Abstract
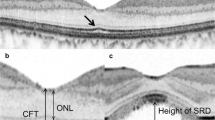
The study aims to investigate the morphological integrity of the outer retinal layers (ORLs) (an ellipsoid layer (EL) + external limiting membrane) and visual prognosis in chronic central serous chorioretinopathy (CSCR) with subretinal fluid (SRF) completely resorbed after half-dose photodynamic therapy (HD PDT) using enhanced-depth imaging (EDI) spectral domain optical coherence tomography (SD-OCT). This retrospective study included 40 eyes of 38 chronic CSCR patients treated with HD PDT between December 2012 and June 2016. However, only 34 eyes (85%) with complete SRF resorption 3 months after HD PDT had their 6th and 12th month data analyzed. Morphological integrity of the ORLs was further analyzed in relation to best-corrected visual acuity (BCVA) and disease duration. Thirty-four eyes of 34 patients (male/female: 82.35/17.65%) with mean age of 49.90 ± 7.80 (32–61) years were studied. The mean logMAR BCVA improved significantly from 0.52 ± 0.31 at baseline to 0.34 ± 0.36 and 0.26 ± 0.26 at the 6th and 12th months after HD PDT, respectively (p < 0.001). The proportion of eyes with completely normal morphological ultrastructural integrity of the ORLs was 44.12% at the 6th month, which increased to 64.71% at the 12th month after HD PDT. However, the EL morphological disruption was associated with significantly lower mean logMAR BCVA 12 months after HD PDT (p = 0.029). The disease duration had no effect on mean logMAR BCVA gain. Even after complete resorption of serous neurosensory retinal detachment after HD PDT in chronic CSCR, the ORLs, especially the EL, may not be anatomically restored. The EL morphological ultrastructural integrity seems to be the most important factor influencing visual prognosis.

Similar content being viewed by others
Data availability
Data are available from the corresponding author on reasonable request.
References
Yannuzzi LA, Slakter JS, Gross NE, Spaide RF, Costa D, Huang SJ, Klancnik JM Jr, Aizman A (2003) Indocyanine green angiography-guided photodynamic therapy for treatment of chronic central serous chorioretinopathy: a pilot study. Retina 23:288–298. https://doi.org/10.1097/00006982-200306000-00002
Yannuzzi LA (1986) Type A behavior and central serous chorioretinopathy. Trans Am Ophthalmol Soc 84:799–845
Piccolino FC, de la Longrais RR, Ravera G, Eandi CM, Ventre L, Abdollahi A, Manea M (2005) The foveal photoreceptor layer and visual acuity loss in central serous chorioretinopathy. Am J Ophthalmol 139:87–99. https://doi.org/10.1016/j.ajo.2004.08.037
Loo RH, Scott IU, Flynn HW Jr, Gass JD, Murray TG, Lewis ML, Rosenfeld PJ, Smiddy WE (2002) Factors associated with reduced visual acuity during long-term follow-up of patients with idiopathic central serous chorioretinopathy. Retina 22:19–24. https://doi.org/10.1097/00006982-200202000-00004
Tan CS, Cheong KX, Sadda SR (2014) Changes in choroidal thickness after photodynamic therapy in patients with central serous chorioretinopathy. Acta Ophthalmol 92:e79. https://doi.org/10.1111/aos.12296
Lee ST, Adelman RA (2011) The treatment of recurrent central serous chorioretinopathy with intravitreal bevacizumab. J Ocul Pharmacol Ther 27:611–614. https://doi.org/10.1089/jop.2011.0045
Bae SH, Heo JW, Kim C, Kim TW, Lee JY, Song SJ, Park TK, Moon SW, Chung H (2011) A randomized pilot study of low-fluence photodynamic therapy versus intravitreal ranibizumab for chronic central serous chorioretinopathy. Am J Ophthalmol 152:784-792.e2. https://doi.org/10.1016/j.ajo.2011.04.008
Koss MJ, Beger I, Koch FH (2012) Subthreshold diode laser micropulse photocoagulation versus intravitreal injections of bevacizumab in the treatment of central serous chorioretinopathy. Eye (Lond) 26:307–314. https://doi.org/10.1038/eye.2011.282
Iacono P, Toto L, Costanzo E, Varano M, Parravano MC (2018) Pharmacotherapy of central serous chorioretinopathy: a review of the current treatments. Curr Pharm Des 24:4864–4873. https://doi.org/10.2174/1381612825666190123165914
Taban M, Boyer DS, Thomas EL, Taban M (2004) Chronic central serous chorioretinopathy: photodynamic therapy. Am J Ophthalmol 137:1073–1080. https://doi.org/10.1016/j.ajo.2004.01.043
Vasconcelos H, Marques I, Santos AR, Melo P, Pires I, Figueira J, de Abreu JF, Cachulo ML, Silva R (2013) Long-term chorioretinal changes after photodynamic therapy for chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 251:1697–1705. https://doi.org/10.1007/s00417-013-2270-2
