Abstract
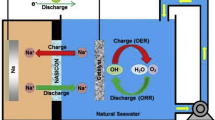
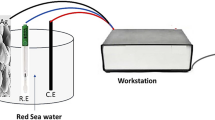
An environmentally friendly cell using polypyrrole-air regenerative cathode and zinc as anode is investigated in the 3% sodium chloride solution. The cell can operate in different charge and discharge mode. Polypyrrole can be reoxidized (doped) with chloride anions either by using dissolved oxygen or by an external power supply, e.g., small photovoltaic cell. In that way, after discharge, capacity retaining can be achieved by using seawater as the electrolyte. During low discharge rate, the delicate balance between solid state diffusion-controlled dedoping and chemical oxidation of polypyrrole produced by hydrogen peroxide is achieved, generating stable voltage plateau. The cell is proposed to operate as a power supply for different sensor devices in two modes. In the low discharge mode (10–20 mA g−1), it can be used for data acquisition, and at the fast discharge mode (up to 2 A g−1) for collecting data transmission.

Charge-discharge curves for different curent densities of Zn|3.5% NaCl(aq)|PPy. Inset: Continuous discharge of the cell under external load of 1 kΩ and constant air supply, during 1-h discharge and 1-h reoxidation charge











Similar content being viewed by others
References
Wilcock WSD, Kauffman PC (1997) Development of a seawater battery for deep-water applications. J Power Sources 66:71–75
Shinohara M, Araki E, Mochizuki M, Kanazawaa T, Suyehiro K (2009) Practical application of a sea-water battery in deep-sea basin and its performance. J Power Sources 187:253–260
Wang N, Wang R, Peng C, Feng Y, Chen B (2012) Effect of hot rolling and subsequent annealing on electrochemical discharge behavior of AP65 magnesium alloy as anode for seawater activated battery. Corros Sci 64:17–27
Zhu Y, Gao P, Teng X, Hu H (2011) S as additive in Mg–Cu2O seawater batteries using Ni foam-supported electrode. Ionics 17:853–857
Koontz RF, Lucero RD (1995) Magnesium water-activated batteries. In: Linden D (ed) Handbook of batteries, 3rd edn. McGraw-Hill, New York Chapter 17
Rao BML, Kobasz W, Hoge WH, Halmen RP, Halliop W, Fitzpatric NP (1992) Advances in aluminum-air salt water batteries. In: Brian E, Murphy OJ, Srinivasan S (eds) Electrochemistry in transition, Conway. Plenum Press, New York, pp 629–639
Maa J, Wen J, Gao J, Li Q (2014) Performance of Al-0.5 Mg-0.02 Ga-0.1 Sn-0.5 Mn as anode for Al-air battery in NaCl solutions. J Power Sources 253:419–423
Wang N, Wang R, Peng C, Peng B, Feng Y, Hu C (2014) Discharge behavior of Mg-Al-Pb and Mg-Al-Pb-In alloys as anodes for Mg-air battery. Electrochim Acta 149:193–205
Mori R (2015) Capacity recovery of aluminium–air battery by refilling salty water with cell structure modification. J Appl Electrochem 45:821–829
Shen PK, Tseung ACC, Kuo C (1994) Development of an aluminum/sea water battery for subsea applications. J Power Sources 47:119–127
Zhang T, Tao Z, Chen J (2014) Magnesium–air batteries: from principle to application. Mater Horiz 1:196–206
Wittmaier D, Danner T, Wagner N, Friedrich KA (2014) Screening and further investigations on promising bi-functional catalysts for metal–air batteries with an aqueous alkaline electrolyte. J Appl Electrochem 44:73–85
Ma Z, Pei P, Wang K, Wang X, Xu H, Liu Y, Peng G (2015) Degradation characteristics of air cathode in zinc air fuel cells. J Power Sources 274:56–64
Song H-K, Palmore GTR (2006) Redox-active polypyrrole: toward polymer-based batteries. Adv Mater 18:1764–1768
Nyström G, Razaq A, Strømme M, Nyholm L, Mihranyan A (2009) Ultrafast all-polymer paper-based batteries. Nano Lett 9(10):3635–3639
Pasta M, Wessells CD, Huggins RA, Yi C (2012) A high-rate and long cycle life aqueous electrolyte battery for grid-scale energy storage. Nat Commun 3:1149
Kong Y, Wang C, Yang Y, Too CO, Wallace GG (2012) A battery composed of a polypyrrole cathode and a magnesium alloy anode—toward a bioelectric battery. Synth Met 162:584–589
Wang C, Zheng W, Yue Z, Too CO, Wallace GG (2011) Buckled, stretchable polypyrrole electrodes for battery applications. Adv Mater 23:3580–3584
Li S, Sultana I, Guo Z, Wang C, Wallace GG, Liu H-K (2013) Polypyrrole as cathode materials for Zn-polymer battery with various biocompatible aqueous electrolytes. Electrochim Acta 95:212–217
Li S, Ping Z, Wang GCY, Wallace GG, Liu HK (2013) Flexible cellulose based polypyrrole–multiwalled carbon nanotube films for bio-compatible zinc batteries activated by simulated body fluids. J Mater Chem A 1:14300–14305
