Abstract
Objectives
Henoch–Schonlein purpura (HSP) is the most common childhood vasculitis, infection is the most essential inducement. We hypothesized that infection could impact the blood routine characteristics and/or outcome of vasculitis. Thus, we aim to find the most common infectious agent in HSP patients and identify convenient indicators to predict renal involvement in HSP patients with infection.
Method
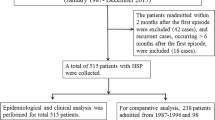
We conducted a retrospective study of 208 HSP children and 98 healthy children. Clinical parameters were compared in those cases.
Results
A total of 68.75% of patients were infected with various pathogens. The mean platelet volume (MPV) (P < 0.02) was lower in HSP patients with infection than patients without infection. Mycoplasma pneumoniae (MP) infection accounted for the largest proportion (45.77%). MPV in HSP nephritis (HSPN) group was lower than in HSP patients (excluded renal involvement) in patients with MP infection. Logistic regression analysis found that age and MPV were risk factors for the occurrence of MP-infected HSPN. The receiver operating characteristic curve (ROC) analysis showed that the combination of MPV with the onset age at the optimal cut-off point had 81% sensitivity in predicting whether HSP patients with MP infection would develop into HSPN.
Conclusions
Our research revealed that MP was the most commonly infected pathogen of children’s HSP. MPV was an essential predictor of nephritis in HSP patients with MP infection. This discovery can prompt clinical treatments as well as reduce costs.
Key Points • Mycoplasma pneumoniae (MP) accounts for the largest proportion in HSP children with infection. • MPV can be used as a predictor for the development of MP-triggered HSP to HSPN. |
Similar content being viewed by others
References
Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, Flores-Suarez LF, Gross WL, Guillevin L, Hagen EC, Hoffman GS, Jayne DR, Kallenberg CG, Lamprecht P, Langford CA, Luqmani RA, Mahr AD, Matteson EL, Merkel PA, Ozen S, Pusey CD, Rasmussen N, Rees AJ, Scott DG, Specks U, Stone JH, Takahashi K, Watts RA (2013) 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum 65(1):1–11. https://doi.org/10.1002/art.37715
Ruperto N, Ozen S, Pistorio A, Dolezalova P, Brogan P, Cabral DA, Cuttica R, Khubchandani R, Lovell DJ, O'Neil KM, Quartier P, Ravelli A, Iusan SM, Filocamo G, Magalhães CS, Unsal E, Oliveira S, Bracaglia C, Bagga A, Stanevicha V, Manzoni SM, Pratsidou P, Lepore L, Espada G, Kone-Paut I, Zulian F, Barone P, Bircan Z, Maldonado Mdel R, Russo R, Vilca I, Tullus K, Cimaz R, Horneff G, Anton J, Garay S, Nielsen S, Barbano G and Martini A (2010) EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part I: overall methodology and clinical characterisation. Ann Rheum Dis 69(5): p. 790–7. https://doi.org/10.1136/ard.2009.116624
Piram M, Maldini C, Biscardi S, De Suremain N, Orzechowski C, Georget E, Regnard D, Koné-Paut I, Mahr A (2017) Incidence of IgA vasculitis in children estimated by four-source capture-recapture analysis: a population-based study. Rheumatology (Oxford) 56(8):1358–1366. https://doi.org/10.1093/rheumatology/kex158
Oni L, Sampath S (2019) Childhood IgA vasculitis (Henoch Schonlein purpura)-advances and knowledge gaps. Front Pediatr 7:257. https://doi.org/10.3389/fped.2019.00257
