Abstract
Patients with 9q34.3 terminal deletion usually show a clinically recognizable phenotype characterized by specific facial features (microcephaly, flat face, arched eyebrows, hypertelorism, short nose, anteverted nostrils, carp mouth and protruding tongue) in combination with severe mental retardation, hypotonia, and other anomalies. We analyzed six unrelated patients with a various 9q34.3 terminal deletion. While having different-sized 9q34.3 deletions, all of these patients shared several distinctive anomalies. These anomalies are likely to arise from a commonly deleted region at distal 9q34.3. Fluorescence in situ hybridization (FISH) analysis using a dozen BAC clones mapped at the 9q34.13-q34.3 region defined the shortest region of deletion overlap (SRO) as a 1-Mb segment proximal to 9qter containing eight known genes. Possible candidate genes delineating specific phenotypes of the 9q34.3 terminal deletion syndrome are discussed.
Similar content being viewed by others
Introduction
Subtelomeric chromosomal rearrangements comprise about half of segmental aneusomies and are one of the major causes of mental retardation (MR). A recent review showed that cryptic subtelomere rearrangements were detected in 131 (5.1%) of 2,585 children with MR (Flint and Knight 2003) and submicroscopic deletions of 4p, 5p, 9p, 16p, 17p, 1p, 2q, and 22q were frequently observed (De Vries et al. 2003). We previously reported five unrelated patients with a 9q34.3 deletion (Dawson et al. 2002; Iwakoshi et al. 2004). In addition, we found another, which was previously reported as a MCA/MR case of satellited chromosome 9 (Harada et al. 1989). All of these patients shared several distinctive anomalies, which are likely to have their origin in a deletion overlap region at distal 9q34.3. Definition of shortest region of deletion overlap (SRO) is therefore very important to delineate the critical region for 9q34.3 terminal deletion syndrome. In the present study, we identified the SRO as a 1-Mb segment proximal to 9qter and discuss candidate genes likely to be responsible for the clinical features manifested in the 9q terminal deletion syndrome. Furthermore, haplotype analysis in order to determine parental origin of the deletion was performed in three patients.
Materials and methods
Subjects
We analyzed six patients with 9q34.3 terminal deletions, and their karyotypes were 46,XY,der(9)t(9;22)(q34.3;q11.21)de novo (Case A: originally described as a case report by Harada et al. 1989), 46,XX. ish del(9)(q34.3)de novo (Case B: described as Patient 1 in Iwakoshi et al. 2004), 46,XY. ish del(9)(q34.3)de novo (Case C: described as N03R003 and Patient 2 in Harada et al. 2004 and Iwakoshi et al. 2004, respectively), 46,XX. ish der(9)t(5;9)(q35.3;q34.3)pat (Case D: described as Patient 1 in Dawson et al. 2002), 46,XX. ish del(9)(q34.3)de novo (Case E: described as Patient 2 in Dawson et al. 2002), and 46,XY. ish der(9)t(6;9)(p25;q34.3)de novo (Case F: described as Patient 3 in Iwakoshi et al. 2004). Clinical features of Cases B, C, D, E, and F were previously described in detail (Iwakoshi et al. 2004). Case A was clinically reevaluated in the present study because 9q34.3 terminal deletion was newly confirmed (Table 1).
Fluorescence in situ hybridization (FISH) analysis
Fluorescence in situ hybridization (FISH) analysis was performed using 13 RPCI-11 human BAC clones (RP11-89B23, 92B21, 265M2, 166H7, 98L5, 432I20, 83N9, 370H5, 769N4, 350O14, 974F22, 417A4, and 424E7) mapped at 9q34.13-q34.3 according to Human Genome Browser 2003 July version (http://www.genome.ucsc.edu/cgi-bin/hgGateway?db=hg11) and a subtelomeric BAC clone, GS-135I17 (Knight et al. 2000) (Fig. 1). BAC clone DNAs labeled with either SpectrumOrange-dUTP or SpectrumGreen-dUTP (Vysis, Downers Grove, IL, USA) by nick translation were denatured and hybridized to metaphase chromosomes of patients at 37°C for 16 h and washed. In order to confirm the common telomeric structure of deleted chromosomes, FISH using telomere-specific probes containing TTAGGG repeats generated by PCR (IJdo et al. 1991) and labeled with a similar manner as BACs was performed in Cases E and F. Fluorescence images were captured using a monochrome CCD camera (Carl Zeiss, Jena, Germany) on Axioplan 2 fluorescence microscope (Carl Zeiss) with appropriate filters and were analyzed with ISIS version 3.4.5 software (MetaSystems, Altlussheim, Germany).
