Abstract:
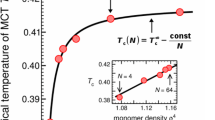
We study the reaction kinetics of end-functionalized polymer chains dispersed in an unreactive polymer melt. Starting from an infinite hierarchy of coupled equations for many-chain correlation functions, a closed equation is derived for the 2nd order rate constant k after postulating simple physical bounds. Our results generalize previous 2-chain treatments (valid in dilute reactants limit) by Doi [#!doi:inter2!#], de Gennes [#!gennes:polreactionsiandii!#], and Friedman and O'Shaughnessy [#!ben:interdil_all_aip!#], to arbitrary initial reactive group density n0 and local chemical reactivity Q. Simple mean field (MF) kinetics apply at short times, .For high Q, a transition occurs to diffusion-controlled (DC) kinetics with (where xt is rms monomer displacement in time t) leading to a density decay . If n0 exceeds the chain overlap threshold, this behavior is followed by a regime where during which k has the same power law dependence in time, , but possibly different numerical coefficient. For unentangled melts this gives while for entangled cases one or more of the successive regimes ,t -3/8 and t -3/4 may be realized depending on the magnitudes of Q and n0. Kinetics at times longer than the longest polymer relaxation time are always MF. If a DC regime has developed before then the long time rate constant is where R is the coil radius. We propose measuring the above kinetics in a model experiment where radical end groups are generated by photolysis.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 2 June 1998 / Revised: 9 July 1998 / Accepted: 10 July 1998
Rights and permissions
About this article
Cite this article
O'Shaughnessy, B., Vavylonis, D. Reaction kinetics in polymer melts. Eur. Phys. J. B 6, 363–372 (1998). https://doi.org/10.1007/s100510050562
Issue Date:
DOI: https://doi.org/10.1007/s100510050562