Abstract
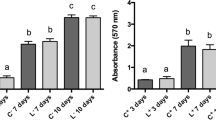
This study aimed to evaluate the effect of different photobiomodulation (PBM) radiant exposures on the viability, proliferation, and gene expression of pulp fibroblasts from human primary teeth (HPF) involved in the pulp tissue repair. HPF were irradiated with Laser InGaAlP (Twin Flex Evolution, MMOptics®) at 660-nm wavelength (red); single time, continuous mode, 0.04-cm2 laser tip area, and 0.225-cm laser tip diameter, keeping the distance of 1 mm between the laser beam and the cell culture. The doses used were between 1.2 and 6.2 J/cm2 and were evaluated at the 6 h, 12 h, and 24 h after PBM. MTT and crystal violet assays evaluated the cell viability and proliferation. RT-PCR verified VEGF and FGF-2 mRNA expression. A blinded examiner analyzed the data through two-way ANOVA followed by Tukey test (p < 0.05). The groups with higher powers (10 mW, 15 mW, 20 mW, and 25 mW), shortest application periods (10 s), and radiant exposures between 2.5 and 6.2 J/cm2 exhibited statistically higher viability than that of the groups with small power (5 mW), longer application period (50 s), and radiant exposure of 6.2 J/cm2 (p < 0.05). VEGF and FGF-2 mRNA expression were observed at the three evaluated periods (6 h, 12 h, and 24 h) and the highest expression was in the shortest period (p < 0.05). All radiant exposures maintained HPF viable. The period of 6 h after irradiation showed statistically greater gene expression for both growth factors than other periods. VEGF mRNA had no differences among the dosimetries studied. The best radiant exposures for FGF-2 gene expression were 2.5 J/cm2 and 3.7 J/cm2.

Similar content being viewed by others
References
Grando Mattuella L, Westphalen Bento L, de Figueiredo JA, Nor JE, de Araujo FB, Fossati AC (2007) Vascular endothelial growth factor and its relationship with the dental pulp. J Endod 33:524–530. https://doi.org/10.1016/j.joen.2007.01.003
Jeanneau C, Lundy FT, El Karim IA, About I (2017) Potential therapeutic strategy of targeting pulp fibroblasts in dentin-pulp regeneration. J Endod 43:S17–S24. https://doi.org/10.1016/j.joen.2017.06.007
Cooper PR, Holder MJ, Smith AJ (2014) Inflammation and regeneration in the dentin-pulp complex: a double-edged sword. J Endod 40:S46–S51. https://doi.org/10.1016/j.joen.2014.01.021
Moretti AB, Sakai VT, Oliveira TM, Fornetti AP, Santos CF, Machado MA et al (2008) The effectiveness of mineral trioxide aggregate, calcium hydroxide and formocresol for pulpotomies in primary teeth. Int Endod J 41:547–555. https://doi.org/10.1111/j.1365-2591.2008.01377.x
Oliveira TM, Moretti AB, Sakai VT, Lourenco Neto N, Santos CF, Machado MA et al (2013) Clinical, radiographic and histologic analysis of the effects of pulp capping materials used in pulpotomies of human primary teeth. Eur Arch Paediatr Dent 14:65–71. https://doi.org/10.1007/s40368-013-0015-x
Taylor GD, Vernazza CR, Abdulmohsen B (2019) Success of endodontic management of compromised first permanent molars in children: a systematic review. Int J Paediatr Dent. https://doi.org/10.1111/ipd.12599
American Academy of Pediatric Dentistry (AAPD) (2014) Reference manual: pulp therapy for primary and immature permanent teeth. 44(6):353–361 Accessed October 8, 2020
Tziafas D, Kodonas K (2010) Differentiation potential of dental papilla, dental pulp, and apical papilla progenitor cells. J Endod 36:781–789. https://doi.org/10.1016/j.joen.2010.02.006
Kim JH, Jeon M, Song JS, Lee JH, Choi BJ, Jung HS et al (2014) Distinctive genetic activity pattern of the human dental pulp between deciduous and permanent teeth. PLoS One 9:e102893. https://doi.org/10.1371/journal.pone.0102893
Marques NC, Neto NL, Rodini Cde O, Fernandes AP, Sakai VT, Machado MA et al (2015) Low-level laser therapy as an alternative for pulpotomy in human primary teeth. Lasers Med Sci 30:1815–1822. https://doi.org/10.1007/s10103-014-1656-7
Niranjani K, Prasad MG, Vasa AA, Divya G, Thakur MS, Saujanya K (2015) Clinical evaluation of success of primary teeth pulpotomy using mineral trioxide aggregate(R), laser and biodentine(TM)—an in vivo study. J Clin Diagn Res 9:ZC35–ZC37. https://doi.org/10.7860/JCDR/2015/13153.5823
