Abstract
Objectives
To evaluate the potential of conventional glass ionomer cement (GIC), Biodentine™, MTA, and Portland cement to induce mineral density changes in carious dentin compared to zinc oxide eugenol control cement (ZOE).
Materials and methods
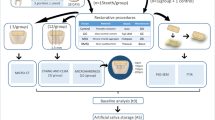
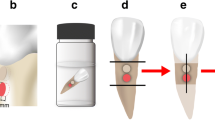
Fifty blocks of bovine root dentin were prepared and a biofilm model using ATCC strains of S.mutans, S.sobrinus, and L.casei was used to promote artificial dentin lesions. After demineralization, the blocks were randomly divided into the five cement groups. Half of the surface of each specimen received the tested material and the other half was covered with wax (control). Samples were stored in phosphate buffered saline solution for 30 days and after that were scanned in a micro-CT with standardized parameters. Dentin mineral density changes were calculated using differences in plot profiles of the exposed and control carious dentin. Friedman’s test, followed by Wilcoxon signed-rank test was used with 5% significance.
Results
Mean ΔZ values for the cements were 48.63 ± 19.09 for the control (ZOE), 63.31 ± 32.59 for Biodentine™, 114.63 ± 72.92 for GIC, 109.56 ± 66.28 for MTA, and 106.88 ± 66.02 for Portland cement. All cements showed a statistically significant increase in ΔZ values compared to the control, but Biodentine™ values were statistically significantly lower compared to GIC and the other calcium silicate cements.
Conclusions
Tested materials present potential to induce mineral density changes in carious bovine dentin. MTA, Portland, and GIC showed higher bioactivity potential than Biodentine™.
Clinical relevance
Based on minimally invasive concept, materials with remineralization potential can be used to preserve diseased but still repairable dental tissue.



Similar content being viewed by others
References
Banerjee A (2013) Minimal intervention dentistry: part 7. Minimally invasive operative caries management: rationale and techniques. Br Dent J 214(3):107–111. https://doi.org/10.1038/sj.bdj.2013.106
Dai L, Liu Y, Salameh Z, Khan S, Mao J, Pashley DH, Tay FR (2011) Can caries-affected dentin be completely remineralized by guided tissue remineralization? Dent Hypotheses 2(2):74–82. https://doi.org/10.5436/j.dehy.2010.1.00011
Hamama HH, Yiu CK, Burrow MF (2015) Caries management: a journey between Black’s principals and minimally invasive concepts. Int J Dent Oral Sci 2(8):120–125
Watson TF, Atmeh AR, Sajini S, Cook RJ, Festy F (2014) Present and future of glass-ionomers and calcium-silicate cements as bioactive materials in dentistry: biophotonics-based interfacial analyses in health and disease. Dent Mater 30(1):50–61. https://doi.org/10.1016/j.dental.2013.08.202
Vallittu PK, Boccaccini AR, Hupa L, Watts DC (2018) Bioactive dental materials-do they exist and what does bioactivity mean? Dent Mater 34(5):693–694. https://doi.org/10.1016/j.dental.2018.03.001
Goldberg M, Kulkarni AB, Young M, Boskey A (2011) Dentin: structure, composition and mineralization. Front Biosci 3:711–735
Liu Y, Li N, Qi Y, Niu LN, Elshafiy S, Mao J, Breschi L, Pashley DH, Tay FR (2011) The use of sodium trimetaphosphate as a biomimetic analog of matrix phosphoproteins for remineralization of artificial caries-like dentin. Dent Mater 27(5):465–477. https://doi.org/10.1016/j.dental.2011.01.008
Koc Vural U, Kiremitci A, Gokalp S (2017) Randomized clinical trial to evaluate MTA indirect pulp capping in deep caries lesions after 24-months. Oper Dent 42(5):470–477. https://doi.org/10.2341/16-110-C
Kind L, Stevanovic S, Wuttig S, Wimberger S, Hofer J, Muller B, Pieles U (2017) Biomimetic remineralization of carious lesions by self-assembling peptide. J Dent Res 96(7):790–797. https://doi.org/10.1177/0022034517698419
Okamoto M, Takahashi Y, Komichi S, Ali M, Yoneda N, Ishimoto T, Nakano T, Hayashi M (2018) Novel evaluation method of dentin repair by direct pulp capping using high-resolution micro-computed tomography. Clin Oral Investig. https://doi.org/10.1007/s00784-018-2374-5
Han L, Okiji T (2011) Uptake of calcium and silicon released from calcium silicate-based endodontic materials into root canal dentine. Int Endod J 44(12):1081–1087. https://doi.org/10.1111/j.1365-2591.2011.01924.x
Laird NM, Mosteller F (1990) Some statistical methods for combining experimental results. Int J Technol Assess Health Care 6(1):5–30
Cohen J (1992) A power primer. Psychol Bull 112(1):155–159
Niu LN, Zhang W, Pashley DH, Breschi L, Mao J, Chen JH, Tay FR (2014) Biomimetic remineralization of dentin. Dent Mater 30(1):77–96. https://doi.org/10.1016/j.dental.2013.07.013
Camilleri J, Sorrentino F, Damidot D (2013) Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement, Biodentine and MTA Angelus. Dent Mater 29(5):580–593. https://doi.org/10.1016/j.dental.2013.03.007