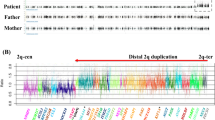
Fluorescence in situ hybridization (FISH) studies confirming the shortest region of deletion overlap (SRO) in six cases of 9q34.3 terminal deletion. a Schematic presentation of the SRO. Top row depicts BAC clones and STR markers used in this study and their genomic location. Solid thick bars from top to bottom indicate deleted segments of Cases A–F, respectively. Bi-directional arrow indicates the SRO. Eight known genes exist within the SRO according to the Human Genome Browser, 2003 July version (http://www.genome.ucsc.edu/cgi-bin/hgGateway?db=hg11). cen centromere, tel end telomeric end. Results of FISH studies revealing the 1 Mb of minimal deletion: b and c show FISH studies using RP11-350O14(Green) and RP11-417A4 (Orange) in Case E, respectively. d and e show FISH results using RP11-350O14 (Green) and RP11-417A4 (Orange) in Case F, respectively. Arrow and arrowhead indicate existence of a signal and absence of a signal, respectively. f and g show the results of FISH using the telomeric-repeat probe in Cases E and F, respectively, indicating that the common telomeric structure was preserved in both cases
Haplotype analysis
In order to confirm parental origin of 9q deletion, we performed haplotype analysis using available parental samples of three cases (B, C, and F) by use of two STR markers located within the deleted region. Sequence information of PCR primers of D9S1838 were described elsewhere (Cormier-Daire et al. 2003). Primer sequence of SC3Q9R, TGCTGTCAAAGGACCTACCC for forward and ACCCCATTCTGAACAAAGCA for reverse were originally designed based on the sequence information from the NCBI Homo sapiens genome view build 33 April 2003 database. PCR was performed in a 20 μl mixture containing 50–75 ng genomic DNA, 1 μM of fluorescent labeled primer and reverse primer, 0.5 U Takara Ex Taq polymerase (Takara Bio, Ohtsu, Japan), 0.2 mM each dNTP and 1X Ex Taq Buffer (Takara Bio). PCR conditions included initial denaturation at 98°C for 2 min, 35 cycles of 98°C for 10 s, 55°C for 30 s, 72°C for 30 s, and a final extension of 72°C for 7 min. PCR products were loaded for electrophoresis in an ABI Prism 377 DNA sequencer (Applied Biosystems, Tokyo, Japan) and analyzed by the manufacturer-supplied GeneScan Analysis and Genotyper Software (Applied Biosystems).
Results and discussion
The FISH analysis revealed that the SRO was defined as a region within 1-Mb from the 9q telomere end (Fig. 1a,b,c,d,e). In addition, the intact common telomere structure with TTAGGG repeats was confirmed in Case E (Fig. 1f) and Case F (Fig. 1g). The SRO is likely to be responsible for typical facial features (flat face, arched eyebrows, hypertelorism, short nose, anteverted nostrils, and open carp mouth), hypotonia, and MR, as all six cases shared them (Table 1). Cormier-Daire et al. (2003) reported a de novo deletion of chromosome 9q34 in two unrelated MR children with early onset of obesity, distinctive facial features, sleep disturbance, and behavioral problems. Their FISH and STR analysis revealed that both children carried a similar-sized deletion of 3 Mb. However, these 3-Mb sized regions were not precisely defined due to lack of informative STR markers in the region between D9S1826 and D9S1838, and FISH analysis of this region was not performed. The SRO identified in this study contains several known genes (Fig. 1a). Among these, NELF, MIZIP, Eu-HMTase1 and CACNA1B could be functionally important, but no mutations associated with human diseases have been reported. NELF encodes nasal embryonic LHRH factor (OMIM *608137). Murine Nelf is expressed in peripheral and central nervous systems during embryonic development, including olfactory sensory cells and LHRH cells, and is suggested to play roles in guidance for olfactory axon projections and neurophilic migration of LHRH cells (Kramer et al. 2000). Although the patients we are describing were not reported to have abnormal olfaction, careful olfactory evaluations may be necessary. MIZIP encodes a melanin-concentrating hormone receptor 1 interacting zinc-finger protein and is expressed ubiquitously, although a higher expression in the brain is reported (Bachner et al. 2002). MIZIP deletion may contribute to MR of 9q34.3 deletion syndrome. Euchromatic histone methyltransferase 1 (Eu-HMTase1, OMIM *607001) is a component of the E2F transcription factor 6 (E2F6, OMIM *602944) (Trimarchi et al. 1998) complex that represses transcription. Eu-HMTase1 methylates lys-9 of histone H3, resulting in epigenetic transcriptional repression by recruiting heterochromatin protein 1 to methylated histones (Ogawa et al. 2002) and may maintain normal brain function/development. The calcium channel, voltage-dependent, N-type, alpha-1B subunit gene (CACNA1B, OMIM *601012) encodes an N-type calcium channel involving neurotransmitter release from neurons (Williams et al. 1992). Recently, Maeno-Hikichi et al. showed that enigma-like LIM domain protein interacts with protein kinase C-epsilon and CACNA1B to form a PKCε-ENH-Ca2+ channel complex (Maeno-Hikichi et al. 2003). Heterozygous deletion of CACNA1B may result in the reduction of N-type channel activity and can be a contributing factor for MR and/or epilepsy.
An autosomal recessive neurological disorder, Joubert syndrome type 1 (JBTS1, OMIM #213300), was mapped close to D9S158 at 9q34.3 (Saar et al. 1999). D9S158 was identified within RP11-83N9, which was deleted in three of our six cases. JBTS1 presents with several clinical features similar to the 9q34.3 deletion syndrome. MR, hypotonia, and protruding tongue are observed in both syndromes, but cerebellar anomalies or ataxia were not evident in the 9q34.3 deletion syndrome. As the JBTS1 locus overlaps with the SRO shown in this study, it is possible that the 9q34.3 deletion syndrome phenotype might be caused by unmasking of a recessive allele for JBTS1. Although the linkage to 9q34.3 in JBTS1 is described in a single family so far, further study is necessary.
The results of the haplotype analysis in addition to previous studies regarding paternity of deletion are shown in Table 2. In Case A, paternal inheritance of his derivative chromosome 9 was traced by use of chromosomal polymorphism as a marker. In Case D, his derivative chromosome 9 originated from a paternal translocation. Cormier-Daire et al. (2003) reported a maternal origin of de novo 9q terminal deletion in one of two cases analyzed, while the other was paternal. We detected a parental origin of deletion in four of six cases in this study. The origin in the other two remains to be elucidated. Both paternal and maternal deletions have been found (Table 2), being supported by the fact that imprinting effects have not been established for chromosome 9 (Ledbetter and Engel 1995).
In conclusion, we delimited the SRO of the 9q34.3 terminal deletion syndrome to a region within 1-Mb from the 9q telomeric end that contains several genes important for human development. It remains to be investigated whether the SRO as defined by this study is representative for other patients with the 9q34.3 deletion syndrome or can be confined to an even smaller segment.
