Abstract
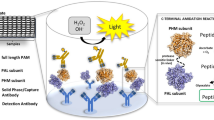
Serum carnosinase (CN-1) measurements are at present mainly performed by assessing enzyme activity. This method is time-consuming, not well suited for large series of samples and can be discordant to measurements of CN-1 protein concentrations. To overcome these limitations, we developed sandwich ELISA assays using different anti-CN-1 antibodies, i.e., ATLAS (polyclonal IgG) and RYSK173 (monoclonal IgG1). With the ATLAS-based assay, similar amounts of CN-1 were detected in serum and both EDTA and heparin plasma. The RYSKS173-based assay detected CN-1 in serum in all individuals at significantly lower concentrations compared to the ATLAS-based assay (range: 0.1–1.8 vs. 1–50 μg/ml, RYSK- vs. ATLAS-based, P < 0.01). CN-1 detection with the RYSK-based assay was increased in EDTA plasma, albeit at significantly lower concentrations compared to ATLAS. In heparin plasma, CN-1 was also poorly detected with the RYSK-based assay. Addition of DTT to serum increased the detection of CN-1 in the RYSK-based assay almost to the levels found in the ATLAS-based assay. Both ELISA assays were highly reproducible (R: 0.99, P < 0.01 and R: 0.93, P < 0.01, for the RYSK- and ATLAS-based assays, respectively). Results of the ATLAS-based assay showed a positive correlation with CN-1 activity (R: 0.62, P < 0.01), while this was not the case for the RYSK-based assay. However, there was a negative correlation between CN-1 activity and the proportion of CN-1 detected in the RYSK-based assay, i.e., CN-1 detected with the RYSK-based assay/CN-1 detected with the ATLAS-based assay × 100% (Spearman–Rang correlation coefficient: −0.6, P < 0.01), suggesting that the RYSK-based assay most likely detects a CN-1 conformation with low CN-1 activity. RYSK173 and ATLAS antibodies reacted similarly in Western blot, irrespective of PNGase treatment. Binding of RYSK173 in serum was not due to differential N-glycosylation as demonstrated by mutant CN-1 cDNA constructs. In conclusion, our study demonstrates a good correlation between enzyme activity and CN-1 protein concentration in ELISA and suggests the presence of different CN-1 conformations in serum. The relevance of these different conformations is still elusive and needs to be addressed in further studies.






Similar content being viewed by others
References
Baguet A, Reyngoudt H, Pottier A, Everaert I et al (2009) Carnosine loading and washout in human skeletal muscles. J Appl Physiol 106:837–842
Bonfanti L, Peretto P, De Marchis S, Fasolo A (1999) Carnosine-related dipeptides in the mammalian brain. Prog Neurobiol 59:333–353
De Marchis S, Modena C, Peretto P, Giffard C et al (2000) Carnosine-like immunoreactivity in the central nervous system of rats during postnatal development. J Comp Neurol 426:378–390
Derave W, Ozdemir MS, Harris RC, Pottier A et al (2007) beta-Alanine supplementation augments muscle carnosine content and attenuates fatigue during repeated isokinetic contraction bouts in trained sprinters. J Appl Physiol 103:1736–1743
Drozak J, Veiga-da-Cunha M, Vertommen D, Stroobant V et al (2010) Molecular identification of carnosine synthase as ATP-grasp domain-containing protein 1 (ATPGD1). J Biol Chem 285:9346–9356
Gardner ML, Illingworth KM, Kelleher J, Wood D (1991) Intestinal absorption of the intact peptide carnosine in man, and comparison with intestinal permeability to lactulose. J Physiol 439:411–422
Hanson HT, Smith EL (1949) Carnosinase; an enzyme of swine kidney. J Biol Chem 179:789–801