Niu LN, Jiao K, Wang TD, Zhang W, Camilleri J, Bergeron BE, Feng HL, Mao J, Chen JH, Pashley DH, Tay FR (2014) A review of the bioactivity of hydraulic calcium silicate cements. J Dent 42(5):517–533. https://doi.org/10.1016/j.jdent.2013.12.015
Reyes-Carmona JF, Felippe MS, Felippe WT (2009) Biomineralization ability and interaction of mineral trioxide aggregate and white Portland cement with dentin in a phosphate-containing fluid. J Endod 35(5):731–736. https://doi.org/10.1016/j.joen.2009.02.011
Simila HO, Karpukhina N, Hill RG (2018) Bioactivity and fluoride release of strontium and fluoride modified Biodentine. Dent Mater 34(1):e1–e7. https://doi.org/10.1016/j.dental.2017.10.005
Pires PM, Santos TP, Fonseca-Goncalves A, Pithon MM, Lopes RT, Neves AA (2018) Mineral density in carious dentine after treatment with calcium silicates and polyacrylic acid based cements. Int Endod J. https://doi.org/10.1111/iej.12941
Corral Nunez C, Covarrubias C, Fernandez E, Oliveira OBJ (2017) Enhanced bioactive properties of BiodentineTM modified with bioactive glass nanoparticles. J Appl Oral Sci 25(2):177–185. https://doi.org/10.1590/1678-77572016-0209
Ngo HC, Mount G, Mc Intyre J, Tuisuva J, Von Doussa RJ (2006) Chemical exchange between glass-ionomer restorations and residual carious dentine in permanent molars: an in vivo study. J Dent 34(8):608–613. https://doi.org/10.1016/j.jdent.2005.12.012
Baig MS, Dowling AH, Cao X, Fleming GJ (2015) A discriminatory mechanical testing performance indicator protocol for hand-mixed glass-ionomer restoratives. Dent Mater 31(3):273–283. https://doi.org/10.1016/j.dental.2014.12.012
Amaral MT, Guedes-Pinto AC, Chevitarese O (2006) Effects of a glass-ionomer cement on the remineralization of occlusal caries--an in situ study. Braz Oral Res 20(2):91–96
Beiruti N, Frencken JE, van ’t Hof MA, van Palenstein Helderman WH (2006) Caries-preventive effect of resin-based and glass ionomer sealants over time: a systematic review. Community Dent Oral Epidemiol 34(6):403–409. https://doi.org/10.1111/j.1600-0528.2006.00321.x
ten Cate JM, van Duinen RN (1995) Hypermineralization of dentinal lesions adjacent to glass-ionomer cement restorations. J Dent Res 74(6):1266–1271. https://doi.org/10.1177/00220345950740060501
Kim YK, Yiu CK, Kim JR, Gu L, Kim SK, Weller RN, Pashley DH, Tay FR (2010) Failure of a glass ionomer to remineralize apatite-depleted dentin. J Dent Res 89(3):230–235. https://doi.org/10.1177/0022034509357172
Li X, De Munck J, Van Landuyt K, Pedano M, Chen Z, Van Meerbeek B (2017) How effectively do hydraulic calcium-silicate cements re-mineralize demineralized dentin. Dent Mater 33(4):434–445. https://doi.org/10.1016/j.dental.2017.01.015
Malkondu O, Karapinar Kazandag M, Kazazoglu E (2014) A review on biodentine, a contemporary dentine replacement and repair material. Biomed Res Int 2014:160951–160910. https://doi.org/10.1155/2014/160951
Maske TT, van de Sande FH, Arthur RA, Huysmans M, Cenci MS (2017) In vitro biofilm models to study dental caries: a systematic review. Biofouling 33(8):661–675. https://doi.org/10.1080/08927014.2017.1354248
Swain MV, Xue J (2009) State of the art of micro-CT applications in dental research. International J Oral Sci 1(4):177–188. https://doi.org/10.4248/IJOS09031
Lee HS, Berg JH, Garcia-Godoy F, Jang KT (2008) Long-term evaluation of the remineralization of interproximal caries-like lesions adjacent to glass-ionomer restorations: a micro-CT study. Am J Dent 21(2):129–132
Neves AA, Lourenco RA, Alves HD, Lopes RT, Primo LG (2015) Caries-removal effectiveness of a papain-based chemo-mechanical agent: a quantitative micro-CT study. Scanning 37(4):258–264. https://doi.org/10.1002/sca.21206
Yassen GH, Platt JA, Hara AT (2011) Bovine teeth as substitute for human teeth in dental research: a review of literature. J Oral Sci 53(3):273–282
Teruel Jde D, Alcolea A, Hernandez A, Ruiz AJ (2015) Comparison of chemical composition of enamel and dentine in human, bovine, porcine and ovine teeth. Arch Oral Biol 60(5):768–775. https://doi.org/10.1016/j.archoralbio.2015.01.014
Acknowledgments
The authors are grateful to the Department of Pediatric Dentistry and Orthodontics in Federal University of Rio de Janeiro (UFRJ) and Laboratory for Nuclear Instrumentation (UFRJ).
Funding
The work was supported by the Department of Pediatric Dentistry and Orthodontics and Laboratory for Nuclear Instrumentation of Federal University of Rio de Janeiro. This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Pesquisa Científica (CNPq), and Fundação Carlos Chagas de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Neves, A.B., Bergstrom, T.G., Fonseca-Gonçalves, A. et al. Mineral density changes in bovine carious dentin after treatment with bioactive dental cements: a comparative micro-CT study. Clin Oral Invest 23, 1865–1870 (2019). https://doi.org/10.1007/s00784-018-2644-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-018-2644-2