Marques NCT, Neto NL, Prado MTO, Vitor LLR, Oliveira RC, Sakai VT et al (2017) Effects of PBM in different energy densities and irradiance on maintaining cell viability and proliferation of pulp fibroblasts from human primary teeth. Lasers Med Sci 32:1621–1628. https://doi.org/10.1007/s10103-017-2301-z
Prado MTO, Vitor LLR, Lourenço Neto N, Marques NCT, Sakai VT, Rios D, Cruvinel T, Oliveira RC, Santos CF, Machado MAAM, Oliveira TM (2019) Effects of different culture media, cell densities and adhesion periods on stem cells from human exfoliated deciduous teeth after photobiomodulation. Laser Phys Lett 16:095601. https://doi.org/10.1088/1612-202X/ab3452
Moore P, Ridgway TD, Higbee RG, Howard EW, Lucroy MD (2005) Effect of wavelength on low-intensity laser irradiation-stimulated cell proliferation in vitro. Lasers Surg Med 36:8–12. https://doi.org/10.1002/lsm.20117
Vitor LLR, Prado MTO, Lourenço Neto N, Oliveira RC, Santos CF, Machado MAAM, Oliveira TM (2018) Photobiomodulation changes type 1 collagen gene expression by pulp fibroblasts. Laser Phys 28:065603. https://doi.org/10.1088/1555-6611/aabd16
Saygun I, Karacay S, Serdar M, Ural AU, Sencimen M, Kurtis B (2008) Effects of laser irradiation on the release of basic fibroblast growth factor (bFGF), insulin like growth factor-1 (IGF-1), and receptor of IGF-1 (IGFBP3) from gingival fibroblasts. Lasers Med Sci 23:211–215. https://doi.org/10.1007/s10103-007-0477-3
Oliveira Prado Bergamo MT, Vitor LLR, Lopes NM, Lourenco Neto N, Dionisio TJ, Oliveira RC et al (2020) Angiogenic protein synthesis after photobiomodulation therapy on SHED: a preliminary study. Lasers Med Sci. https://doi.org/10.1007/s10103-020-02975-7
Ginani F, Soares DM, Barreto MP, Barboza CA (2015) Effect of low-level laser therapy on mesenchymal stem cell proliferation: a systematic review. Lasers Med Sci 30:2189–2194. https://doi.org/10.1007/s10103-015-1730-9
Goldberg M, Njeh A, Uzunoglu E (2015) Is pulp inflammation a prerequisite for pulp healing and regeneration? Mediat Inflamm 2015:347649. https://doi.org/10.1155/2015/347649
Rombouts C, Giraud T, Jeanneau C, About I (2017) Pulp vascularization during tooth development, regeneration, and therapy. J Dent Res 96:137–144. https://doi.org/10.1177/0022034516671688
Shimabukuro Y, Ueda M, Ozasa M, Anzai J, Takedachi M, Yanagita M et al (2009) Fibroblast growth factor-2 regulates the cell function of human dental pulp cells. J Endod 35:1529–1535. https://doi.org/10.1016/j.joen.2009.08.010
Fernandes AP, Lourenco Neto N, Teixeira Marques NC, Silveira Moretti AB, Sakai VT, Cruvinel Silva T et al (2015) Clinical and radiographic outcomes of the use of low-level laser therapy in vital pulp of primary teeth. Int J Paediatr Dent 25:144–150. https://doi.org/10.1111/ipd.12115
Ansari G, Safi Aghdam H, Taheri P, Ghazizadeh Ahsaie M (2018) Laser pulpotomy—an effective alternative to conventional techniques—a systematic review of literature and meta-analysis. Lasers Med Sci 33:1621–1629. https://doi.org/10.1007/s10103-018-2588-4
Fernandes A, Marques NCT, Vitor LLR, Prado MTO, Oliveira RC, Machado MAAM, Oliveira TM (2018) Cellular response of pulp fibroblast to single or multiple photobiomodulation applications. Laser Phys 28:065604. https://doi.org/10.1088/1555-6611/aab952
Volpato LER, Oliveira RC, Espinosa MM, Bagnato VS, Machado MAAM (2011) Viability of fibroblasts cultured under nutritional stress irradiated with red laser, infrared laser, and red light emitting diode. J Biomed Opt 16:075004. https://doi.org/10.1117/1.3602850
Souza LM, Rinco UG, Aguiar DAC, de Almeida Junior LA, Silva LC, Oliveira TM, Sakai VT, Marques NCT (2018) Effect of low-level laser therapy on viability and proliferation of stem cells from exfoliated deciduous teeth under different nutritional conditions. Laser Phys 28:025603 (5pp). https://doi.org/10.1088/1555-6611/aa8e79
Ferreira LS, Diniz IMA, Maranduba CMS, Miyagi SPH, Rodrigues M, Moura-Netto C, Marques MM (2019) Short-term evaluation of photobiomodulation therapy on the proliferation and undifferentiated status of dental pulp stem cells. Lasers Med Sci 34(4):659–666. https://doi.org/10.1007/s10103-018-2637-z
