Abstract.
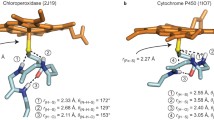
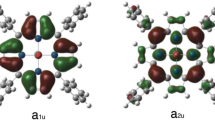
Optical absorption and resonance Raman spectra have been investigated for enzymatic intermediates, compounds I and II, of chloroperoxidase (CPO) which contains a thiolate-ligated iron porphyrin. Compound I of CPO (CPO-I), an oxoferryl porphyrin π cation radical, gave an apparently asymmetric single-peaked Soret band at 367 nm, for which band fitting analyses revealed the presence of two transition bands around 365 and 415 nm. Compound II of CPO (CPO-II), an oxoferryl neutral porphyrin, gave a split Soret spectrum with two bands (blue and red Soret bands) at 373 and 436 nm. Thus both CPO-I and CPO-II can be categorized as hyperporphyrins. The maximum extinction coefficients (εb and εr) and energies (E b and E r) of the blue and red Soret bands of CPO-II were found to fall on an εb/εr versus E b–E r correlation line derived from data reported for six-coordinate ferrous derivatives of cytochrome P450 and CPO. Corresponding data for CPO-I did not fall on the correlation line. Resonance enhancement of the FeIV=O stretching (νFeO) Raman band was found for CPO-I when Raman scattering was excited at wavelengths within both transition bands around 365 and 415 nm, while the νFeO Raman band was not identified for CPO-II at any of the excitation wavelengths examined here. These findings suggest that the thiolate axial ligand causes Soret band splitting of CPO-II through configuration interaction between the sulfur→porphyrin eg* charge transfer and porphyrin a1u,a2u→eg* transitions, while the FeO portion is important in determining the shape of the Soret band of CPO-I.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Egawa, T., Proshlyakov, D., Miki, H. et al. Effects of a thiolate axial ligand on the π→π* electronic states of oxoferryl porphyrins: a study of the optical and resonance Raman spectra of compounds I and II of chloroperoxidase. J. Biol. Inorg. Chem. 6, 46–54 (2001). https://doi.org/10.1007/s007750000181
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s007750000181