Abstract
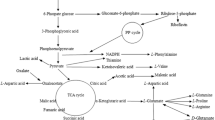
This study was to establish an integrated process for the co-production of γ-aminobutyric acid (GABA) and live probiotics. Six probiotic bacteria were screened and Bacillus subtilis ATCC 6051 showed the highest GABA-producing capacity. The optimal temperature and initial pH value for GABA production in B. subtilis were found to be 30 °C and 8.0, respectively. A variety of carbon and nitrogen sources were tested, and potato starch and peptone were the preferred carbon and nitrogen sources for GABA production, respectively. The concentrations of carbon source, nitrogen source and substrate (sodium l-glutamate) were then optimized using the response surface methodology. The GABA titer and concentration of viable cells of B. subtilis reached 19.74 g/L and 6.0 × 108 cfu/mL at 120 h. The GABA titer represents the highest production of GABA in B. subtilis. This work thus demonstrates a highly efficient co-production process for GABA and probiotic B. subtilis cells.



Similar content being viewed by others
References
Ay F, Catalkaya EC, Kargi F. A statistical experiment design approach for advanced oxidation of Direct Red azo-dye by photo-Fenton treatment. J. Hazard Mater. 162: 230–236 (2009)
Chen Z, Xie J, Hu MY, Tang J, Shao ZF, Li MH. Protective effects of γ -aminobutyric acid (GABA) on the small intestinal mucosa in heat-stressed Wenchang chicken. J. Anim. Plant Sci. 25: 78–87 (2015)
Cheng JB, Bu DP, Wang JQ, Sun XZ, Pan L, Zhou LY, Liu W. Effects of rumen-protected gamma-aminobutyric acid on performance and nutrient digestibility in heat-stressed dairy cows. J. Dairy Sci. 97: 5599–5607 (2014)
Dhakal R, Bajpai VK, Baek KH. Production of GABA (γ-aminobutyric acid) by microorganisms: a review. Braz. J. Microbiol. 43: 1230–1241 (2012)
Diana M, Quílez J, Rafecas M. Gamma-aminobutyric acid as a bioactive compound in foods: a review. J. Funct Foods 10: 407–420 (2014)
Filotheou A, Nanou K, Papaioannou E, Roukas T, Kotzekidou P, Liakopoulou-Kyriakides M. Application of response surface methodology to improve carotene production from synthetic medium by Blakeslea trispora in submerged fermentation. Food Bioprocess Tech. 5: 1189–1196 (2010)
Gan R-Y, Li H-B, Gunaratne A, Sui Z-Q, Corke H. Effects of fermented edible seeds and their products on human health: bioactive components and bioactivities. Compr. Rev. Food Sci. Food Saf. 16: 489–531 (2017)
Ghasemi S, Ahmadzadeh M. Optimisation of a cost-effective culture medium for the large-scale production of Bacillus subtilis UTB96. Arch. Phytopathol. Plant Protect. 46: 1552–1563 (2013)
Janisiewicz WJ, Korsten L. Biological control of postharvest diseases of fruits. Annu. Rev. Phytopathol. 40: 411–441 (2002)
Li H, Cao Y. Lactic acid bacterial cell factories for gamma-aminobutyric acid. Amino Acids 39: 1107–1116 (2010)
Li MF, Guo SJ, Yang XH, Meng QW, Wei XJ. Exogenous gamma-aminobutyric acid increases salt tolerance of wheat by improving photosynthesis and enhancing activities of antioxidant enzymes. Biol. Plant. 60: 123–131 (2015)
Li Y, Fan Y, Ma Y, Zhang Z, Yue H, Wang L, Li J, Jiao Y. Effects of exogenous γ-aminobutyric acid (GABA) on photosynthesis and antioxidant system in pepper (Capsicum annuum L.) seedlings under low light stress. J. Plant Growth Regul. 36: 436–449 (2017)
Limón RI, Peñas E, Torino MI, Martínez-Villaluenga C, Dueñas M, Frias J. Fermentation enhances the content of bioactive compounds in kidney bean extracts. Food Chem. 172: 343–352 (2015)
Malekzadeh P, Khara J, Heydari R. Alleviating effects of exogenous Gamma-aminobutiric acid on tomato seedling under chilling stress. Physiol. Mol. Biol. Plants 20: 133–137 (2014)
Olmos J, Paniagua-Michel J. Bacillus subtilis a potential probiotic bacterium to formulate functional feeds for aquaculture. J. Microb. Biochem. Technol. 6: 361–365 (2014)
