Abstract
Introduction
Studies regarding multiple chronic lacunes (MCLs) and clinical outcome according to stroke etiology are scarce. We sought to evaluate the association between MCL and short-term/long-term clinical outcomes according to stroke etiology.
Patients and methods
We analyzed a prospectively collected stroke registry of acute ischemic stroke patients over 4 years. The enrolled patients were classified as having large artery atherosclerosis (LAA), small vessel occlusion (SVO), cardioembolic (CE) stroke, and other etiology. The early neurological deterioration (END) and favorable outcome at 3 months were assessed.
Results
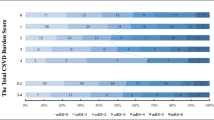
A total of 1070 patients were enrolled. Patients with MCL had significantly more END compared to those without MCL both in total population (adjusted odds ratio (OR), 1.7; 95% confidence interval [CI], 1.1–2.5; p = 0.013*) and in the LAA group (adjusted OR, 2.3; 95% CI, 1.3–4.2, p < 0.006). Patients with MCL had a significantly lower OR for favorable outcome at 3 months compared to those without MCL both in total population (adjusted OR, 0.7; 95% CI, 0.5–1.0, p = 0.035) and in the LAA group (adjusted OR, 0.6; 95% CI, 0.3–1.0, p = 0.043). However, MCL was not associated with END or long-term functional outcome in patients with SVO, CE, or other etiology.
Conclusions
The presence of MCL was an independent predictive factor for END as well as long-term poor functional outcome in acute ischemic stroke patients. These associations were only observed in patients with LAA, not in those with SVO, CE, or other etiology.



Similar content being viewed by others
Data availability
The de-identified data supporting the findings of this study are available upon reasonable request to the corresponding author.
References
Appleton JP, Woodhouse LJ, Adami A, Becker JL, Berge E, Cala LA, Casado AM, Caso V, Christensen HK, Dineen RA, Gommans J, Koumellis P, Szatmari S, Sprigg N, Bath PM, Wardlaw JM, Investigators E (2020) Imaging markers of small vessel disease and brain frailty, and outcomes in acute stroke. Neurology 94(5):e439–e452. https://doi.org/10.1212/WNL.0000000000008881
Arba F, Inzitari D, Ali M, Warach SJ, Luby M, Lees KR, Collaboration SVI (2017) Small vessel disease and clinical outcomes after IV rt-PA treatment. Acta Neurol Scand 136(1):72–77. https://doi.org/10.1111/ane.12745
Arba F, Palumbo V, Boulanger JM, Pracucci G, Inzitari D, Buchan AM, Hill MD, Investigators C (2016) Leukoaraiosis and lacunes are associated with poor clinical outcomes in ischemic stroke patients treated with intravenous thrombolysis. Int J Stroke 11(1):62–67. https://doi.org/10.1177/1747493015607517
Bu N, Khlif MS, Lemmens R, Wouters A, Fiebach JB, Chamorro A, Ringelstein EB, Norrving B, Laage R, Grond M, Wilms G, Brodtmann A, Thijs V (2021) Imaging markers of brain frailty and outcome in patients with acute ischemic stroke. Stroke 52(3):1004–1011. https://doi.org/10.1161/STROKEAHA.120.029841
Duan Y, Chen F, Lin L, Wei W, Huang Y (2015) Leukoaraiosis rather than lacunes predict poor outcome and chest infection in acute ischemic stroke patients. Int J Clin Exp Med 8(10):19304–19310
Griessenauer CJ, McPherson D, Berger A, Cuiper P, Sofoluke N, Adams MD, Kunaprayoon S, Zand R, Li J, Abedi V, Goren O, Schirmer CM, Donahue K, Nardin M, Giese AK, Schirmer MD, Rost NS, Hendrix P (2020) Effects of white matter hyperintensities on 90-day functional outcome after large vessel and non-large vessel stroke. Cerebrovascular diseases (Basel, Switzerland) 49(4):419–426. https://doi.org/10.1159/000509071