Piram M, Mahr A (2013) Epidemiology of immunoglobulin A vasculitis (Henoch-Schönlein): current state of knowledge. Curr Opin Rheumatol 25(2):171–178. https://doi.org/10.1097/BOR.0b013e32835d8e2a
Gardner-Medwin JM, Dolezalova P, Cummins C, Southwood TR (2002) Incidence of Henoch-Schönlein purpura, Kawasaki disease, and rare vasculitides in children of different ethnic origins. Lancet 360(9341):1197–1202. https://doi.org/10.1016/s0140-6736(02)11279-7
Hwang HH, Lim IS, Choi BS, Yi DY (2018) Analysis of seasonal tendencies in pediatric Henoch-Schönlein purpura and comparison with outbreak of infectious diseases. Medicine (Baltimore) 97(36):e12217. https://doi.org/10.1097/md.0000000000012217
Jauhola O, Ronkainen J, Koskimies O, Ala-Houhala M, Arikoski P, Hölttä T, Jahnukainen T, Rajantie J, Ormälä T, Nuutinen M (2010) Clinical course of extrarenal symptoms in Henoch-Schonlein purpura: a 6-month prospective study. Arch Dis Child 95(11):871–876. https://doi.org/10.1136/adc.2009.167874
Blanco R, Martínez-Taboada VM, Rodríguez-Valverde V, García-Fuentes M, González-Gay MA (1997) Henoch-Schönlein purpura in adulthood and childhood: two different expressions of the same syndrome. Arthritis Rheum 40(5):859–864. https://doi.org/10.1002/art.1780400513
López-Mejías R, Castañeda S, Genre F, Remuzgo-Martínez S, Carmona FD, Llorca J, Blanco R, Martín J, González-Gay MA (2018) Genetics of immunoglobulin-A vasculitis (Henoch-Schönlein purpura): an updated review. Autoimmun Rev 17(3):301–315. https://doi.org/10.1016/j.autrev.2017.11.024
Karadağ ŞG, Çakmak F, Çil B, Tanatar A, Sönmez HE, Kıyak A, Yavuz S, Çakan M, and Aktay Ayaz N (2021) The relevance of practical laboratory markers in predicting gastrointestinal and renal involvement in children with Henoch-Schönlein Purpura. Postgrad Med 133(3): p. 272–277. https://doi.org/10.1080/00325481.2020.1807161
Yakut HI, Kurt T, Uncu N, Semsa Cayci F, and Celikel Acar B (2020) Predictive role of neutrophil to lymphocyte ratio and mean platelet volume in Henoch-Schönlein purpura related gastrointestinal and renal involvement. Arch Argent Pediatr 118(2):139–142. https://doi.org/10.5546/aap.2020.eng.139
Ozen S, Ruperto N, Dillon MJ, Bagga A, Barron K, Davin JC, Kawasaki T, Lindsley C, Petty RE, Prieur AM, Ravelli A, Woo P (2006) EULAR/PReS endorsed consensus criteria for the classification of childhood vasculitides. Ann Rheum Dis 65(7):936–941. https://doi.org/10.1136/ard.2005.046300
Elmas AT, Tabel Y (2016) Platelet counts in children with Henoch-Schonlein purpura–relationship to renal involvement. J Clin Lab Anal 30(1):71–74. https://doi.org/10.1002/jcla.21817
Özdemir ZC, Çetin N, Kar YD, Öcal HO, Bilgin M, Bör Ö (2020) Hemotologic indices for predicting internal organ involvement in Henoch-Schönlein purpura (IgA vasculitis). J Pediatr Hematol Oncol 42(1):e46–e49. https://doi.org/10.1097/mph.0000000000001571
Ekinci RMK, Balci S, Sari Gokay S, Yilmaz HL, Dogruel D, Altintas DU, and Yilmaz M (2019) Do practical laboratory indices predict the outcomes of children with Henoch-Schönlein purpura? Postgrad Med 131(4):295–298. https://doi.org/10.1080/00325481.2019.1609814
Park CH, Han DS, Jeong JY, Eun CS, Yoo KS, Jeon YC, Sohn JH (2016) The optimal cut-off value of neutrophil-to-lymphocyte ratio for predicting prognosis in adult patients with Henoch-Schönlein purpura. PLoS ONE 11(4):e0153238. https://doi.org/10.1371/journal.pone.0153238
