Abstract
Objective
The aim of this study was to evaluate the immunohistochemical expressions of PD1, CD4+, and CD8+ in premalignant lesions (OPML) that were transformed into oral squamous cell carcinoma OSCC (OPML-OSCC), in OSCC and also in premalignant lesions that were not transformed into OSCC (OPML-NOSSC).
Materials and methods
Retrospective analyses were performed in order to verify the demographic characteristics of the patients. CD4, CD8, and PD1 IMH studies were carried out on OPML and OSCC samples from 11 patients with OPML-OSCC and OPML, together with samples from 14 patients with OPML-NOSCC. The differences between OPML-OSCC and OPML-NOSCC were analyzed.
Results
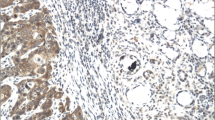
Non-homogenous leukoplakia, together with the related oral subsite, and the lack of an exposure to tobacco, were all associated with malignant transformations. There were no statistical differences in the PD1 expression and the CD4+ cells in OPML-OSCC and OPML-NOSCC. A significant increment in the CD8+ cells was noted in the OPML that evolved into carcinomas when compared with OPML-NOSCC (p = 0.05), whereas there were higher CD8+ cells levels in the carcinomas when compared with the OPML that evolved into carcinomas (p = 0.027).
Conclusions
CD8+ cells infiltrate more in OPML-NOSCC than in OPML-OSCC. Carcinoma is more infiltrated by CD8+ cells than its associated OPML.
Clinical relevance
Understanding immunological factors associated with malignant transformation of oral premalignant lesions can open a new way to treat this disease.


Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917. https://doi.org/10.1002/ijc.25516
Neville B, Damm D, Allen C, Bouquot J (2009) Oral and maxillofacial pathology. Saunders/Elsevier, St. Louis
Warnakulasuriya S1, Johnson NW, van der Waal I (2007 Nov) Nomenclature and classification of potentially malignant disorders of the oral mucosa. J Oral Pathol Med 36(10):575–580. https://doi.org/10.1111/j.1600-0714.2007.00582.x
Napier SS, Speight PM (2008) Natural history of potentially malignant oral lesions and conditions: an overview of the literature. J Oral Pathol Med 37:1–10. https://doi.org/10.1111/j.1600-0714.2007.00579.x
Wojtowicz ME, Dunn BK, Umar A (2016) Immunologic approaches to cancer prevention-current status, challenges, and future perspectives. Semin Oncol 43:161–172. https://doi.org/10.1053/j.seminoncol.2015.11.001
Willian WNJ (2012) Oral premalignant lesions: any progress with systemic therapies? Curr Opin Oncol 24:205–210. https://doi.org/10.1097/CCO.0b013e32835091bd
Quan H, Fang L, Pan H, Deng Z, Gao S, Liu O, Wang Y, Hu Y, Fang X, Yao Z, Guo F, Lu R, Xia K, Tang Z (2016) An adaptive immune response driven by mature, antigen-experienced T and B cells within the microenvironment of oral squamous cell carcinoma. Int J Cancer 138:2952–2962. https://doi.org/10.1002/ijc.30019
Kaur J, Jacobs R (2015 Aug) Proinflammatory cytokine levels in oral lichen planus, oral leukoplakia, and oral submucous fibrosis. J Korean Assoc Oral Maxillofac Surg 41(4):171–175. https://doi.org/10.5125/jkaoms.2015.41.4.171
Mori K, Haraguchi S, Hiori M, Shimada J, Ohmori Y (2015) Tumor-associated macrophages in oral premalignant lesions coexpress CD163 and STAT1 in a Th1-dominated microenvironment. BMC Cancer 15:573. https://doi.org/10.1186/s12885-015-1587-0
Lee JJ, Lin CL, Chen TH, Kok SH, Chang MC, Jeng JH. Changes in peripheral blood lymphocyte phenotypes distribution in patients with oral cancer/oral leukoplakia in Taiwan Int J Oral Maxillofac Surg 2010 39(8):806-814. doi: https://doi.org/10.1016/j.ijom.2010.04.045.