Park JH, Kim IH. Effects of dietary gamma-aminobutyric acid on egg production, egg quality, and blood profiles in layer hens. Vet. Med. 60: 629–634 (2016)
Park KB, Oh SH. Enhancement of gamma-aminobutyric acid production in Chungkukjang by applying a Bacillus subtilis strain expressing glutamate decarboxylase from Lactobacillus brevis. Biotechnol. Lett. 28: 1459–1463 (2006)
Pervaiz I, Ahmad S, Mukhtar MF, Arshad A, Imran M, Mahmood W. Microbial biotransformation of dexamethasone by Bacillus subtilis (ATCC 6051). Pharm. Chem. J. 49: 405–408 (2015)
Pham VD, Lee SH, Park SJ, Hong SH. Production of gamma-aminobutyric acid from glucose by introduction of synthetic scaffolds between isocitrate dehydrogenase, glutamate synthase and glutamate decarboxylase in recombinant Escherichia coli. J. Biotechnol. 207: 52–57 (2015)
Sheng L, Shen D, Luo Y, Sun X, Wang J, Luo T, Zeng Y, Xu J, Deng X, Cheng Y. Exogenous gamma-aminobutyric acid treatment affects citrate and amino acid accumulation to improve fruit quality and storage performance of postharvest citrus fruit. Food Chem. 216: 138–145 (2017)
Stülke J, Hillen W. Regulation of carbon catabolism in Bacillus species. Annu. Rev. Microbiol. 54: 849–883 (2000)
Suwanmanon K, Hsieh PC. Isolating Bacillus subtilis and optimizing its fermentative medium for GABA and nattokinase production. CyTA J. Food. 12: 282–290 (2014a)
Suwanmanon K, Hsieh PC. Effect of gamma-aminobutyric acid and nattokinase-enriched fermented beans on the blood pressure of spontaneously hypertensive and normotensive Wistar-Kyoto rats. J. Food Drug Anal. 22: 485–491 (2014b)
Tajabadi N, Ebrahimpour A, Baradaran A, Rahim RA, Mahyudin NA, Manap MY, Bakar FA, Saari N. Optimization of gamma-aminobutyric acid production by Lactobacillus plantarum Taj-Apis362 from honeybees. Molecules 20: 6654–6669 (2015)
Torino MI, Limon RI, Martinez-Villaluenga C, Makinen S, Pihlanto A, Vidal-Valverde C, Frias J. Antioxidant and antihypertensive properties of liquid and solid state fermented lentils. Food Chem. 136: 1030–1037 (2013)
Wang HB, Zhang LW, Luo J, Yu LJ. Rapid and environmentally-friendly extraction of carotenoids from Blakeslea trispora. Biotechnol. Lett. 37: 2173–2178 (2015)
Wang L, Li P, Zhang Z, Chen Q, Aguilar ZP, Xu H, Yang L, Xu F, Lai W, Xiong Y, Wei H. Rapid and accurate detection of viable Escherichia coli O157:H7 in milk using a combined IMS, sodium deoxycholate, PMA and real-time quantitative PCR process. Food Control 36: 119–125 (2014a)
Wang Y, Luo Z, Huang X, Yang K, Gao S, Du R. Effect of exogenous γ-aminobutyric acid (GABA) treatment on chilling injury and antioxidant capacity in banana peel. Sci. Hort. 168: 132–137 (2014b)
Zhang G, Ren A, Wu F, Yu H, Shi L, Zhao M. Ethylene promotes mycelial growth and ganoderic acid biosynthesis in Ganoderma lucidum. Biotechnol. Lett. 39: 269–275 (2017)
Zhang Q, Xiang J, Zhang L, Zhu X, Evers J, van der Werf W, Duan L. Optimizing soaking and germination conditions to improve gamma-aminobutyric acid content in japonica and indica germinated brown rice. J. Funct. Foods. 10: 283–291 (2014)
Acknowledgements
This work was financially supported by a Grant-In-Aid (16GRNT26430067) from the American Heart Association (USA), the Agricultural and Social Development Program of Hangzhou Science and Technology Bureau of Zhejiang Province (China), the Young College Teachers Studying Abroad fund (Grant No. 3-2016) of Hubei Province (China), Jianghan University Doctoral Research Startup Fund Project (Grant No. 1017-06330003), and Major Technical Innovation Project of Hubei Province (China) (Grant No. 2017ABA147).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wang, H., Huang, J., Sun, L. et al. An efficient process for co-production of γ-aminobutyric acid and probiotic Bacillus subtilis cells. Food Sci Biotechnol 28, 155–163 (2019). https://doi.org/10.1007/s10068-018-0461-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-018-0461-7