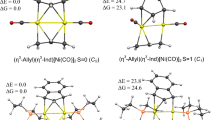
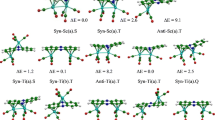
Abstract
This paper is focused on the examination of the bonding properties of a series of [M(NHC)2X2] (M = nd8 transition metal; X = Cl, Br and I) complexes in normal, abnormal and mixed C∩C coordination modes. Structures have been optimised in gas phase using B3LYP, M06 and P3BW91 functionals. Two basis sets have been used: the LanL2DZ and a mixed basis set (LanL2DZ for nd8 transition metals as well as halogen atoms and 6-311+G(d,p) for other atoms). Results obtained indicate that the B3PW91 bond distances are closer to experimental data. The complexation energies obtained for each binding mode increase in the order: Ni2+ < Pd2+ < Pt2+, independently of the halogen atom adopted. From the Quantum Theory of Atoms in Molecule (QTAIM) approach, the instability has been found to follow this trend: M − X < M − C. The analysis of metal-ligand interactions using the natural bond orbital (NBO) revealed that the strongest metal-ligand interactions are observed in the normal binding mode. The NCH → MX2 donation terms were found to be interestingly predominant compared with back donation ones in the complexes studied, except in Pt chloride ones. The contribution of electrostatic interaction energy between the above fragments (∆Eelstat term) is in the range 57.48–63.95% traducing the fact that the interactions are mostly electrostatic.

Graphical abstract






Similar content being viewed by others
References
Arduengo AJ, Harlow RL, Kline M (1991). J Am Chem Soc 113:361–363
Crudden CM, Allen DP (2004). Coord Chem Rev 248:2247–2273
Peris E, Crabtree RH (2004). Coord Chem Rev 248:2239–2246
Pugh D, Danopoulos AA (2007). Coord Chem Rev 251:610–641
Díez-González S, Marion N, Nolan SP (2009). Chem Rev 109:3612–3676
Visbal R, Gimeno MC (2014). Chem Soc Rev 43:3551–3574
Hopkinson MN, Richter C, Schedler M, Glorius F (2014). Nature 510:485–496
Herrmann WA (2002). Angew Chem Int Ed 41:1290–1309
Munz D, Meyer D, Strassner T (2013). Organometallics 32:3469–3480
Ingrosso G, Midollini N (2003). J Mol Catal A-Chem 204-205:425–431
Munz D, Strassner T (2014). Top Catal 57:1372–1376
Muehlhofer M, Strassner T, Herrmann WA (2002). Angew Chem Int Ed 41:1745–1747
Liu JQ, Gou XX, Han YF (2018). Chem Asian J 13:2257–2276
Hindi KM, Panzner MJ, Tessier CA, Cannon CL, Youngs WJ (2009). Chem Rev 109:3859–3884
Gautier A, Cisnetti F (2012). Metallomics 4:23–32
Cisnetti F, Gautier A (2013). Angew Chem Int Ed Eng 52:11976–11978
Liu W, Gust R (2013). Chem Soc Rev 42:755–773
Nolan SP (2011). Acc Chem Res 44:91–100
Johnson NA, Southerland MR, Youngs WJ (2017). Molecules 22(8):1263–1283
Chardon E, Puleo GL, Dahm G, Fournel S, Guichard G, Bellemin-Laponnaz S (2012). ChemPlusChem 77:1028–1038
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, Castedo M, Kroemer G (2012). Oncogene 31:1869–1883
Ahrens S, Zeller A, Taige M, Strassner T (2006). Organometallics 25:5409–5415
Poverenov E, Efremenko I, Leitus G, Martin JM, Milstein D (2013). Organometallics 32:4813–4819
Hou X-F, Wang Y-N, Gottker-Schnetmann I (2011) N-Heterocyclic carbene palladium catalyzed regioselective oxidative trifluoroacetoxylation of unactivated methylene sp3 C–H bonds in linear alkyl esters. Organometallics 30:6053–6056. https://doi.org/10.1021/om200484g
Micksch M, Strassner T (2012) Palladium(II) complexes with chelating biscarbene ligands in the catalytic Suzuki–Miyaura cross coupling reaction. Eur J Inorg Chem 2012:5872–5880. https://doi.org/10.1002/ejic.201200940
Nonnenmacher M, Kunz D, Rominger F, Oeser T (2007). J Organomet Chem 692:2554–2563
