Abstract
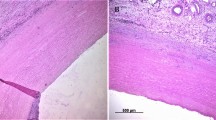
Incorporation of surrounding tissues after implantation of synthetic vascular prostheses potentially varies in accordance with implanted prostheses. To evaluate post-implant tissue incorporation, we examined surgical, histological and ultrastructural findings after implantation in animal models. Three types of commercially available prostheses were tested (Gelweave™; Group G, J Graft SHIELD NEO®; Group J and Triplex®; Group T). Prostheses were implanted into Sprague–Dawley rats subcutaneously or sutured on abdominal aorta of Japanese white rabbits. The tissues were surgically examined for adhesion and were subjected to histological evaluations for cellular and tissue infiltration and ultrastructural observations by scanning electron microscopy (SEM). Group G exhibited less tendency in adhesion formation in early phase (rat: G vs J, P < 0.0001; G vs T, P < 0.0001/rabbit: G vs J, P < 0.0001; G vs T, P = 0.059). In late phase, Group J showed highest adhesion (rat: G vs J, P = 0.0004; J vs T, P = 0.015/rabbit: G vs J, P = 0.0015; J vs T, P = 0.0044). In group G, a gap was observed between implants and surrounding tissues forming capsulation, whereas other groups exhibited tissue infiltration inside of the implants wall which were also confirmed by SEM. The tissue permeation toward the implants and adhesion was positively correlated (P < 0.0001). Surrounding tissue conformation varied in accordance with the type of prostheses. It is desirable to elucidate characteristics of each prosthesis to select suitable grafts for each patient to achieve a better surgical outcome.





Similar content being viewed by others
References
Fernandez FG, Shahian DM, Kormos R, Jacobs JP, D’Agostino RS, Mayer JE Jr, et al. The Society of Thoracic Surgeons National Database 2019 Annual Report. Ann Thorac Surg. 2019;108:1625–32.
Tamura A, Yamaguchi A, Yuri K, Noguchi K, Naito K, Nagano H, et al. Clinical experience with a new vascular graft free from biodegradable material. Interact Cardiovasc Thorac Surg. 2011;12:758–61.
Lederle FA, Freischlag JA, Kyriakides TC, Matsumura JS, Padberg FT Jr, Kohler TR, et al. Long-term comparison of endovascular and open repair of abdominal aortic aneurysm. N Engl J Med. 2012;367:1988–97.
Zhou KZ, Maingard J, Phan K, Kok HK, Lee MJ, Brooks DM, et al. The 100 most cited articles in the endovascular treatment of thoracic and abdominal aortic aneurysms. J Vasc Surg. 2018;68:1566–81.
Heydorn WH, Daniel JS, Wade CE. A new look at pericardial substitutes. J Thorac Cardiovasc Surg. 1987;94:291–6.
Ukpabi P, Marois Y, King M, Deng X, Martin L, Laroche G, et al. The gelweave polyester arterial prosthesis. Can J Surg. 1995;38:322–31.
Munakata H, Yamanaka K, Okada K, Okita Y. Successful surgical treatment of aortoesophageal fistula after emergency thoracic endovascular aortic repair: aggressive debridement including esophageal resection and extended aortic replacement. J Thorac Cardiovasc Surg. 2013;146:235–7.
Ohata T, Ueda H, Kobayashi K, Fukuda H, Miyamoto Y. Terumo-Triplex grafts for total arch replacement: analysis of postoperative graft performance. J Artif Organs. 2012;15:240–3.
Watt SM, Gschmeissner SE, Bates PA. PECAM-1: its expression and function as a cell adhesion molecule on hemopoietic and endothelial cells. Leuk Lymphoma. 1995;17:229–44.
Mathisen SR, Wu HD, Sauvage LR, Usui Y, Walker MW. An experimental study of eight current arterial prostheses. J Vasc Surg. 1986;4:33–41.
Patel M, Arnell RE, Sauvage LR, Wu HD, Shi Q, Wechezak AR, et al. Experimental evaluation of ten clinically used arterial prostheses. Ann Vasc Surg. 1992;6:244–51.
