Abstract
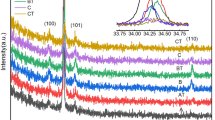
Aluminum-doped ZnO thin films with pebble-like structures have been successfully deposited on glass substrates by successive ionic layer adsorption reaction method. The effect of percentage composition of the aluminum dopant on the flower-like clusters of the ZnO nanostructures on the structure, morphology, and optical properties was investigated. The ZnO thin films which were crystallized in hexagonal wurtzite structures with crystallite sizes of 44, 51, 56, and 43 nm for the intrinsic and 1, 3, and 5% Al-doped ZnO thin films, respectively. Preferred orientation of crystallites is in all cases in [001] direction perpendicular to the sample surface The Raman spectroscopy revealed decrease in the intensity of the ZnO characteristic peak due to the substitution of the Zn2+ atoms by the Al3+ and attributed to potential fluctuations of the alloy disorder. The introduction of the Al3+ dopant significantly increased the optical band gap.

Graphical abstract







Similar content being viewed by others
References
Munan MY, Wang HXH, Pei Z, Gong J, Sun C (2006) Growth control of ZnO films on glass by chemical bath deposition. Surf Rev Lett 13:369–373
Ozgur U, Alivoc YI, Liu C, Teke A, Reshchikov MA, Dogan S, Avrutin V, Cho JS, Morkoc H (2005) A comprehensive review of ZnO materials and devices. J Appl Phys 98:0413011–0413103
Jimenez-Gonzalez AE (1997) Modification of ZnO thin films by Ni, Cu, and Cd Doping. J Solid State Chem 128:176–180
Zhang Q, Dandeneau CS, Zhou X, Cao G (2009) ZnO nanostructures for dye-sensitized solar cells. Adv Mater 21(41):4087–4108
Nishino J, Ohshio S, Kamata K (1992) Preparation of Aluminum-Doped Zinc Oxide Films by a Normal-Pressure CVD Method. J Am Ceram Soc 75(12):3469–3472
Mondal S, Bhattacharyya SR, Mitra P (2013) Effect of Al doping on microstructure and optical band gap of ZnO thin film synthesized by successive ion layer adsorption and reaction. Pramana J Phys 80(2):315–326
Pathan HM, Lockhande CD (2004) Deposition of metal chalcogenide thin films by successive ionic layer adsorption and reaction (SILAR) method. Bull Mater Sci 27(2):85–111
Gao XD, Li XM, Yu WD (2004) Synthesis and optical properties of ZnO nanocluster porous films deposited by modified SILAR method. Appl Surf Sci 229:275–281
Mondal S, Kanta KP, Mitra P (2008) Preparation of Al-doped ZnO (AZO) thin film by SILAR. J Phys Sci 12:221–229
Jambure SB, Patil SJ, Deshpande AR, Lokhande CD (2014) A comparative study of physico-chemical properties of CBD and SILAR grown ZnO thin films. Mater Res Bull 49:420–425
Kofstadt P (1983) Ch. 1 Nonstoichimetry, diffusion and electrical conductivity in binary metal oxides. Krieger, Malabar, FL, p 7
Kamalasanan MN, Chandra S (1996) Sol-gel synthesis of ZnO thin films. Thin Solid Films 288:112–115
Raidou A, Aggour M, Qachaou A, Laanab L, Fahoume M (2010) Preparation and characterisation of ZnO thin films deposited by SILAR method. J Condensed Matter 12(2):125–139
Amutha C, Dhanalakshmi A, Lawrence B, Kulathuraan K, Ramadas V, Natarajan B (2014) Influence of concentration on structural and optical characteristics of nanocrystalline ZnO thin films synthesized by Sol-Gel dip coating method. Progress in Nanotechnology and Nanomaterials 3:13–18
Harris GB (1952) X. Quantitative measurement of preferred orientation in rolled uranium bars. Philos Mag 43:113–123
Ma JG, Liu YC, Mu R, Zhang JY, Lu YM, Shen DZ, Fan XW (2004) Method of control of nitrogen content in ZnO films: Structure and photoluminescence properties. J Vac Sci Technol B 22:94–98
Zhang JP, Zhang LD, Zhu LQ, Zhang Y, Liu M, Wang XJ (2007) Characterization of ZnO: N films prepared by annealing sputtered zinc oxynitride films at different temperatures. J Appl Phys 102:1149031–1149037