Marques NP, Lopes CS, Marques NCT, Cosme-Silva L, Oliveira TM et al (2019) A preliminary comparison between the effects of red and infrared laser irradiation on viability and proliferation of SHED. Lasers Med Sci 34(3):465–471
Silva PCS, Marques NP, Farina MT, Oliveira TM, Duque C et al (2019) Laser treatment contributes to maintain membrane integrity in stem cells from human exfoliated deciduous teeth (shed) under nutritional deficit. Lasers Med Sci 34(1):15–21
Almeida-Junior LA, Marques NCT, Prado MTO, Oliveira TM, Sakai VT (2019) Effect of single and multiple doses of low-level laser therapy on viability and proliferation of stem cells from human exfoliated deciduous teeth (SHED). Lasers Med Sci 34:1917. https://doi.org/10.1007/s10103-019-02836-y
Almeida-Lopes L, Rigau J, Zangaro RA, Guidugli-Neto J, Jaeger MM (2001) Comparison of the low level laser therapy effects on cultured human gingival fibroblasts proliferation using different irradiance and same fluence. Lasers Surg Med 29:179–184. https://doi.org/10.1002/lsm.1107
Eduardo Fde P, Bueno DF, de Freitas PM, Marques MM, Passos-Bueno MR, Eduardo C de P, et al. (2008) Stem cell proliferation under low intensity laser irradiation: a preliminary study. 40:433-438. DOI: https://doi.org/10.1002/lsm.20646
Theocharidou A, Bakopoulou A, Kontonasaki E, Papachristou E, Hadjichristou C, Bousnaki M et al (2017) Odontogenic differentiation and biomineralization potential of dental pulp stem cells inside Mg-based bioceramic scaffolds under low-level laser treatment. Lasers Med Sci 32:201–210. https://doi.org/10.1007/s10103-016-2102-9
El Nawam H, El Backly R, Zaky A, Abdallah A (2019) Low-level laser therapy affects dentinogenesis and angiogenesis of in vitro 3D cultures of dentin-pulp complex. Lasers Med Sci 34:1689–1698. https://doi.org/10.1007/s10103-019-02804-6
Karu TI (2008) Mitochondrial signaling in mammalian cells activated by red and near-IR radiation. Photochem Photobiol 84:1091–1099. https://doi.org/10.1111/j.1751-1097.2008.00394.x
AlGhamdi KM, Kumar A, Moussa NA (2012) Low-level laser therapy: a useful technique for enhancing the proliferation of various cultured cells. Lasers Med Sci 27:237–249. https://doi.org/10.1007/s10103-011-0885-2
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Gillies RJ, Didier N, Denton M (1986) Determination of cell number in monolayer cultures. Anal Biochem 159:109–113. https://doi.org/10.1016/0003-2697(86)90314-3
Karu T (1999) Primary and secondary mechanisms of action of visible to near-IR radiation on cells. J Photochem Photobiol B 49:1–17. https://doi.org/10.1016/S1011-1344(98)00219-X
Farivar S, Malekshahabi T, Shiari R (2014) Biological effects of low level laser therapy. J Lasers Med Sci 5:58–62. https://doi.org/10.22037/jlms.v5i2.5540
Prindeze NJ, Moffatt LT, Shupp JW (2012) Mechanisms of action for light therapy: a review of molecular interactions. Exp Biol Med (Maywood) 237:1241–1248. https://doi.org/10.1258/ebm.2012.012180
Szezerbaty SKF, de Oliveira RF, Pires-Oliveira DAA, Soares CP, Sartori D, Poli-Frederico RC (2018) The effect of low-level laser therapy (660 nm) on the gene expression involved in tissue repair. Lasers Med Sci 33:315–321. https://doi.org/10.1007/s10103-017-2375-7
Yanagita M, Kojima Y, Kubota M, Mori K, Yamashita M, Yamada S et al (2014) Cooperative effects of FGF-2 and VEGF-A in periodontal ligament cells. J Dent Res 93:89–95. https://doi.org/10.1177/0022034513511640
Funding
This study was funded by State of São Paulo Research Foundation (Grant #2015-19696-0; #2017/11396-3; #2018/20316-6).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards (protocol CAAE 53785716.6.0000.5417).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bergamo, M.T., Vitor, L.L.R., Dionísio, T.J. et al. Could the photobiomodulation therapy induce angiogenic growth factors expression from dental pulp cells?. Lasers Med Sci 36, 1751–1758 (2021). https://doi.org/10.1007/s10103-021-03291-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-021-03291-4