Abstract
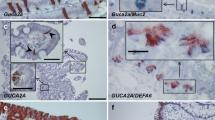
Brush cells (BCs) are relatively rare cells that are sparsely distributed throughout the mammalian digestive and respiratory systems. BCs have been identified in the rodent large intestine, but these cells have not been characterized by immunocytochemistry or electron microscopy. We previously demonstrated that rat bile duct BCs had strong immunoreactivity for six proteins that function in HCO −3 secretion and thus assumed that BCs secrete NaHCO3. It is well known that the gastrointestinal (GI) tract secretes NaHCO3, but it is not known whether BCs of the GI tract also express proteins related to HCO −3 secretion. Thus, in the present study, using double immunostaining for cytokeratin 18, a specific marker for BCs, we investigated protein expression in BCs from the rodent GI tract. We show that BCs from the GI tract express six proteins related to HCO −3 secretion: cystic fibrosis transmembrane conductance regulator (CFTR), Cl−/HCO −3 exchanger, Na+/HCO −3 cotransporter, carbonic anhydrase II, Na+/H+ exchanger (NHE) 1, and NHE3. These results suggest that BCs from the GI tract secrete NaHCO3. In addition, we examined BCs from the rat cecum using electron microscopy (EM). Transmission EM (TEM) showed that BCs have long microvilli, a well-developed tubulovesicular system, and an abundant cytoskeleton. Scanning EM revealed that BCs were scattered on the luminal surface of the cecum and had numerous long microvilli.
Similar content being viewed by others
References
Rhodin JAG, Dalhamn T (1956) Electron microscopy of the tracheal ciliated mucosa in rat. Z Zellforsch 44:345–412
Ogata T (2000) Mammalian tuft (brush) cells and chloride cells of other vertebrate share a similar structure and cytochemical reactivities. Acta Histochem Cytochem 33:439–449
Sbarbati A, Osculati F (2005) A new fate for old cells: brush cells and related elements. J Anat 206:349–358
Sato A (2007) Tuft cells. Anat Sci Int 82f:187–199
Isomäki AM (1973) A new cell type (tuft cell) in the gastrointestinal mucosa of the rat. Acta Pathol Microbiol Scand Sect A (Suppl) 240:1–35
Nabeyama A, Leblond CP (1974) “Caveolated cells” characterized by deep surface invaginations and abundant filaments in mouse gastro-intestinal epithelia. Am J Anat 140:147–166
Sato A, Miyoshi S (1997) Fine structure of tuft cells of the main excretory duct epithelium in the rat submandibular gland. Anat Rec 248:325–331
Luciano L, Groos S, Reale E (2003) Brush cells of rodent gallbladder and stomach epithelia express neurofilaments. J Histochem Cytochem 51:187–198
Hijiya K (1978) Electron microscope study of the alveolar brush cell. J Electron Microsc 27:223–227
Höfer D, Drenckhahn D (1998) Identification of the taste cell Gprotein, α-gustducin, in brush cells of the rat pancreatic duct system. Histochem Cell Biol 110:303–309
Ogata T (2005) Sodium bicarbonate secretion indicated by ultrastructural cytochemical localization of HCO −3 , Cl−, and Na+ ions on rat bile duct brush cells. Med Mol Morphol 38:243–250
Ogata T (2006) Bicarbonate secretion by rat bile duct brush cells indicated by immunohistochemical localization of CFTR, anion exchanger AE2, Na+/HCO −3 cotransporter, carbonic anhydrase II, Na+/H+ exchangers NHE1 and NHE3, H+/K+-ATPase, and Na+/K+-ATPase. Med Mol Morphol 39:44–48
Argent BE, Case RM (1994) Pancreatic ducts. Cellular mechanism and control of bicarbonate secretion. In: Johnson LR (ed) Physiology of the gastrointestinal tract, 3rd edn. Raven Press, New York, pp 1473–1497
Marino CR, Gorelick FS (2003) Pancreatic and salivary glands. In: Boron WF, Boulpaep EL (eds) Medical physiology. Saunders, Philadelphia, pp 908–930
Steward MC, Ishiguro H, Case RM (2005) Mechanisms of bicarbonate secretion in the pancreatic duct. Annu Rev Physiol 67:377–409
Höfer D, Drenckhahn D (1996) Cytoskeletal markers allowing discrimination between brush cells and other epithelial cells of the gut including enteroendocrine cells. Histochem Cell Biol 105:405–412
Flemström G, Garner A (1982) Gastroduodenal HCO −3 transport: characteristics and proposed role in acidity regulation and mucosal protection. Am J Physiol 242:G183–G193
Kawamata K, Hayashi H, Suzuki Y (2006) Chloride-dependent bicarbonate secretion in the mouse large intestine. Biomed Res 27:15–21
Hatta S, Sakamoto J, Horio Y (2002) Ion channels and diseases. Med Electron Microsc 35:117–126
Roussa E, Romero MF, Schmitt BM, Boron WF, Alper SL, Thévenod F (1999) Immunolocalization of anion exchanger AE2 and Na+-HCO −3 cotransporter in rat parotid and submandibular glands. Am J Physiol 277:G1288–G1296
Roussa E, Alper SL, Thévenod F (2001) Immunolocalization of anion exchanger AE2, Na+/H+ exchangers NHE1 and NHE4, and vacuolar type H+-ATPase in rat pancreas. J Histochem Cytochem 49:463–474
Marino CR, Jeanes V, Boron WF, Schmitt BM (1999) Expression and distribution of the Na+-HCO −3 cotransporter in human pancreas. Am J Physiol 277:G487–G494
Thévenod F, Roussa E, Schmitt BM, Romero MF (1999) Cloning and immunolocalization of a rat pancreatic Na+ bicar bonate cotransporter. Biochem Biophys Res Commun 264: 291–298
Satoh H, Moriyama N, Hara C, Yamada H, Horita S, Kunimi M, Tsukamoto K, Iso-o N, Inatomi J, Kawakami H, Kudo A, Endou H, Igarashi T, Goto A, Fujita T, Seki G (2003) Localization of Na+-HCO −3 cotransporter (NBC-1) variants in rat and human pancreas. Am J Physiol 284:C729–C737
Kumpulainen T, Jalovaara P (1981) Immunohistochemical localization of carbonic anhydrase isoenzymes in the human pancreas. Gastroenterology 80:796–799
Parkkila S, Parkkila AK, Juvonen T, Rajaniemi H (1994) Distribution of the carbonic anhydrase isoenzymes I, II, and VI in the human alimentary tract. Gut 35:645–650
Spicer SS, Ge ZH, Tashian RE, Hazen-Martin DJ, Schulte BA (1990) Comparative distribution of carbonic anhydrase isozymes III and II in rodent tissues. Am J Anat 187:55–64
Wattel W, Geuze JJ (1978) The cells of the rat gastric groove and cardia. An ultrastructural and carbohydrate histochemical study, with special reference to the fibrillovesicular cells. Cell Tissue Res 186:375–391
Luciano L, Reale E, Ruska H (1968) Über eine glykogenhaltige Bürstenzelle in Rectum der Ratte. Z Zellforsh 91:153–158
Sato A, Miyoshi S (1988) Ultrastructure of the main excretory duct epithelia of the rat parotid and submandibular glands with a review of the literature. Anat Rec 220:239–251
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okamoto, K., Hanazaki, K., Akimori, T. et al. Immunohistochemical and electron microscopic characterization of brush cells of the rat cecum. Med Mol Morphol 41, 145–150 (2008). https://doi.org/10.1007/s00795-008-0412-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-008-0412-0