Grgur BN, Gvozdenović MM, Stevanović J, Jugović BZ, Marinović VM (2008) Polypyrrole as possible electrode materials for the aqueous-based rechargeable zinc batteries. Electrochim Acta 53(14):4627–4632
Audebert P (2010) Recent trends in polypyrrole electrochemistry, nanostructuration, and applications. In: Cosnier S, Karyakin A (eds) Electropolymerization: concepts, materials and applications. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, pp 77–91
Saidman SB (2003) Influence of anion and pH on the electrochemical behavior of polypyrrole synthesised in alkaline media. Electrochim Acta 48:1719–1726
Wallace GG, Tsekouras G, Wang C (2010) Inherently conducting polymers via electropolymerization for energy conversion and storage. In: Cosnier S, Karyakin A (eds) Electropolymerization: concepts, materials and applications. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, pp 215–240
Wang GJ, Yang LC, Qu QT, Wang B, Wu YP, Holze R (2010) An aqueous rechargeable lithium battery based on doping and intercalation mechanisms. J Solid State Electrochem 14:865–869
Kitani A, Kaya M, Sasaki K (1986) Performance study of aqueous polyaniline batteries. J Electrochem Soc 133(6):1069–1073
Barsukov VZ, Khomenko VG, Katashinskii AS, Motronyuk TI (2004) Catalytic activity of polyaniline in the molecular oxygen reduction: its nature and mechanism. Russ J Electrochem 40(11):1170–1173
Khomenko VG, Barsukov VZ, Katashinskii AS (2005) The catalytic activity of conducting polymers toward oxygen reduction. Electrochim Acta 50:1675–1683
Barsukov VZ, Khomenko VG, Katashinskii AS, Motronyuk TI (2006) New concept for the metal-air batteries using composites: conducting polymers/extended graphite as catalyst. In: Barsukov IV et al (eds) New carbon based materials for electrochemical energy storage systems. Springer, Netherlands, pp 89–104
Khomenko VG, Lykhnytskyi KV, Barsukov VZ (2013) Oxygen reduction at the surface of polymer/carbon and polymer/carbon/spinel catalysts in aqueous solutions. Electrochim Acta 104:391–399
Wu A, Venancio EC, MacDiarmid AG (2007) Polyaniline and polypyrrole oxygen reversible electrodes. Synth Met 157:303–310
Grgur BN (2014) Metal|polypyrrole battery with the air regenerated positive electrode. J Power Sources 272:1053–1060
Vernitskaya TV, Efimov ON (1997) Polypyrrole: a conducting polymer; its synthesis, properties, and applications. Russ Chem Rev 66(5):443–457
Carrasco PM, Cortazar M, Ochoteco E, Calahorra E, Pomposo JA (2007) Comparison of surface and bulk doping levels in chemical polypyrroles of low, medium and high conductivity. Surf Interface Anal 39:26–32
Stanković R, Pavlović O, Vojnović M, Jovanović S (1994) The effect of preparation conditions on the properties of electrochemically synthesized thick films of polypyrrole. Eur Polym J 30(3):385–393
Jia X, Wang C, Zhao C, Ge Y, Wallace GG (2016) Toward biodegradable Mg–air bioelectric batteries composed of silk fibroin–polypyrrole film. Adv Funct Mater 26:1454–1462
Weidlich C, Mangold K-M, Jüttner K (2005) EQCM study of the ion exchange behaviour of polypyrrole with different counterions in different electrolytes. Electrochim Acta 50:1547–1552
Mazeikiene R, Malinauskas A (2002) Kinetics of the electrochemical degradation of polypyrrole. Polym Degrad Stab 75:255–258
Careem MA, Velmurugu Y, Skaarup S, West K (2006) A voltammetry study on the diffusion of counter ions in polypyrrole films. J Power Sources 159:210–214
Alumaa A, Hallik A, Sammelselg V, Tamm J (2007) On the improvement of stability of polypyrrole films in aqueous solutions. Synth Met 157:485–491
Trišović T, Gajić-Krstajić LJ, Krstajić N, Vojnović M (2001) On the kinetics of the hydrogen evolution reaction on zinc in sulfate solutions. J Serb Chem Soc 66(11–12):811–823
Acknowledgements
The research was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia, under the research project “Electrochemical synthesis and characterization of nanostructured functional materials for applications in new technologies” No. ON172046. Ali H. Al-Eggiely and Alsadek A. Alguail are grateful to the Libyan Ministry of Higher Education and Scientific Research for the material supports during PhD study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Eggiely, A.H., Alguail, A.A., Gvozdenović, M.M. et al. Seawater zinc/polypyrrole-air cell possessing multifunctional charge-discharge characteristics. J Solid State Electrochem 21, 2769–2777 (2017). https://doi.org/10.1007/s10008-017-3597-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3597-0