Liu HF, Chua SJ, Hu GX, Gong H (2011) Deposition and Characterizations of ZnO Thin Films on Al2O3 (0001) Substrates with III-Arsenide Intermediating Layers. The Open Applied Physics Journal 4:41–44
Min Y, An C, Kim SK, Song J, Hwang CS (2010) Growth and Characterization of Conducting ZnO Thin Films by Atomic Layer Deposition. Bull Kor Chem Soc 31(9):2503–2508
Damen TC, Porto SPS, Tell B (1966) Raman Effect in Zinc Oxide. Phys Rev 142:570–574
Mei YF, Siu GG, Ricky KY, Paul K, Li ZM, Tang ZK (2006) Room-temperature electrosynthesized ZnO thin film with strong (002) orientation and its optical properties. Appl Surf Sci 252:2973–2977
Ursaki VV, Tiginyanu IM, Zalamai VV, Masalov VM, Samarov EN, Emelchenko GA, Briones F (2004) Photoluminescence and resonant Raman scattering from ZnO-opal structures. J Appl Phys 96:1001–1006
Calleja JM, Cardona M (1977) Resonant Raman scattering in ZnO. Phys Rev B 16:3753–3761
Samanta PK, Patra SK, Ghosh A, Chaudhuri PR (2009) Visible emission from ZnO nanorods synthesized by a simple wet chemical method. International Journal of NanoScience and Nanotechnology 1:81–90
Hammouda A, Canizares A, Simon P, Boughalout A, Kechouane M (2012) Improving the sensitivity of Raman signal of ZnO thin films deposited on silicon substrate. Vib Spectrosc 62:217–221
Desai MA, Sartale SD (2015) Facile soft solution route to engineer hierarchical morphologies of ZnO nanostructures. Cryst Growth Des 15(10):4813–4820. doi:10.1021/acs.cgd.5b00561
Al Asmar R, Atanas JP, Ajaka M, Zaatar Y, Ferblantier G, Sauvajol JL, Jabbour J, Juillaget S, Foucaran A (2005) Characterization and Raman investigations on high-quality ZnO thin films fabricated by reactive electron beam evaporation technique. J Cryst Growth 279:394–402
Khan ZR, Khan MS, Zulfequar M, Khan MS (2011) Optical and Structural Properties of ZnO Thin Films Fabricated by Sol-Gel Method. Mater Sci Appl 2:340–345
Merrigan JA (1982) Sunlight to electricity: photovoltaic technology for business prospectus. The Massachusetts Institute of Technology Press, Massachusetts
Richard CN (1978) Solar energy conversion: the solar cell. Elsevier Scientific Publishing Company, Amsterdam
Gümü C, Ozkendir OM, Kavak H, Ufuktepe Y (2006) Structural and optical properties of zinc oxide thin films prepared by spray pyrolysis method. J Optoelectron Adv Mater 8(1):299–303
Mitra P, Mondal S (2013) Structural and morphological characterization of ZnO thin films synthesized by SILAR. Progress in Theoretical and Applied Physics 1:17–31
Yazdanmehr M, Asadabadi SJ, Nourmohammadi A, Ghasemzadeh M, Rezvanian M (2012) Electronic structure and bandgap of ?-Al2O3 compound using mBJ exchange potential. Nanoscale Res Lett 7:4881–4891
Acknowledgements
We graciously acknowledge the grant for this project by TETFUND under contract number TETF/DESS/UNN/NSUKKA/STI/VOL.I/B4.33.The results were also partially developed within the CENTEM project, reg. no. CZ.1.05/2.1.00/03.0088 co-funded by the ERDF within the OP RDI programme, and in the follow-up sustainability stage, supported through CENTEM+ (LO1402) by financial means from the Czech Ministry of Education, Youth and Sports under the National Sustainability Programme I. We also thank Engr. Emeka Okwuosa for the generous sponsorship of April 2014 and July, 2016 conference/workshops on Applications of Nanotechnology to Energy, Health and Environment conference.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Offiah, S.U., Agbo, S.N., Sutta, P. et al. Study of the extrinsic properties of ZnO:Al grown by SILAR technique. J Solid State Electrochem 21, 2621–2628 (2017). https://doi.org/10.1007/s10008-017-3514-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3514-6