Cardillo Piccolino F, Eandi CM, Ventre L, Rigault de la Longrais RC, Grignolo FM (2003) Photodynamic therapy for chronic central serous chorioretinopathy. Retina 23:752–763. https://doi.org/10.1097/00006982-200312000-00002
Michels S, Hansmann F, Geitzenauer W, Schmidt-Erfurth U (2006) Influence of treatment parameters on selectivity of verteporfin therapy. Invest Ophthalmol Vis Sci 47:371–376. https://doi.org/10.1167/iovs.05-0354
Cheng CK, Chang CK, Peng CH (2017) Comparison of photodynamic therapy using half-dose of verteporfin or half-fluence of laser light for the treatment of chronic central serous chorioretinopathy. Retina 37:325–333. https://doi.org/10.1097/IAE.0000000000001138
Tsai MJ, Hsieh YT (2014) Half-time photodynamic therapy for central serous chorioretinopathy. Optom Vis Sci 91:1140–1145. https://doi.org/10.1097/OPX.0000000000000360
Shin JY, Woo SJ, Yu HG, Park KH (2011) Comparison of efficacy and safety between half-fluence and full-fluence photodynamic therapy for chronic central serous chorioretinopathy. Retina 31:119–126. https://doi.org/10.1097/IAE.0b013e3181e378f2
Prünte C, Flammer J (1996) Choroidal capillary and venous congestion in central serous chorioretinopathy. Am J Ophthalmol 121:26–34. https://doi.org/10.1016/s0002-9394(14)70531-8
Ozdemir I, Eren A, Ersöz G (2019) Outer nuclear layer thickness at the central fovea relation with symptom duration in central serous chorioretinopathy. Int Ophthalmol 39:1323–1328. https://doi.org/10.1007/s10792-018-0950-y
Gemenetzi M, De Salvo G, Lotery AJ (2010) Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye (Lond) 24:1743–1756. https://doi.org/10.1038/eye.2010.130
Tsai DC, Chen SJ, Huang CC, Chou P, Chung CM, Huang PH, Lin SJ, Chen JW, Chen TJ, Leu HB, Chan WL (2013) Epidemiology of idiopathic central serous chorioretinopathy in Taiwan, 2001–2006: a population-based study. Plos One 8:e66858. https://doi.org/10.1371/journal.pone.0066858
Haga F, Maruko R, Sato C, Kataoka K, Ito Y, Terasaki H (2017) Long-term prognostic factors of chronic central serous chorioretinopathy after half-dose photodynamic therapy: a 3-year follow-up study. Plos One 12:e0181479. https://doi.org/10.1371/journal.pone.0181479
Ruiz-Moreno JM, Lugo FL, Armadá F, Silva R, Montero JA, Arevalo JF, Arias L, Gómez-Ulla F (2010) Photodynamic therapy for chronic central serous chorioretinopathy. Acta Ophthalmol 88:371–376. https://doi.org/10.1111/j.1755-3768.2008.01408.x
Lai FH, Ng DS, Bakthavatsalam M, Chan VC, Young AL, Luk FO, Tsang CW, Brelén ME (2016) A multicenter study on the long-term outcomes of half-dose photodynamic therapy in chronic central serous chorioretinopathy. Am J Ophthalmol 170:91–99. https://doi.org/10.1016/j.ajo.2016.07.026
Yu J, Jiang C, Xu G (2019) Correlations between changes in photoreceptor layer and other clinical characteristics in central serous chorioretinopathy. Retina 39:1110–1116. https://doi.org/10.1097/IAE.0000000000002092
Moon JW, Yu HG, Kim TW, Kim HC, Chung H (2009) Prognostic factors related to photodynamic therapy for central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol 247:1315–1323. https://doi.org/10.1007/s00417-009-1104-8
Ohkuma Y, Hayashi T, Sakai T, Watanabe A, Tsuneoka H (2013) One-year results of reduced fluence photodynamic therapy for central serous chorioretinopathy: the outer nuclear layer thickness is associated with visual prognosis. Graefes Arch Clin Exp Ophthalmol 251:1909–1917. https://doi.org/10.1007/s00417-013-2289-4
Matušková V, Vysloužilová D, Uher M (2018) Half-fluence photodynamic therapy for chronic central serous chorioretinopathy: predisposing factors for visual acuity outcomes. Semin Ophthalmol 33:690–699. https://doi.org/10.1080/08820538.2017.1416414
Chan WM, Lai TY, Lai RY, Tang EW, Liu DT, Lam DS (2008) Safety enhanced photodynamic therapy for chronic central serous chorioretinopathy: one-year results of a prospective study. Retina 28:85–93. https://doi.org/10.1097/IAE.0b013e318156777f
Serizawa S, Ohkoshi K, Minowa Y, Soejima K (2016) Interdigitation zone band restoration after treatment of diabetic macular edema. Curr Eye Res 41:1229–1234. https://doi.org/10.3109/02713683.2015.1113430
Hata M, Oishi A, Shimozono M, Mandai M, Nishida A, Kurimoto Y (2013) Early changes in foveal thickness in eyes with central serous chorioretinopathy. Retina 33:296–301. https://doi.org/10.1097/IAE.0b013e31826710a0
Matsumoto H, Kishi S, Otani T, Sato T (2008) Elongation of photoreceptor outer segment in central serous chorioretinopathy. Am J Ophthalmol 145:162–168. https://doi.org/10.1016/j.ajo.2007.08.024