Huo YC, Li Q, Zhang WY, Zou N, Li R, Huang SY, Wang HQ, Song KY, Zhang RR, Qin XY (2019) Total small vessel disease burden predicts functional outcome in patients with acute ischemic stroke. Front Neurol 10:808. https://doi.org/10.3389/fneur.2019.00808
Ryu WS, Jeong SW, Kim DE (2020) Total small vessel disease burden and functional outcome in patients with ischemic stroke. PLoS One 15(11):e0242319. https://doi.org/10.1371/journal.pone.0242319
Xu T, Wang Y, Yuan J, Chen Y, Luo H (2021) Small vessel disease burden and outcomes of mechanical thrombectomy in ischemic stroke: a systematic review and meta-analysis. Front Neurol 12:602037. https://doi.org/10.3389/fneur.2021.602037
Reijmer YD, Freeze WM, Leemans A, Biessels GJ, Utrecht Vascular Cognitive Impairment Study G (2013) The effect of lacunar infarcts on white matter tract integrity. Stroke 44(7):2019–2021. https://doi.org/10.1161/STROKEAHA.113.001321
Pierpaoli C, Barnett A, Pajevic S, Chen R, Penix LR, Virta A, Basser P (2001) Water diffusion changes in Wallerian degeneration and their dependence on white matter architecture. Neuroimage 13(6 Pt 1):1174–1185. https://doi.org/10.1006/nimg.2001.0765
Munoz DG (1991) The pathological basis of multi-infarct dementia. Alzheimer Dis Assoc Disord 5(2):77–90. https://doi.org/10.1097/00002093-199100520-00005
Adams HP Jr, Davis PH, Leira EC, Chang KC, Bendixen BH, Clarke WR, Woolson RF, Hansen MD (1999) Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology 53(1):126–131. https://doi.org/10.1212/wnl.53.1.126
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, 3rd Marsh EE (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24(1):35–41. https://doi.org/10.1161/01.str.24.1.35
Kang DW, Chalela JA, Ezzeddine MA, Warach S (2003) Association of ischemic lesion patterns on early diffusion-weighted imaging with TOAST stroke subtypes. Arch Neurol 60(12):1730–1734. https://doi.org/10.1001/archneur.60.12.1730
Bruno A, Shah N, Lin C, Close B, Hess DC, Davis K, Baute V, Switzer JA, Waller JL, Nichols FT (2010) Improving modified Rankin Scale assessment with a simplified questionnaire. Stroke 41(5):1048–1050. https://doi.org/10.1161/STROKEAHA.109.571562
Helleberg BH, Ellekjær H, Rohweder G, Indredavik B (2014) Mechanisms, predictors and clinical impact of early neurological deterioration: the protocol of the Trondheim early neurological deterioration study. BMC Neurol 14(1):201. https://doi.org/10.1186/s12883-014-0201-4
Fisher CM (1982) Lacunar strokes and infarcts: a review. Neurology 32(8):871–876. https://doi.org/10.1212/wnl.32.8.871
Regenhardt RW, Das AS, Lo EH, Caplan LR (2018) Advances in understanding the pathophysiology of lacunar stroke: a review. JAMA Neurol 75(10):1273–1281. https://doi.org/10.1001/jamaneurol.2018.1073
Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, Lindley RI, O’Brien JT, Barkhof F, Benavente OR, Black SE, Brayne C, Breteler M, Chabriat H, Decarli C, de Leeuw FE, Doubal F, Duering M, Fox NC, Greenberg S, Hachinski V, Kilimann I, Mok V, Oostenbrugge R, Pantoni L, Speck O, Stephan BC, Teipel S, Viswanathan A, Werring D, Chen C, Smith C, van Buchem M, Norrving B, Gorelick PB, Dichgans M, nEuroimagingSTfRVco (2013) Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol 12(8):822–838. https://doi.org/10.1016/S1474-4422(13)70124-8
Miyamoto N, Tanaka R, Ueno Y, Watanabe M, Kurita N, Hira K, Shimada Y, Kuroki T, Yamashiro K, Urabe T, Hattori N (2017) Analysis of the usefulness of the WORSEN Score for predicting the deterioration of acute ischemic stroke. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 26(12):2834–2839. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.07.005