Harris RC, Tallon MJ, Dunnett M, Boobis L et al (2006) The absorption of orally supplied beta-alanine and its effect on muscle carnosine synthesis in human vastus lateralis. Amino Acids 30:279–289
Hill CA, Harris RC, Kim HJ, Harris BD et al (2007) Influence of beta-alanine supplementation on skeletal muscle carnosine concentrations and high intensity cycling capacity. Amino Acids 32:225–233
Janssen B, Hohenadel D, Brinkkoetter P, Peters V et al (2005) Carnosine as a protective factor in diabetic nephropathy: association with a leucine repeat of the carnosinase gene CNDP1. Diabetes 54:2320–2327
Lenney JF, George RP, Weiss AM, Kucera CM et al (1982) Human serum carnosinase: characterization, distinction from cellular carnosinase, and activation by cadmium. Clin Chim Acta 123:221–231
Margolis FL, Grillo M, Grannot-Reisfeld N, Farbman AI (1983) Purification, characterization and immunocytochemical localization of mouse kidney carnosinase. Biochim Biophys Acta 744:237–248
Murphey WH, Patchen L, Lindmark DG (1972) Carnosinase: a fluorometric assay and demonstration of two electrophoretic forms in human tissue extracts. Clinica Chimica Acta 42:309–314
Park YJ, Volpe SL, Decker EA (2005) Quantitation of carnosine in human plasma after dietary consumption of beef. J Agric Food Chem 53:4736–4739
Peters V, Kebbewar M, Jansen EW, Jakobs C et al (2010) Relevance of allosteric conformations and homocarnosine concentration on carnosinase activity. Amino Acids 38:1607–1615
Riedl E, Koeppel H, Brinkkoetter P, Sternik P et al (2007) A CTG polymorphism in the CNDP1 gene determines the secretion of serum carnosinase in Cos-7 transfected cells. Diabetes 56:2410–2413
Riedl E, Koeppel H, Pfister F, Peters V et al (2010) N-glycosylation of carnosinase influences protein secretion and enzyme activity: implications for hyperglycemia. Diabetes 59:1984–1990
Sauerhofer S, Yuan G, Braun GS, Deinzer M et al (2007) l-carnosine, a substrate of carnosinase-1, influences glucose metabolism. Diabetes 56:2425–2432
Stout JR, Cramer JT, Zoeller RF, Torok D et al (2007) Effects of beta-alanine supplementation on the onset of neuromuscular fatigue and ventilatory threshold in women. Amino Acids 32:381–386
Tabakman R, Lazarovici P, Kohen R (2002) Neuroprotective effects of carnosine and homocarnosine on pheochromocytoma PC12 cells exposed to ischemia. J Neurosci Res 68:463–469
Teufel M, Saudek V, Ledig JP, Bernhardt A et al (2003) Sequence identification and characterization of human carnosinase and a closely related non-specific dipeptidase. J Biol Chem 278:6521–6531
Vistoli G, Pedretti A, Cattaneo M, Aldini G et al (2006) Homology modeling of human serum carnosinase, a potential medicinal target, and MD simulations of its allosteric activation by citrate. J Med Chem 49:3269–3277
Wassif WS, Sherwood RA, Amir A, Idowu B et al (1994) Serum carnosinase activities in central nervous system disorders. Clin Chim Acta 225:57–64
Wood T (1957) Carnosine and carnosinase in rat tissue. Nature 180:39–40
Acknowledgments
This study was supported by grants from the Deutsche Forschungsgemeinschaft (Graduiertenkolleg 880 to E.R. and F.P. and DFG SA 2143/1-1 to S.J.H.) and the Deutsche Diabetes Gesellschaft (Projektförderung 2008 to E.R.).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
K. Adelmann and D. Frey contributed equally to this work.
Rights and permissions
About this article
Cite this article
Adelmann, K., Frey, D., Riedl, E. et al. Different conformational forms of serum carnosinase detected by a newly developed sandwich ELISA for the measurements of carnosinase concentrations. Amino Acids 43, 143–151 (2012). https://doi.org/10.1007/s00726-012-1244-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1244-8