McCall AS, Wang H, Desper JM, Kraft S (2011). J Am Chem Soc 133:1832–1848
Jacobsen H, Correa A, Poater A, Cavallo L (2009). Coord Chem Rev 253:687–703
Jagadeesan R, Velmurugan G, Venuvanalingam P (2015). RSC Adv 5:80661–80667
Munz D, Strassner T (2014). Angew Chem Int Ed 53:2485–2488
Meyer D, Taige MA, Zeller A, Hohlfeld K, Ahrens S, Strassner T (2009). Organometallics 28:2142–2149
Taige MA, Zeller A, Ahrens S, Goutal S, Herdtweck E, Strassner T (2007). J Organomet Chem 692:1519–1529
Strassner T, Muehlhofer M, Zeller A, Herdtweck E, Herrmann WA (2004). J Organomet Chem 689:1418–1424
Weinhold F, Klein RA (2012). Mol Phys 110:565–579
Schreckenbach G, Ziegler TJ, Li J (1995). Int J Quantum Chem 56:477–488
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JRMontgomery Jr JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi JJ, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth A, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2009) Gaussian 09 Revision B.04. Gaussian Inc, Pittsburgh: PA (licence g09)
Ivanciuc O, Balaban AT (1998) Graph theory in chemistry. In: PVR S, Allinger NL, Kollmann PA, Clark T, HFS S, Gasteiger J, Schreiner PR (eds) The encyclopedia of computational chemistry. John Wiley & Sons, Chichester 1169–1190
Lu T, Chen F (2012). J Comput Chem 33:580–592
Reed AE, Curtiss LA, Weinhold F (1988). Chem Rev 88:899–926
Xiao M, Lu T (2015). J Adv Phys Chem 4:111–124
Parr RG, Szentpaly LV, Liu S (1999). J Am Chem Soc 12:1922–1924
Parr RG, Yang W (1989) Density-functional theory of atoms and molecules. Oxford University Press, New York
Pearson RG (1997) Chemical hardness. Wiley-VCH, Oxford
Morokuma K (1971). J Chem Phys 55:1236–1244
Kitaura K, Morokuma KA (1976). Int J Quantum Chem 10:325–340
Ziegler T, Rauk A (1977). Theoret Chim Acta 46:1–10
Ruedenberg K (1962). Rev Mod Phys 34:326–376
Heitler W, London F (1927). Z Phys 44:455–472
Clark T, Murray JS, Politzer P (2018). Phys Chem Chem Phys 20:30076–30082
Morokuma K, Kitaura K (1981) In: Politzer P, Truhlar DG (eds) Chemical applications of atomic and molecular electrostatic potentials. Plenum, New York, pp 215–242
Politzer P, Murray JS, Clark T (2015). J Mol Model 21:1–10
Heckenroth M, Kluser E, Neels A, Albrecht M (2008). Dalton Trans 44:6242–6249
Shimojo F, Wu Z, Nakano A, Kalia RK, Vashishta P (2010). J Chem Phys 132:1–7
Perdew JP, Yue W (1989). Phys Rev B 40:3399–3399
Wysokinski R, Michalska D (2001). J Comput Chem 22:901–912
Herrmann WA, Reisinger C-P, Spiegler M (1998). J Organomet Chem 557:93–96
Lebel H, Janes MK, Charette AB, Nolan SP (2004). J Am Chem Soc 126:5046–5047
Chianese AR, Crabtree RH (2004). ACS Symp Ser 885:169–183
Chianese AR, Kovacevic A, Zeglis BM, Faller JW, Crabtree RH (2004). Organometallics 23:2461–2468
Bayat M, Yaghoobi F, Salehzadeh S, Hokmi S (2011). Polyhedron 30:2809–2814
Samantaray MK, Roy D, Patra A, Saikh M, Sunoj RB, Ghosh P (2006). J Organomet Chem 691:3797–3805
De Bruin TJ, Milet A, Greene AE, Gimbert Y (2004). J Organomet Chem 69:1075–1080
Vyboishchikov SF, Frenking G (1998). Chem Eur J 4:1428–1438
Boehme C, Frenking G (1998). Organometallics. 17:5801–5809
Elschenbroich C, Salzer A (1992) Organometallics, a concise introduction2nd edn. VCH Press, Weinheim, Germany
Bickelhaupt FM, Baerends EJ (2000). Rev Comput Chem 15:1–86
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 208 kb)
Rights and permissions
About this article
Cite this article
Moto Ongagna, J., Tamafo Fouegue, A.D., Ateba Amana, B. et al. B3LYP, M06 and B3PW91 DFT assignment of nd8 metal-bis-(N-heterocyclic carbene) complexes. J Mol Model 26, 246 (2020). https://doi.org/10.1007/s00894-020-04500-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-04500-7