Jonas RA, Schoen FJ, Levy RJ, Castaneda AR. Biological sealants and knitted Dacron: porosity and histological comparisons of vascular graft materials with and without collagen and fibrin glue pretreatments. Ann Thorac Surg. 1986;41:657–63.
Lei ZY, Liu T, Li WJ, Shi XH, Fan DL. Biofunctionalization of silicone rubber with microgroove-patterned surface and carbon-ion implantation to enhance biocompatibility and reduce capsule formation. Int J Nanomedicine. 2016;11:5563–72.
Tabata M, Shimokawa T, Fukui T, Manabe S, Sato Y, Takanashi S. New uncoated vascular prosthesis reduces mediastinal tube drainage after thoracic aortic surgery. Ann Thorac Surg. 2011;91:899–902.
Knoner G, Rolfe BE, Campbell JH, Parkin SJ, Heckenberg NR, Rubinsztein-Dunlop H. Mechanics of cellular adhesion to artificial artery templates. Biophys J. 2006;91:3085–96.
Lucke S, Hoene A, Walschus U, Kob A, Pissarek JW, Schlosser M. Acute and chronic local inflammatory reaction after implantation of different extracellular porcine dermis collagen matrices in rats. Biomed Res Int . 2015;2015:938059.
Taksaudom N, Ketwong M, Lertprasertsuke N, Kongkaew A. Postoperative pericardial adhesion prevention using collagen membrane in pigs: a pilot study. Open J Cardiovasc Surg. 2017;9:1179065217720909.
Hamaji M, Kojima F, Komatsu T, Tsuruyama T, Date H, Nakamura T. A synthetic bioabsorbable sheet may prevent postoperative intrapleural adhesions following thoracotomy: a canine model. Interact Cardiovasc Thorac Surg. 2014;19:914–20.
Kamitani T, Masumoto H, Kotani H, Ikeda T, Hyon SH, Sakata R. Prevention of retrosternal adhesion by novel biocompatible glue derived from food additives. J Thorac Cardiovasc Surg. 2013;146:1232.
De Paulis R, Riebman JB, Deleuze P, Mohammad FS, Burns GL, Morea M, et al. The use of an artificial pericardium with a total artificial heart. J Cardiovasc Surg (Torino). 1990;31:202–8.
DiEgidio P, Friedman HI, Gourdie RG, Riley AE, Yost MJ, Goodwin RL. Biomedical implant capsule formation: lessons learned and the road ahead. Ann Plast Surg. 2014;73:451–60.
Rowe NM, Impellizzeri P, Vaynblat M, Lawson NM, Kim YD, Sierra M, et al. Studies in thoracic aortic graft infections: the development of a porcine model and a comparison of collagen-impregnated dacron grafts and cryopreserved allografts. J Thorac Cardiovasc Surg. 1999;118:857–65.
Matsumoto H, Sugiyama S, Shibazaki A, Tanaka R, Takashima K, Noishiki Y, et al. Experimental study of materials for patch graft on right ventricular outflow tract under extracorporeal circulation in dogs—comparison between Denacol EX-313-treated bovine jugular vein graft and expanded polytetrafluoroethylene (EPTFE) graft. J Vet Med Sci. 2001;63:961–5.
Acknowledgements
The authors thank Mr. Shuichi Miyake, Ms. Yasuyo Matsubara, Mr. Haruyasu Koda and Ms. Keiko Furuta (Kyoto University) for the technical support. This work was supported by collaborative research grants from Terumo Corp. and Japan Lifeline Co. Ltd, and Invited Research Project of Institute for Advancement of Clinical Transnational Science, Kyoto University Hospital (to Kenji Minatoya).
Author information
Authors and Affiliations
Contributions
KM and HM contributed to the study conception and design. Material preparation, data collection and analysis were performed by YF, SH and HM. Data analyses were performed by KY and TI. The first draft of the manuscript was written by YF and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial relationship with the organization that sponsored the research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fujiwara, Y., Hirao, S., Yamazaki, K. et al. Evaluations for surrounding tissue incorporation after implantation of synthetic vascular prostheses in animal models. J Artif Organs 25, 24–33 (2022). https://doi.org/10.1007/s10047-021-01283-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-021-01283-3