Naftolin F, Martin J, Sonnenschein C (1978) Effects of a single injection of estradiol valerate on the hypothalamic arcuate nucleus and on reproductive function in the female rat. Endocrinology 103(2):501–512. https://doi.org/10.1210/endo-103-2-501
Brawer JR, Munoz M, Farookhi R (1986) Development of the polycystic ovarian condition (PCO) in the estradiol valerate-treated rat. Biol Reprod 35(3):647–655. https://doi.org/10.1095/biolreprod35.3.647
Carriere PD, Brawer JR, Farookhi R (1988) Pituitary gonadotropin-releasing hormone receptor content in rats with polycystic ovaries. Biol Reprod 38(3):562–567. https://doi.org/10.1095/biolreprod38.3.562
Simard M, Brawer JR, Farookhi R (1987) An intractable, ovary-independent impairment in hypothalamo-pituitary function in the estradiol-valerate-induced polycystic ovarian condition in the rat. Biol Reprod 36(5):1229–1237. https://doi.org/10.1095/biolreprod36.5.1229
Stener-Victorin E, Lundeberg T, Waldenstrom U, Manni L, Aloe L, Gunnarsson S, Janson PO (2000) Effects of electro-acupuncture on nerve growth factor and ovarian morphology in rats with experimentally induced polycystic ovaries. Biol Reprod 63:1497–1503. https://doi.org/10.1095/biolreprod63.5.1497
Lara HE, Ferruz JL, Luza S, Bustamante DA, Borges Y, Ojeda SR (1993) Activation of ovarian sympathetic nerves in polycystic ovary syndrome. Endocrinology 133:2690–2695. https://doi.org/10.1210/en.133.6.2690
Walters KA, Allan CM, Handelsman DJ (2012) Rodent models for human polycystic ovary syndrome. Biol reprod 86(5):149–141. https://doi.org/10.1095/biolreprod.111.097808
Masoumipoor M, Jameie SB, Janzadeh A, Nasirinezhad F, Soleimani M, Kerdary M (2014) Effects of 660-and 980-nm low-level laser therapy on neuropathic pain relief following chronic constriction injury in rat sciatic nerve. Lasers Med Sci 29:1593–1598. https://doi.org/10.1007/s10103-014-1552-1
Barbosa D, de Souza RA, Xavier M, da Silva FF, Arisawa EÂL, Villaverde AGJB (2013) Effects of low-level laser therapy (LLLT) on bone repair in rats: optical densitometry analysis. Lasers Med Sci 28:651–656. https://doi.org/10.1007/s10103-012-1125-0
Dumitrescu R, Mehedintu C, Briceag I, Purcarea VL, Hudita D (2015) The polycystic ovary syndrome: an update on metabolic and hormonal mechanisms. J Med Life Sci 8(2):142
Karimi Jashni H, Kargar Jahromi H, Bagheri Z (2016) The effect of palm pollen extract on polycystic ovary syndrome (POS) in rats. Int J Med Res Health Sci 5(5):317–321
Speroff L, Fritz MA (2005) Clinical gynecologic endocrinology and infertility. In: Philadelphia PA (ed) Anovulation and the polycystic ovary, 7th edn. Williams &Wilkins Lippincott
Esmaeilinezhad Z, Babajafari S, Sohrabi Z, Eskandari MH, Amooee S, Barati-Boldaji R (2019) Effect of synbiotic pomegranate juice on glycemic, sex hormone profile and anthropometric indices in PCOS: a randomized, triple blind, controlled trial. Nutr Metab Cardiovasc Dis 29(2):201–208. https://doi.org/10.1016/j.numecd.2018.07.002
Di Pietro M, Pascuali N, Parborell F, Abramovich D (2018) Ovarian angiogenesis in polycystic ovary syndrome. Reproduction 155(5):199–209. https://doi.org/10.1530/REP-17-0597
Abramovich D, Irusta G, Bas D, Cataldi NI, Parborell F, Tesone M (2012) Angiopoietins/TIE2 system and VEGF are involved in ovarian function in a DHEA rat model of polycystic ovary syndrome. Endocrinology 153(7):3446–3456. https://doi.org/10.1210/en.2012-1105
Liebert AD, Bicknell BT, Adams RD (2014) Protein conformational modulation by photons: a mechanism for laser treatment effects. Med Hypotheses 82(3):275–281. https://doi.org/10.1016/j.mehy.2013.12.009
Shin HS, Lee C, Choi JCY (2012) Effects of LED light spectra on the growth of the yellowtail clownfish, Amphiprion clarkia. Fish Sci 78:549–556. https://doi.org/10.1007/s12562-012-0482-8
Huang L, Tang Y, Xing D (2013) Activation of nuclear estrogen receptors induced by low-power laser irradiation via PI3-K/Akt signaling cascade. J Cell Physiol 228:1045–1059. https://doi.org/10.1002/jcp.24252
Berisha B, Schams D (2005) Ovarian function in ruminants. Domest Anim Endocrinol 29(2):305–317. https://doi.org/10.1016/j.domaniend.2005.02.035
Hawkins D, Abrahamse H (2006) Effect of multiple exposures of low-level laser therapy on the cellular responses of wounded human skin fibroblasts. Photomed Laser Surg 24(6):705–771. https://doi.org/10.1089/pho.2006.24.705
Funding
This study received support from Atatürk University Scientific Research Projects (TSA-2019-7440).
Author information
Authors and Affiliations
Contributions
Bülent Polat, Damla Tuğçe Okur: literature search, study design, data collection and interpretation, and manuscript preparation. Damla Tuğçe Okur, Armağan Çolak, Kader Yilmaz: statistical analysis, data collection, interpretation, and manuscript preparation. Selim Çomaklı, Mustafa Özkaraca: histopathological analysis and evaluation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Polat, B., Okur, D.T., Çolak, A. et al. The effects of low-level laser therapy on polycystic ovarian syndrome in rats: three different dosages. Lasers Med Sci 38, 177 (2023). https://doi.org/10.1007/s10103-023-03847-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-023-03847-6