Jaszczura M, Góra A, Grzywna-Rozenek E, Barć-Czarnecka M, Machura E (2019) Analysis of neutrophil to lymphocyte ratio, platelet to lymphocyte ratio and mean platelet volume to platelet count ratio in children with acute stage of immunoglobulin A vasculitis and assessment of their suitability for predicting the course of the disease. Rheumatol Int 39(5):869–878. https://doi.org/10.1007/s00296-019-04274-z
Gayret OB, Erol M, and Tekin Nacaroglu H (2016) The relationship of neutrophil-lymphocyte ratio and platelet-lymphocyte ratio with gastrointestinal bleeding in Henoch-Schonlein purpura. Iran J Pediatr 26(5): p. e8191. https://doi.org/10.5812/ijp.8191
Makay B, Türkyilmaz Z, Duman M, Unsal E (2009) Mean platelet volume in Henoch-Schönlein purpura: relationship to gastrointestinal bleeding. Clin Rheumatol 28(10):1225–1228. https://doi.org/10.1007/s10067-009-1219-7
Kapsoritakis AN, Koukourakis MI, Sfiridaki A, Potamianos SP, Kosmadaki MG, Koutroubakis IE, Kouroumalis EA (2001) Mean platelet volume: a useful marker of inflammatory bowel disease activity. Am J Gastroenterol 96(3):776–781. https://doi.org/10.1111/j.1572-0241.2001.03621.x
Liang QC, Jin D, Li Y, Wang RT (2014) Mean platelet volume and platelet distribution width in vascular dementia and Alzheimer’s disease. Platelets 25(6):433–438. https://doi.org/10.3109/09537104.2013.831064
Xu M, He XY, Huang P (2020) The relationship between the mean platelet volume and carotid atherosclerosis and prognosis in patients with acute cerebral infarction. Biomed Res Int 2020:6685740. https://doi.org/10.1155/2020/6685740
Gang L, Yanyan Z, Zhongwei Z, Juan D (2017) Association between mean platelet volume and hypertension incidence. Hypertens Res 40(8):779–784. https://doi.org/10.1038/hr.2017.30
Gasparyan AY, Ayvazyan L, Mikhailidis DP, Kitas GD (2011) Mean platelet volume: a link between thrombosis and inflammation? Curr Pharm Des 17(1):47–58. https://doi.org/10.2174/138161211795049804
Bitton A, Peppercorn MA (1995) Emergencies in inflammatory bowel disease. Crit Care Clin 11(2):513–529
Shi X, Li WC, Mo LJ, Li XH, Luo YZ, Qin LQ, Yang Z, Mo WN (2018) Altered mean platelet volume in children with Henoch-Schonlein purpura and its association with disease activity. Ann Clin Biochem 55(3):368–372. https://doi.org/10.1177/0004563217727015
Kocaoglu C (2017) Could platelet indices have predictive value in Henoch-Schonlein purpura? J Clin Anal Med 8(4). https://doi.org/10.4328/jcam.4874
Özel A, Bostan Gayret Ö, Erol M, Yiğit Ö, and Mete F (2018) Are mean platelet volume and neutrophil-to-lymphocyte ratio valuable in the early detection of system involvements in Henoch-Schönlein purpura? Med Bull Haseki 56(4): p. 279–285. https://doi.org/10.4274/haseki.4164
Robbins G, Barnard DL (1983) Mean platelet volume changes in infection. J Clin Pathol 36(11):1320. https://doi.org/10.1136/jcp.36.11.1320-a
Douda T, Bures J, Rejchrt S, Kopácová M, Pecka M, Malý J (2006) Mean platelet volume (MPV) in Crohn’s disease patients. Cas Lek Cesk 145(11):870–873
Mete E, Akelma AZ, Cizmeci MN, Bozkaya D, Kanburoglu MK (2014) Decreased mean platelet volume in children with acute rotavirus gastroenteritis. Platelets 25(1):51–54. https://doi.org/10.3109/09537104.2013.764493
Rigante D, Castellazzi L, Bosco A, Esposito S (2013) Is there a crossroad between infections, genetics, and Henoch-Schönlein purpura? Autoimmun Rev 12(10):1016–1021. https://doi.org/10.1016/j.autrev.2013.04.003
Lahita RG (1997) Influence of age on Henoch Schönlein purpura. Lancet 350(9085):1116–1117. https://doi.org/10.1016/s0140-6736(05)63784-1