Alawneh JA, Moustafa RR, Baron JC (2009) Hemodynamic factors and perfusion abnormalities in early neurological deterioration. Stroke 40(6):e443-450. https://doi.org/10.1161/STROKEAHA.108.532465
Gwak DS, Kwon JA, Shim DH, Kim YW, Hwang YH (2021) Perfusion and diffusion variables predict early neurological deterioration in minor stroke and large vessel occlusion. J Stroke 23(1):61–68. https://doi.org/10.5853/jos.2020.01466
Seners P, Ben Hassen W, Lapergue B, Arquizan C, Heldner MR, Henon H, Perrin C, Strambo D, Cottier JP, Sablot D, Girard Buttaz I, Tamazyan R, Preterre C, Agius P, Laksiri N, Mechtouff L, Bejot Y, Duong DL, Mounier-Vehier F, Mione G, Rosso C, Lucas L, Papassin J, Aignatoaie A, Triquenot A, Carrera E, Niclot P, Obadia A, Lyoubi A, Garnier P, Crainic N, Wolff V, Tracol C, Philippeau F, Lamy C, Soize S, Baron JC, Turc G, Collaborators M-S (2021) Prediction of early neurological deterioration in individuals with minor stroke and large vessel occlusion intended for intravenous thrombolysis alone. JAMA Neurol 78(3):321–328. https://doi.org/10.1001/jamaneurol.2020.4557
Sung SM, Kang YJ, Cho HJ, Kim NR, Lee SM, Choi BK, Cho G (2020) Prediction of early neurological deterioration in acute minor ischemic stroke by machine learning algorithms. Clin Neurol Neurosurg 195:105892. https://doi.org/10.1016/j.clineuro.2020.105892
Xie X, Xiao J, Wang Y, Pan L, Ma J, Deng L, Yang J, Ren L (2021) Predictive model of early neurological deterioration in patients with acute ischemic stroke: a retrospective cohort study. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 30(3):105459. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105459
Davalos A, Cendra E, Teruel J, Martinez M, Genis D (1990) Deteriorating ischemic stroke: risk factors and prognosis. Neurology 40(12):1865–1869. https://doi.org/10.1212/wnl.40.12.1865
Kwan J, Hand P (2006) Early neurological deterioration in acute stroke: clinical characteristics and impact on outcome. QJM 99(9):625–633. https://doi.org/10.1093/qjmed/hcl082
Appleton JP, Woodhouse LJ, Adami A, Becker JL, Berge E, Cala LA, Casado AM, Caso V, Christensen HK, Dineen RA, Gommans J, Koumellis P, Szatmari S, Sprigg N, Bath PM, Wardlaw JM (2020) Imaging markers of small vessel disease and brain frailty, and outcomes in acute stroke. Neurology 94(5):e439–e452. https://doi.org/10.1212/wnl.0000000000008881
Author information
Authors and Affiliations
Contributions
HL, TK, and JK contributed to the conception and design of the study. YDK, SN, YHC, IU, and SWC contributed to the analysis of the data. HL and TK contributed to drafting the text and preparing the tables and figure.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee and the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Conflict of interest
None.
Informed consent
All aspects of this retrospective study were approved by the Institutional Review Board of The Catholic University of Korea (IRB). The requirement for informed consent was formally waived by the IRB due to the retrospective nature of the study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, Hj., Kim, T., Koo, J. et al. Multiple chronic lacunes predicting early neurological deterioration and long-term functional outcomes according to TOAST classification in acute ischemic stroke. Neurol Sci 44, 611–619 (2023). https://doi.org/10.1007/s10072-022-06446-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06446-5