References
Bachner D, Kreienkamp HJ, Richter D (2002) MIZIP, a highly conserved, vertebrate specific melanin-concentrating hormone receptor 1 interacting zinc-finger protein. FEBS Lett 28:124–128
Cormier-Daire V, Molinari F, Rio M, Raoul O, de Blois MC, Romana S, Vekemans M, Munnich A, Colleaux L (2003) Cryptic terminal deletion of chromosome 9q34: a novel cause of syndromic obesity in childhood? J Med Genet 40:300–303
Dawson AJ, Putnam S, Schultz J, Riordan D, Prasad C, Greenberg CR, Chodirker BN, Mhanni AA, Chudley AE (2002) Cryptic chromosome rearrangements detected by subtelomere assay in patients with mental retardation and dysmorphic features. Clin Genet 62:488–494
De Vries BB, Winter R, Schinzel A, Ravenswaaij-Arts C (2003) Telomeres: a diagnosis at the end of the chromosomes. J Med Genet 40:385–398
Flint J, Knight S (2003) The use of telomere probes to investigate submicroscopic rearrangements associated with mental retardation. Curr Opin Genet Dev 13:310–316
Harada N, Abe K, Kondoh T, Hirota T, Niikawa N (1989) Satellited chromosome 9 in a boy with multiple anomalies. Jpn J Hum Genet 34:297–305
Harada N, Hatchwell E, Okamoto N, Tsukahara M, Kurosawa K, Kawame H, Kondoh T, Ohashi H, Tsukino R, Kondoh Y, Shimokawa O, Ida T, Nagai T, Fukushima Y, Yoshiura K, Niikawa N, Matsumoto N (2004) Subtelomere specific microarray based comparative genomic hybridisation: a rapid detection system for cryptic rearrangements in idiopathic mental retardation. J Med Genet 41:130–136
IJdo JW, Wells RA, Baldini A, Reeders ST (1991) Improved telomere detection using a telomere repeat probe (TTAGGG)n generated by PCR. Nucleic Acid Res 19:4780
Iwakoshi M, Okamoto N, Harada N, Nakamura T, Yamamori S, Fujita H, Niikawa N, Matsumoto N (2004) 9q34.3 deletion syndrome in three unrelated children. Am J Med Genet 30:278–283
Knight SJ, Lese CM, Precht KS, Kuc J, Ning Y, Lucas S, Regan R, Brenan M, Nicod A, Lawrie NM, Cardy DL, Nguyen H, Hudson TJ, Riethman HC, Ledbetter DH, Flint J (2000) An optimized set of human telomere clones for studying telomere integrity and architecture. Am J Hum Genet 67:320–332
Kramer PR, Wray S (2000) Novel gene expressed in nasal region influences outgrowth of olfactory axons and migration of luteinizing hormone-releasing hormone (LHRH) neurons. Genes Dev 14:1824–1834
Ledbetter DH, Engel E (1995) Uniparental disomy in humans: development of an imprinting map and its implications for prenatal diagnosis. Hum Mol Genet 4:1757–1764
Maeno-Hikichi Y, Chang S, Matsumura K, Lai M, Lin H, Nakagawa N, Kuroda S, Zhang JF (2003) A PKC epsilon-ENH-channel complex specifically modulates N-type Ca2+ channels. Nat Neurosci 6:468–475
Ogawa H, Ishiguro K, Gaubatz S, Livingston DM, Nakatani Y (2002) A complex with chromatin modifiers that occupies E2F- and Myc-responsive genes in G0 cells. Science 10:1034–1035
Saar K, Al-Gazali L, Sztriha L, Rueschendorf F, Nur-E-Kamal M, Reis A, Bayoumi R (1999) Homozygosity mapping in families with Joubert syndrome identifies a locus on chromosome 9q34.3 and evidence for genetic heterogeneity. Am J Hum Genet 65:1666–1671
Trimarchi JM, Fairchild B, Verona R, Moberg K, Andon N, Lees JA (1998) E2F-6, a member of the E2F family that can behave as a transcriptional repressor. Proc Natl Acad Sci USA 95:2850–2855
Williams ME, Brust PF, Feldman DH, Patthi S, Simerson S, Maroufi A, McCue AF, Velicelebi G, Ellis SB, Harpold MM (1992) Structure and functional expression of an omega-conotoxin-sensitive human N-type calcium channel. Science 257:389–395
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harada, N., Visser, R., Dawson, A. et al. A 1-Mb critical region in six patients with 9q34.3 terminal deletion syndrome. J Hum Genet 49, 440–444 (2004). https://doi.org/10.1007/s10038-004-0166-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10038-004-0166-z
Keywords
This article is cited by
-
The sociability spectrum: evidence from reciprocal genetic copy number variations
Molecular Autism (2020)
-
KMT2C/D COMPASS complex-associated diseases [KCDCOM-ADs]: an emerging class of congenital regulopathies
Clinical Epigenetics (2020)
-
Mikrodeletionssyndrom 9q34 – ein charakteristischer Phänotyp
Monatsschrift Kinderheilkunde (2007)