Heineke MH, Ballering AV, Jamin A, Ben Mkaddem S, Monteiro RC, and Van Egmond M (2017) New insights in the pathogenesis of immunoglobulin A vasculitis (Henoch-Schönlein purpura). Autoimmun Rev 16(12): p. 1246–1253. https://doi.org/10.1016/j.autrev.2017.10.009
Trapani S, Micheli A, Grisolia F, Resti M, Chiappini E, Falcini F, De Martino M (2005) Henoch Schonlein purpura in childhood: epidemiological and clinical analysis of 150 cases over a 5-year period and review of literature. Semin Arthritis Rheum 35(3):143–153. https://doi.org/10.1016/j.semarthrit.2005.08.007
Liu LJ, Yu J, Li YN (2015) Clinical characteristics of Henoch-Schönlein purpura in children. Zhongguo Dang Dai Er Ke Za Zhi 17(10):1079–1083
Wang JJ, Xu Y, Liu FF, Wu Y, Samadli S, Wu YF, Luo HH, Zhang DD, Hu P (2020) Association of the infectious triggers with childhood Henoch-Schonlein purpura in Anhui province, China. J Infect Public Health 13(1):110–117. https://doi.org/10.1016/j.jiph.2019.07.004
Shang XP, Wu JG, Cheng Y, Hu HB (2021) Epidemiology and clinical characteristics of Henoch-Schönlein purpura associated with mycoplasma pneumoniae infection in 131 children in Hubei Province, China. Mediterr J Hematol Infect Dis 13(1):e2021037. https://doi.org/10.4084/mjhid.2021.037
Wang Y, Zhang Y, Lu W, Wang L (2018) Serum tumor necrosis factor-α and interferon-γ levels in pediatric Mycoplasma pneumoniae pneumonia: a systematic review and meta-analysis. Can Respir J 2018:8354892. https://doi.org/10.1155/2018/8354892
Kuźma-Mroczkowska E, Pańczyk-Tomaszewska M, Szmigielska A, Szymanik-Grzelak H, Roszkowska-Blaim M (2015) Mycoplasma pneumoniae as a trigger for Henoch-Schönlein purpura in children. Cent Eur J Immunol 40(4):489–492. https://doi.org/10.5114/ceji.2015.56976
Hu P, Guan Y, Lu L (2015) Henoch-Schönlein purpura triggered by Mycoplasma pneumoniae in a female infant. Kaohsiung J Med Sci 31(3):163–164. https://doi.org/10.1016/j.kjms.2014.12.006
Jung DY, Kwon YR, Yu MH, Namgoong MK (2017) Clinical features and prognosis of Henoch-Schönlein purpura in children and adults: a 13-year retrospective study at a single centre. Childhood Kidney Diseases 21(2):61–68. https://doi.org/10.3339/jkspn.2017.21.2.61
Calviño MC, Llorca J, García-Porrúa C, Fernández-Iglesias JL, Rodriguez-Ledo P, González-Gay MA (2001) Henoch-Schönlein purpura in children from northwestern Spain: a 20-year epidemiologic and clinical study. Medicine (Baltimore) 80(5):279–290. https://doi.org/10.1097/00005792-200109000-00001
Gonzalez-Gay MA, Calviño MC, Vazquez-Lopez ME, Garcia-Porrua C, Fernandez-Iglesias JL, Dierssen T, Llorca J (2004) Implications of upper respiratory tract infections and drugs in the clinical spectrum of Henoch-Schönlein purpura in children. Clin Exp Rheumatol 22(6):781–784
Fretzayas A, Sionti I, Moustaki M, Papadimitriou A, Nicolaidou P (2008) Henoch-Schönlein purpura: a long-term prospective study in Greek children. J Clin Rheumatol 14(6):324–331. https://doi.org/10.1097/RHU.0b013e31817a240a
Acknowledgements
The authors wish to express their gratitude to Dr Wenyuan Yang for his help in adjusting the format.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, C., Luo, L., Fu, M. et al. Analysis of children with Henoch–Schonlein purpura secondary to infection. Clin Rheumatol 41, 803–810 (2022). https://doi.org/10.1007/s10067-021-06007-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-021-06007-9