Fan CC, Wang TY, Cheng YA, Jiang SS, Cheng CW, Lee AY, Kao TY (2013) Expression of E-cadherin, twist and p53 and their prognostic value in patients with oral squamous cell carcinoma. J Cancer Res Clin Oncol 139:1735–1744. https://doi.org/10.1007/s00432-013-1499-9
Rhodus NL, Kerr RA, Patel K (2014) Leukoplakia, premalignancy and squamous cell carcinoma. Dent Clin N Am 58:315–340. https://doi.org/10.1016/j.cden.2013.12.004
Anderson A, Ishak N (2015) Marked variation in malignant transformation rates of oral leukoplakia. Evid-Based Dent 16:102–103. https://doi.org/10.1038/sj.ebd.6401128
Dietrich T, Reichart PA, Scheifele C (2004) Clinical risk factors of oral leukoplakia in a representative sample of the US population. Oral Oncol 40:158–163. https://doi.org/10.1016/S1368-8375(03)00145-3
Howlader N, Noone AM, Krapcho M, Miller D, Bishop K, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA (eds). SEER Cancer statistics review, 1975-2013, National Cancer Institute
Johnson SD, Levingston C, Young MRI (2016) Premalignant oral lesion cells elicit increased cytokine production and activation of T-cells. Anticancer Res 36:3261–3270
Woodford D, Johnson SD, Costa AM, Young MR (2014) An inflammatory cytokine milieu is prominent in premalignant oral lesions, but subsides when lesions progress to squamous cell carcinoma. J Clin Cell Immunol 5:1–17. https://doi.org/10.4172/2155-9899.1000230
Badoual C, Hans S, Rodriguez J, Peyrard S, Klein C, Agueznay NEH, Mosseri V, Laccourreye O, Bruneval P, Fridman WH, Brasnu DF, Tartour E (2006) Prognostic value of tumour-infiltrating CD4+ T-cell subpopulations in head and neck cancers. Clin Cancer Res 12:465–472. https://doi.org/10.1158/1078-0432.CCR-05-1886
Öhman J, Mowjood R, Larsson L, Kovacs A, Magnusson B, Kjeller G, Jontell M, Hasseus B (2015) Presence of CD3-positive T-cells in oral premalignant leukoplakia indicates prevention of cancer transformation. Anticancer Res 35:311–318
Piva MR, Souza LB, Martins-Filho PR, Soares RC, Santos TS, Andrade ESS (2011) Role of inflammation in oral carcinogenesis (part I): histological grading of malignancy using a binary system. Oncol Lett 2:1225–1231. https://doi.org/10.3892/ol.2011.382
Maleki S, Schlecht NF, Keller C, Diaz J, Moss J, Prystowsky MB, Macian F, Brandwein-Gensler M (2011) Lymphocytic host response to oral squamous cell carcinoma: an adaptive T-cell response at the tumour interface. Head Neck Pathol 5:117–122. https://doi.org/10.1007/s12105-011-0247-1
Gannot G, Gannot I, Vered H, Buchner A, Keisari Y (2002) Increase in immune cell infiltration with progression of oral epithelium from hyperkeratosis to dysplasia and carcinoma. Br J Cancer 86:1444–1448
Aghbari SMH, Abushouk AI, Shakir OG, Zayed SO, Attia A. Correlation between tissue expression of microRNA-137 and CD8 in oral lichen planus.Clin Oral Investig [15 Oct 2017, 22(3):1463–1467]. DOI: https://doi.org/10.1007/s00784-017-2252-6
Enomoto A, Sato E, Yasuda T, Isomura T, Nagao T, Chikazu D (2018 Apr) Intraepithelial CD8+ lymphocytes as a predictive diagnostic biomarker for the remission of oral lichen planus. Hum Pathol 74:43–53. https://doi.org/10.1016/j.humpath.2017.12.008
Brahmbhatt B, Vora H, Shah B (2016) Infiltration of T cell subsets during oral carcinogenesis. Indian J Oral Sci 7:28–41
Cho YA, Yoon HJ, Lee JI, Hong SP, Hong SD (2011) Relationship between the expressions of PD-L1 and tumour-infiltrating lymphocytes in oral squamous cell carcinoma. Oral Oncol 47:1148–1153. https://doi.org/10.1016/j.oraloncology.2011.08.007
Lin YM, Sung WW, Hsieh MJ, Tsai SC, Lai HW, Yang SM, Shen KH, Chen MK, Lee H, Yeh KT, Chen CJ (2015) High PD-L1 expression correlates with metastasis and poor prognosis in oral Ssquamous cell carcinoma. PLoS One 10:e0142656. https://doi.org/10.1371/journal.pone.0142656
Straub M, Drecoll E, Pfarr N, Weichert W, Langer R, Hapfelmeier A, Götz C, Wolff KD, Kolk A, Specht K (2016) CD274/PD-L1 gene amplification and PD-L1 protein expression are common events in squamous cell carcinoma of the oral cavity. Oncotarget 7: 12024–12034. Doi: https://doi.org/10.18632/oncotarget.7593
Ryan BM, Faupel-Badger JM (2016) The hallmarks of premalignant conditions: a molecular basis for cancer prevention. Semin Oncol 43:22–35
Funding
This study was supported by grants from the FAPEMIG (Grant n. RED-00011-14).
Author information
Authors and Affiliations
Contributions
RIMAR, LVM, ALFC conceptualized and designed the study; LVM, ALFC, JMAS, AML, and AAS performed clinical examinations of the patients; RIMAR, LVM, and AGS performed statistical analyses, analyzed and interpreted the data, and drafted the manuscript; RIMAR, EFCM, and MAM acquire financing; LFBP, GFL, and FMM executed the immunohistochemical analysis and revised the medical data. HBS contributed to the analysis, drafted illustration, and revised the manuscript. All authors revised the manuscript for important content and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author declare that they have no conflict of interest.
Ethical approval
The clinical data of the patients were obtained from the medical records. Informed consent was obtained from living patients and the study was performed in accordance with the Declaration of Helsinki. This study also was approved by the Ethical Committee of the Federal University of São João del-Rei, Brazil (Protocol Number: 940876–2015).
Informed consent
Written informed consent was obtained from all patients when alive, and the clinical data were obtained from the medical records. Since this was a retrospective archive study, the Ethics Committee waived the need for an informed consent, and a policy of strict anonymity and confidentiality were assured. All information obtained was used exclusively for the purpose of this study and was not shared with other individuals or organizations.
Rights and permissions
About this article
Cite this article
Chaves, A.L.F., Silva, A.G., Maia, F.M. et al. Reduced CD8+ T cells infiltration can be associated to a malignant transformation in potentially malignant oral epithelial lesions. Clin Oral Invest 23, 1913–1919 (2019). https://doi.org/10.1007/s00784-018-2622-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-018-2622-8