Torres-Costa S, Penas S, Cerqueira AR, Brandão E, Carneiro Â, Rocha-Sousa A, Falcão-Reis F (2021) Long term outer retinal changes in central serous chorioretinopathy submitted to half-dose photodynamic therapy. Photodiagnosis Photodyn Ther 34:102235. https://doi.org/10.1016/j.pdpdt.2021.102235
Nicoló M, Eandi CM, Alovisi C, Grignolo FM, Traverso CE, Musetti D, Cardillo Piccolino F (2014) Half-fluence versus half-dose photodynamic therapy in chronic central serous chorioretinopathy. Am J Ophthalmol 157:1033–1037. https://doi.org/10.1016/j.ajo.2014.01.022
Altinel MG, Kanra AY, Totuk OMG, Ardagil A, Kabadayi K (2021) Comparison of half-dose versus half-fluence versus standard photodynamic therapy in chronic central serous chorioretinopathy. Photodiagnosis Photodyn Ther 33:102081. https://doi.org/10.1016/j.pdpdt.2020.102081
Lee JH, Lee SC, Lee CS (2020) Comparison of half-time and half-irradiance photodynamic therapy in nonresolving central serous chorioretinopathy. J Ocul Pharmacol Ther 36:109–115. https://doi.org/10.1089/jop.2019.0003
Alkin Z, Perente I, Ozkaya A, Alp D, Agca A, Aygit ED, Korkmaz S, Yazici AT, Demirok A (2014) Comparison of efficacy between low-fluence and half-dose verteporfin photodynamic therapy for chronic central serous chorioretinopathy. Clin Ophthalmol 8:685–690. https://doi.org/10.2147/OPTH.S58617
Erikitola OC, Crosby-Nwaobi R, Lotery AJ, Sivaprasad S (2014) Photodynamic therapy for central serous chorioretinopathy. Eye (Lond) 28:944–957. https://doi.org/10.1038/eye.2014.134
Reibaldi M, Boscia F, Avitabile T, Russo A, Cannemi V, Uva MG, Reibaldi A (2009) Low-fluence photodynamic therapy in longstanding chronic central serous chorioretinopathy with foveal and gravitational atrophy. Eur J Ophthalmol 19:154–158. https://doi.org/10.1177/112067210901900126
Ziemssen F, Heimann H (2012) Evaluation of verteporfin pharmakokinetics—redefining the need of photosensitizers in ophthalmology. Expert Opin Drug Metab Toxicol 8:1023–1041. https://doi.org/10.1517/17425255.2012.701617
Wang MS, Sander B, Larsen M (2002) Retinal atrophy in idiopathic central serous chorioretinopathy. Am J Ophthalmol 133:787–793. https://doi.org/10.1016/s0002-9394(02)01438-1
Funding
The authors declare that they have received no public or private financial support or involvement in the products, methods, or materials mentioned in this manuscript.
Author information
Authors and Affiliations
Contributions
Study design and recruitment of participants: JM, HHG, SN, and YC; photodynamic therapy and follow-up of patients: JM and SN; data analysis: HHG, ZO, JM, and YC; reviewing, editing, and verifying the accuracy of the manuscript: HHG, JM, SN, and ZO; complete access to all study data and accountability for data integrity and accuracy of data analysis: JM, HHG, SN, ZO, and YC. HHG = Hamidu Hamisi Gobeka; JM = Jale Mentes; SN = Serhad Nalcaci; ZO = Zafer Oztas; and YC = Yigit Cay.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the principles of the Helsinki Declaration and all applicable regulations, and approved by Ege University Faculty of Medicine Ethics Committee (Approval Number: 02.06.2021: 21–5.1 T/55). Each participant signed a written informed consent form.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
The authors note that human study participants have given informed consent to the release of the images.
Competing interests
The authors declare no competing interests.
Financial interest
All authors certify that they have no association or participation with any organization or individual with any financial interest or non-financial interest in the subject matter or materials discussed in this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Prior presentation
The study manuscript has not been published or is not being considered for publication elsewhere. Its abstract, however, was accepted and orally presented at the 50th Turkish Ophthalmology Society (TOD) Congress, 9-13 November 2016, Antalya, Turkey, under the ID: SS-TR-37.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gobeka, H.H., Mentes, J., Nalcaci, S. et al. Morphological integrity of the outer retinal layers and visual prognosis in chronic central serous chorioretinopathy after half-dose photodynamic therapy: a qualitative SD-OCT analysis. Lasers Med Sci 38, 9 (2023). https://doi.org/10.1007/s10103-022-03676-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-022-03676-z